Nuclear reactions Using the strong nuclear force to


















- Slides: 33

Nuclear reactions Using the strong nuclear force to produce useful energy Micro-world Macro-world Lecture 17

Strong Nuclear Force • It is very strong – It overcomes the electrical repulsion between positively charged protons that are only 10 -15 m apart. • It acts over a very short range – It is not felt by nucleons when they are more than 10 -15 m apart. • It is selective – It is felt by neutrons & protons, but not by electrons

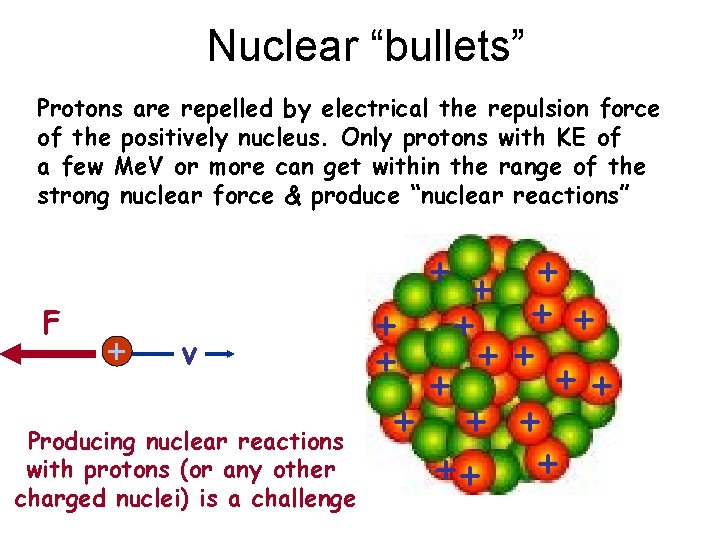
Nuclear “bullets” Protons are repelled by electrical the repulsion force of the positively nucleus. Only protons with KE of a few Me. V or more can get within the range of the strong nuclear force & produce “nuclear reactions” F + v Producing nuclear reactions with protons (or any other charged nuclei) is a challenge ++ ++ + + ++ +

Neutron induced nuclear reactions Neutrons don’t feel the electrical force so even very slow, low-energy neutrons can strike the nucleus & produce “nuclear reactions” v Low energy neutrons are effective nuclear “bullets” ++ ++ + + ++ +

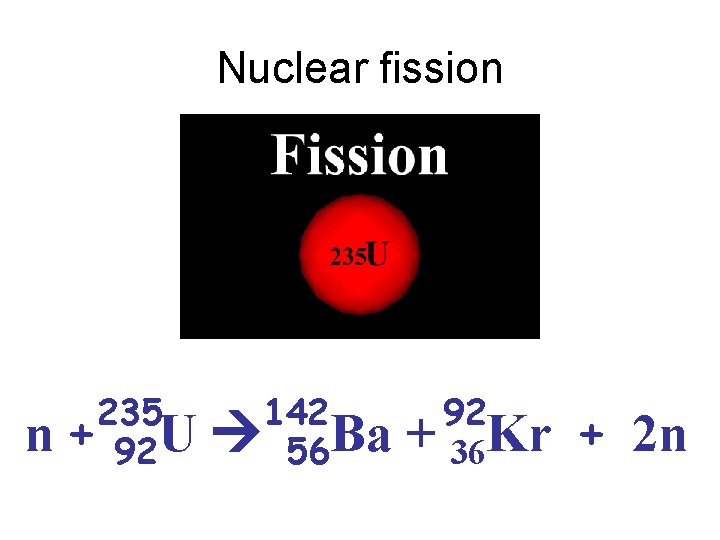
Nuclear fission 235 n + 92 U 142 56 Ba 92 + 36 Kr + 2 n

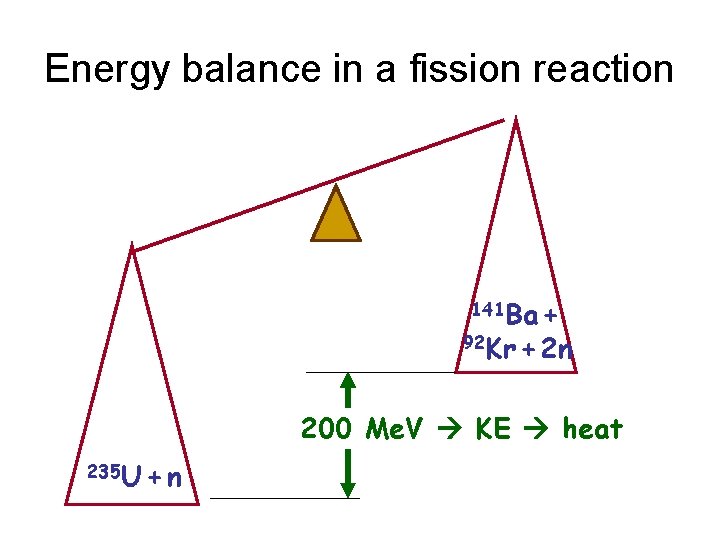
Energy balance in a fission reaction 141 Ba + 92 Kr + 2 n 200 Me. V KE heat 235 U + n

Chain reaction Use the neutrons produced by one fission to initiate another fission Enrico Fermi

Requirements for A-bomb • Fissionable material: • Critical mass • Mechanism 235 U or 239 Pu

Critical Mass Mcrit Enriched 239 Pu 235 U 50 kg 10 kg

Fissionable Material Fortunately, only certain nuclear isotopes undergo the fission process: 235 U only 0. 7% of naturally occurring U (99. 3% is 238 U, which doesn’t fission) 239 Pu doesn’t occur naturally, but is produced in nuclear reactors …. There are other fissionable isotopes, e. g. 232 Th, but they are very rare 233 U &

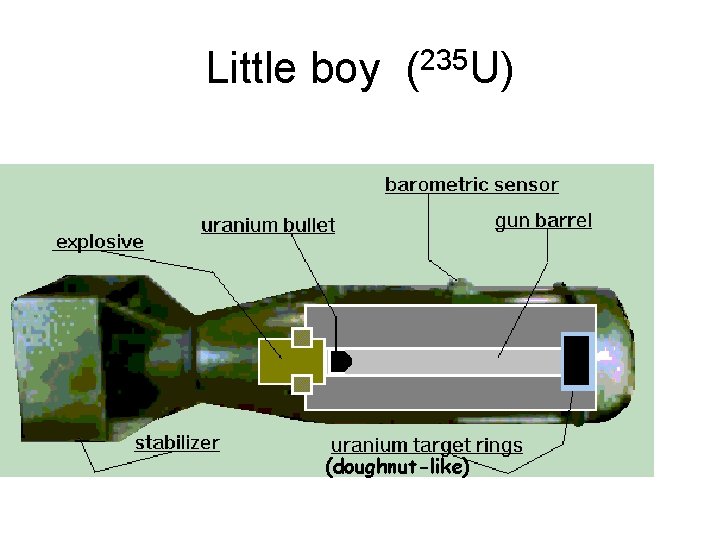
Little boy (235 U) (doughnut-like)

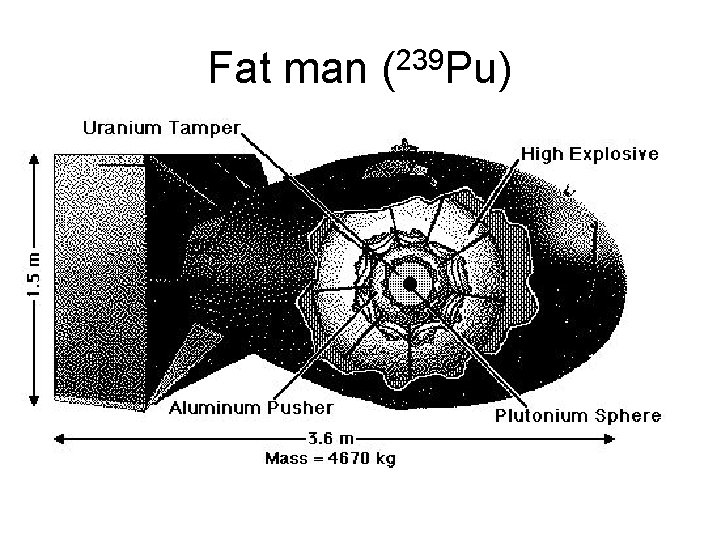
Fat man (239 Pu)

Devastation Hiroshima Aug 6 1945 8: 15 AM 80, 000 people killed immediately; ~100, 000 people were exposed to lethal radiation & died painful slow deaths

Hiroshima aftermath

Devastation Nagasaki Aug 9 1945 10: 45 AM 39, 000 people killed immediately; ~70, 000 people were exposed to lethal radiation & died painful slow deaths

Nagasaki aftermath

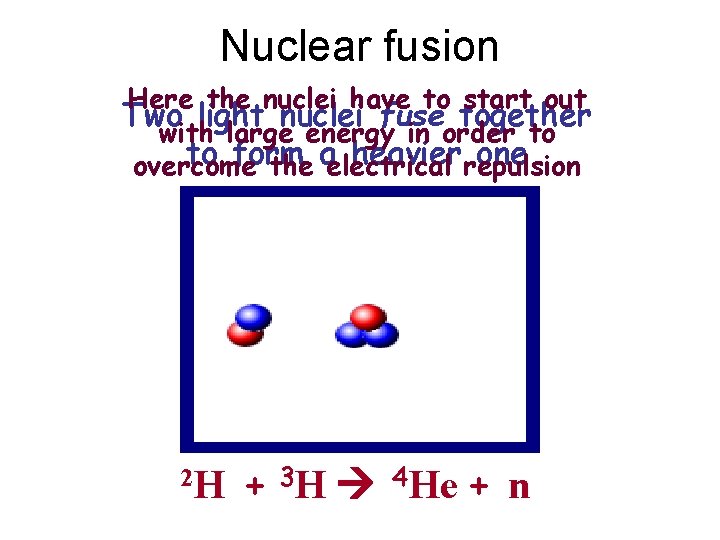
Nuclear fusion Here the nuclei have to start out Two light nuclei fuse together with large energy in order to to form heavierrepulsion one overcome the aelectrical 2 H + 3 H 4 He + n

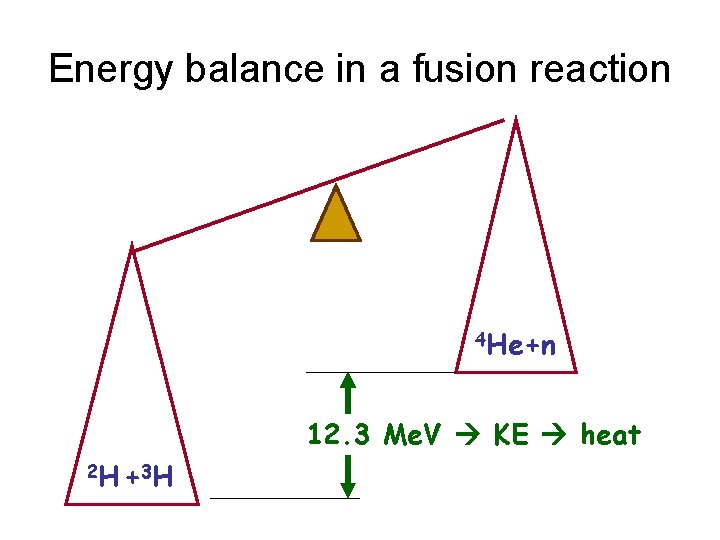
Energy balance in a fusion reaction 4 He+n 12. 3 Me. V KE heat 2 H + 3 H

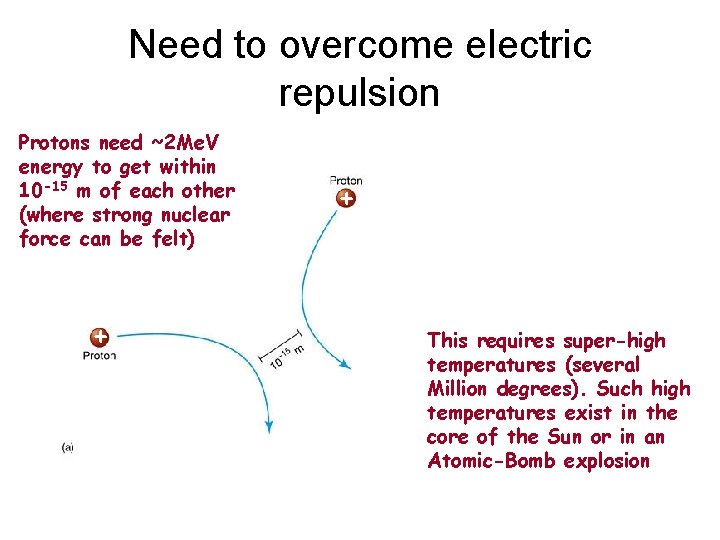
Need to overcome electric repulsion Protons need ~2 Me. V energy to get within 10 -15 m of each other (where strong nuclear force can be felt) + + This requires super-high temperatures (several Million degrees). Such high temperatures exist in the core of the Sun or in an Atomic-Bomb explosion

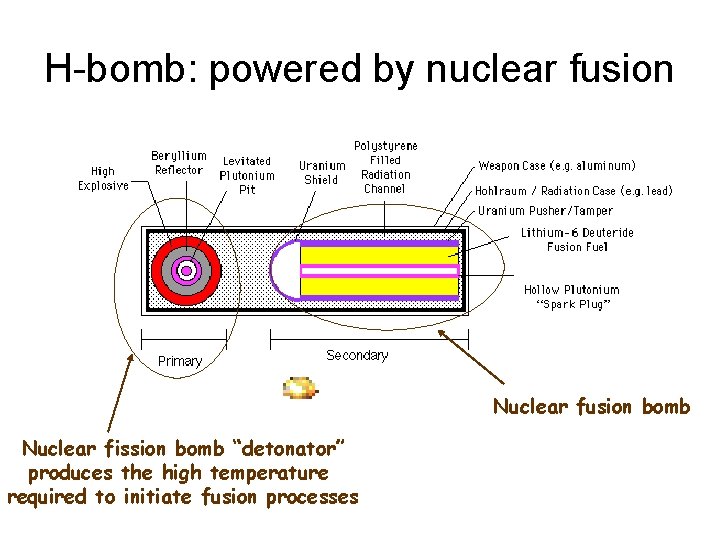
H-bomb: powered by nuclear fusion Nuclear fusion bomb Nuclear fission bomb “detonator” produces the high temperature required to initiate fusion processes

Brighter than 1000 suns 1000 times the power of an A-bomb!!

Dangers of teaching nuclear physics Oh, and I suppose it was me who said ‘what harm could it be to give the chickens a book on nuclear physics? ’

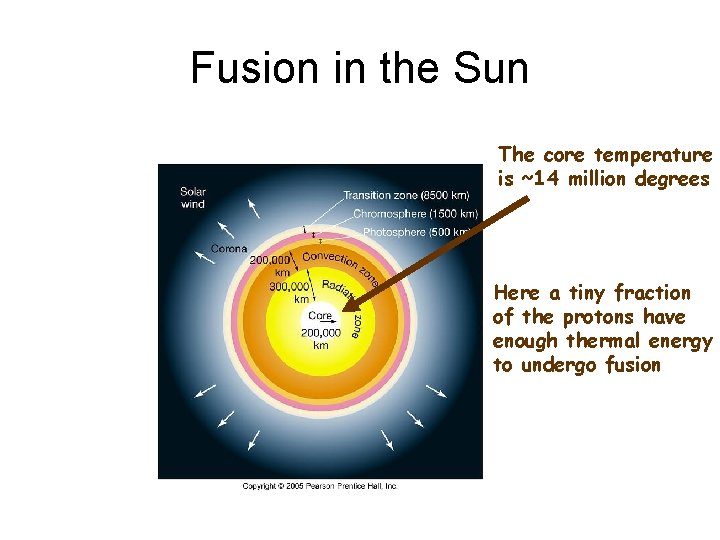
Fusion in the Sun The core temperature is ~14 million degrees Here a tiny fraction of the protons have enough thermal energy to undergo fusion

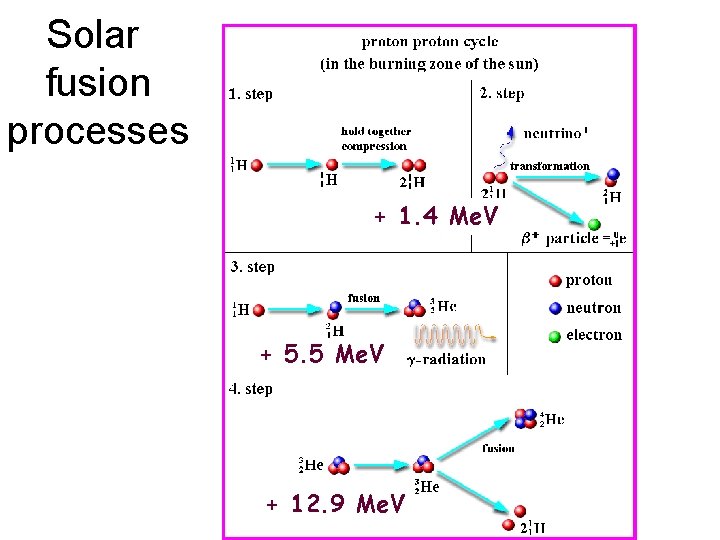
Solar fusion processes + 1. 4 Me. V + 5. 5 Me. V + 12. 9 Me. V

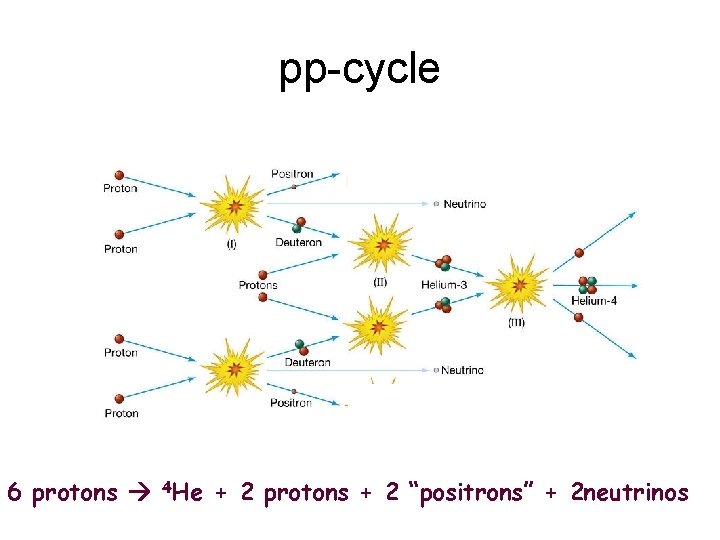
pp-cycle 6 protons 4 He + 2 protons + 2 “positrons” + 2 neutrinos

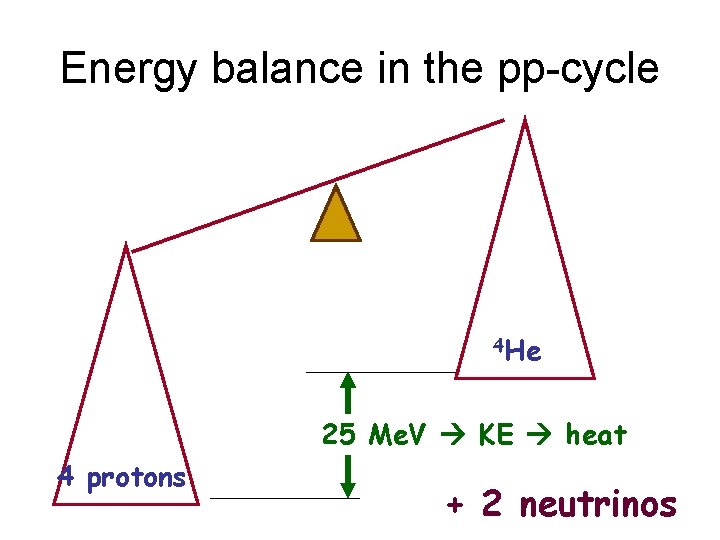
Energy balance in the pp-cycle 4 He 25 Me. V KE heat 4 protons + 2 neutrinos

How do we know what goes on inside the Sun?

Superkamiokande

Superkamiokande

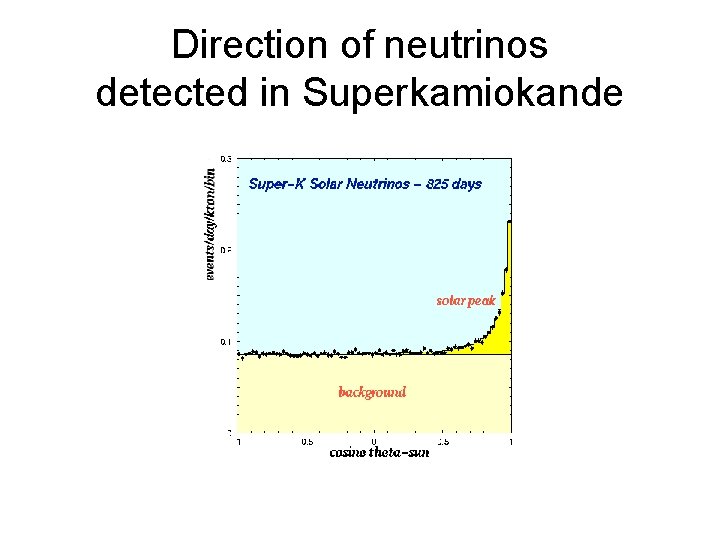
Direction of neutrinos detected in Superkamiokande

Sun as seen by a neutrino detector

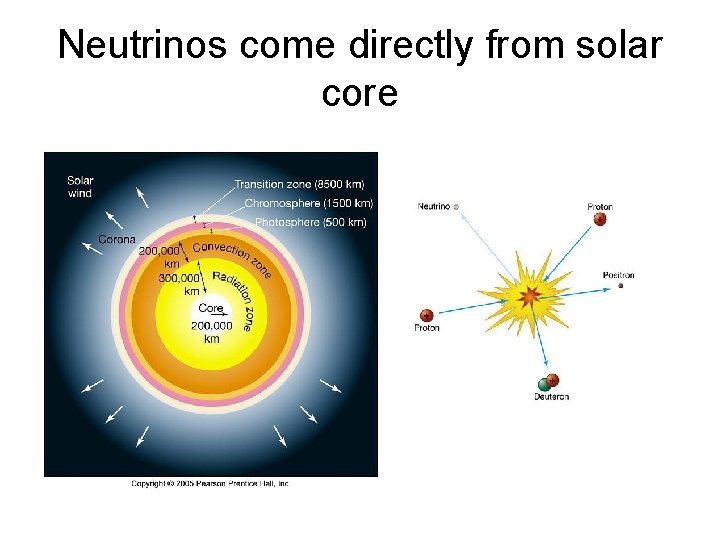
Neutrinos come directly from solar core

Neutrinos are everywhere Ttest