Table of Contents Chapter 19 Acids and Bases

Table of Contents Chapter 19: Acids and Bases 19. 1: Acids and Bases 19. 2: Strengths of Acids and Bases 19. 3: What is p. H? 19. 4: Neutralization
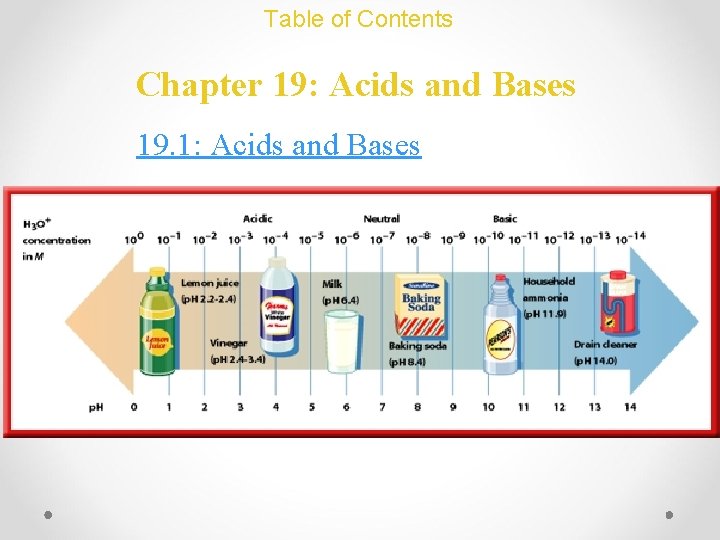
Table of Contents Chapter 19: Acids and Bases 19. 1: Acids and Bases

Acids and Bases: Basic Concepts Properties of Acids and Bases ACIDS • sour • Turn litmus red • React with & corrodes metals • Lemon juice, vinegar, etc. BASES • • bitter slippery Turns litmus blue Don’t react with metals • Soaps, etc.

Acids and Bases: Basic Concepts The Arrhenius model • Acids • H is the first element in a formula • Ionizes in aqueous solution to form H+ ions • Increases the concentration H+ ions • Also can produce hydronium ion, H 3 O+.

Acids and Bases: Basic Concepts The Arrhenius model • Hydrolysis: HCl dissolves in water, produces hydronium ions • In an acid, any hydrogen atom that can be transferred to water is called an acidic hydrogen.

Acids and Bases: Basic Concepts Monoprotic Acids • Monoprotic acids contain only one acidic hydrogen.

Acids and Bases: Basic Concepts Diprotic and Triprotic Acids • Polyprotic acids: contains several acidic hydrogens. • Diprotic acids: contain two acidic hydrogens. • Triprotic acids: contain three acidic hydrogens.

Acids and Bases: Basic Concepts Arrhenius Bases – • Base: produces hydroxide ions (OH ) when it dissolves in water. • The simplest kind: water-soluble ionic compound (like Na. OH) that contains the hydroxide ion as the negative ion. • Dissociates in solution to release OH-

Table of Contents Chapter 19: Acids and Bases 19. 2: Strengths of Acids and Bases

Acids and Bases: Basic Concepts Strength Is Not Concentration • weak and strong: compare the strengths of acids and bases • dilute and concentrated: describe the concentration of solutions. • The combination of strength and concentration ultimately determines the behavior of the solution.

Acids and Bases: Basic Concepts Strong Bases & Acids • Strong Acids and Bases • Completely dissociates in water • Completely breaks into ions Mg(OH)2(aq)→ Mg 2+(aq) + 2 OH–(aq) HCl(aq)→ H+(aq) + Cl–(aq)

List of strong acids and bases • • • HCl – hydrochloric acid HBr - hydrobromic acid HI – hydroiodic acid HNO 3 – nitric acid H 2 SO 4 – sulfuric acid HCl. O 4 – perchloric acid • Groups 1 & 2 o Na. OH o KOH o Li. OH o Rb. OH o Cs. OH o Ca(OH)2 o Ba(OH)2 o Sr(OH)2

Acids and Bases: Basic Concepts Weak Bases & Acids • Weak Acids and Bases • Reaches equilibrium • Most molecules don’t react with water to break into ions • Acetic acid (HC 2 H 3 O 2); HCHO 2 • Ammonia (NH 3); Al(OH)3; Fe(OH)3

Acids and Bases: Basic Concepts Weak Acids • The concentration of the un-ionized acid is always the greatest of the three concentrations. (Comparing strong acids and weak acids)

Acids and Bases: Basic Concepts A Broader Definition of Acids and Bases • Brønsted-Lowry model • Acid = a proton (hydrogen-ion) donor • When a Brønsted-Lowry acid donates a hydrogen ion, a conjugate base is formed. • Base = a proton (hydrogen-ion) acceptor • When a Brønsted-Lowry base accepts a hydrogen ion, a conjugate acid is formed.

Acids and Bases: Basic Concepts A Broader Definition of Acids and Bases • Conjugate acid-base pair: Two substances related to each other by the donating and accepting of a single hydrogen ion. strong weak Acid – conjugate base Base – conjugate acid strong weak
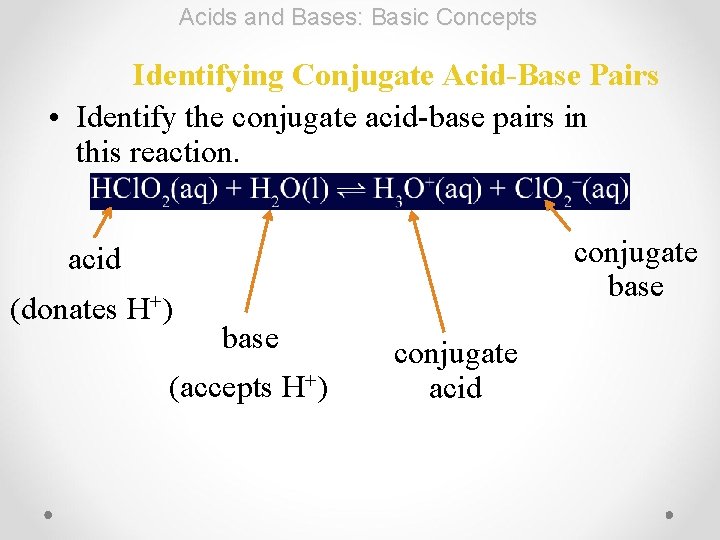
Acids and Bases: Basic Concepts Identifying Conjugate Acid-Base Pairs • Identify the conjugate acid-base pairs in this reaction. conjugate base acid (donates H+) base (accepts H+) conjugate acid

PRACTICE identifying CA & CB • NH 4+(aq) + OH-(aq) acid base NH 3(aq) + H 2 O(l) • CO 32 -(aq) + H 2 O(l) HCO 3 -(aq) +OH-(aq) base acid • HSO 4 -(aq) + H 2 O(l) acid base CB CA CA CB H 3 O+(aq) +SO 42 -(aq) CA CB Fin de la première journée

Fin de la ère 1 journée

Table of Contents Chapter 19: Acids and Bases 19. 3: What is p. H?

Acids and Bases: Basic Concepts Starter • Define Arrhenius and Brønsted-Lowry acids and bases • What is the difference between weak and strong acids and bases? • What are some characteristics of acids? bases? Today, you’ll need a scientific/graphing calculator!

Acids and Bases: Basic Concepts The ion product constant for water • Self-ionization of water: H 2 O(l) 2 H 2 O(l) H+(aq) + OH-(aq) H 3 O+(aq) + OH-(aq) Kw = [H+][OH-] = 1. 0 x 10– 14 • Just “plug and chug” to solve for concentrations of either ion
![Acids and Bases: Basic Concepts The ion product constant for water • When [H+] Acids and Bases: Basic Concepts The ion product constant for water • When [H+]](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-23.jpg)
Acids and Bases: Basic Concepts The ion product constant for water • When [H+] > [OH-], the solution is acidic • When [H+] < [OH-], the solution is basic • When [H+] = [OH-], the solution is neutral
![Practice finding [OH ] + [H ] & • The concentration of either the Practice finding [OH ] + [H ] & • The concentration of either the](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-24.jpg)
Practice finding [OH ] + [H ] & • The concentration of either the H+ ion or OH- ion is given. For each solution, calculate [H+] or [OH-]. State whether the solution is acidic, basic, or neutral. 1. [H+] = 1. 0 x 10 -5 M 2. [H+] = 1. 0 x 10 -13 M 3. [OH-] = 1. 0 x 10 -7 M 4. [OH-] = 1. 0 x 10 -3 M [OH-] = 1. 0 x 10 -9 M, acidic [OH-] = 1. 0 x 10 -1 M, basic [H+] = 1. 0 x 10 -7 M, neutral [H+] = 1. 0 x 10 -11 M, basic

Acids and Bases: Basic Concepts Measuring p. H • p. H measures acidity. • Indicators register different colors at different p. Hs. • p. H meters are instruments that measure the exact p. H of a solution.

Acids and Bases: Basic Concepts The p. H Scale p. H: • A mathematical scale • Measures concentration of hydronium (H 3 O+) ions in a solution • Range: 0 to 14

Acids and Bases: Basic Concepts Interpreting the p. H Scale • p. H = 7 is neutral. • p. H < 7 is acidic. As p. H drops from 7, it becomes more acidic. • p. H > 7 is basic. As p. H increases from 7, the solution becomes more basic.
![Acids and Bases: Basic Concepts The p. H Scale [H+] – 11 = 1 Acids and Bases: Basic Concepts The p. H Scale [H+] – 11 = 1](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-28.jpg)
Acids and Bases: Basic Concepts The p. H Scale [H+] – 11 = 1 x 10 M – 4 [H 3 O+ ] = 1 x 10 M [H 3 O+ ] – 4 = 1. 1 x 10 M p. H =11 p. H = 4 p. H = 3. 96

Acids and Bases: Additional Concepts p. H and p. OH • p. H scale: expresses the acidity of a solution. • p. OH scale: expresses the basicity of a solution.

Acids and Bases: Additional Concepts p. H and p. OH • The p. H and p. OH values for a solution may be – + determined if either [H ] or [OH ] is known.
![Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] • Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] •](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-31.jpg)
Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] • EXAMPLE: If a carbonated soft drink has a – 4 hydrogen ion concentration of 7. 3 x 10 M, what are the p. H and p. OH of the soft drink? Known: [H+] Calculate p. H
![Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] The Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] The](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-32.jpg)
Acids and Bases: Additional Concepts CALCULATING p. H and p. OH from [H+] The carbonated soft drink is acidic.

Practice calculating p. H and p. OH • Calculate the p. H and p. OH of aqueous solutions having the following ion concentrations. 1. [OH-] = 1. 0 x 10 -6 M 2. [OH-] = 6. 5 x 10 -4 M 3. [H+] = 3. 6 x 10 -9 M 4. [H+-] = 0. 025 M p. OH = 6. 00, p. H = 8. 00 p. OH = 3. 19, p. H = 10. 81 p. H = 8. 44, p. OH = 5. 56 p. H = 1. 60, p. OH = 12. 40

Fin de la ème 2 journée • On Web. Assign Ch 19, SKIP 3, 11, 18, 20, 21 • We will correct Ch 19 practice problems 1 -12 tomorrow at the beginning of class
![p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p. p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p.](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-35.jpg)
p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p. OH=-log[OH-] [H+] 1. 0 x 10 -14 = [H+][OH-]

WARM-UP • Calculate the p. H and p. OH of aqueous solutions having the following ion concentrations. o [OH-] = 6. 5 x 10 -4 M p. OH = 3. 19, p. H = 10. 81 p. H = 1. 60, p. OH = 12. 40 o [H+-] = 0. 025 M The concentration of either the H+ ion or OH- ion is given. For each solution, calculate [H+] or [OH-]. State whether the solution is acidic, basic, or neutral. [OH-] = 1. 0 x 10 -9 M, acidic [H+] = 1. 0 x 10 -5 M [H+] = 1. 0 x 10 -11 M, basic [OH-] = 1. 0 x 10 -3 M
![Acids and Bases: Additional Concepts CALCLUATING ion concentrations from p. H + [H ] Acids and Bases: Additional Concepts CALCLUATING ion concentrations from p. H + [H ]](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-37.jpg)
Acids and Bases: Additional Concepts CALCLUATING ion concentrations from p. H + [H ] = -p. H 10 [OH-] = 10 -p. OH
![Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-38.jpg)
Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H • What are [H+] and [OH–] in a solution with a p. H of 9. 70? • Find [H+]. + [H ] = -p. H 10 = 10 -9. 70 – 10 + [H ] = 2. 0 x 10 M
![Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-39.jpg)
Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H – + • What are [H ] and [OH ] in a solution with a p. H of 9. 70? • Determine p. OH
![Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-40.jpg)
Acids and Bases: Additional Concepts [H+] – Calculating and [OH ] from p. H – + • What are [H ] and [OH ] in a solution with a p. H of 9. 70? – • Find [OH ] = -p. OH 10 = 10 -4. 30 As expected, [OH–] > [H+] in this basic solution.

Practice finding ion concentrations • The p. H is given for three solutions. Calculate [H+] and [OH-] in each solution. 1. p. H = 2. 37 [H+] =4. 3 x 10 -3 M [OH-]=2. 3 x 10 -12 M 2. p. H = 11. 05 [H+] = 8. 9 x 10 -12 M [OH-]=1. 1 x 10 -3 M 3. p. H = 6. 50 [H+] =3. 2 x 10 -7 M [OH-]=3. 2 x 10 -8 M

Acids and Bases: Additional Concepts CALCLUATING p. H of strong acids and bases • Remember, strong acids and bases completely break into their ions in water HCl(aq)→ H+ For every HCl molecule, 1 [H+] = [HCl] – Cl (aq) + H+ ion is produced. So, Mg(OH)2(aq)→ Mg 2+(aq) + 2 OH–(aq) For every Mg(OH)2 molecule, 2 OH- ions are produced. So, [Mg(OH)2] = 2[OH-]

Practice calculating p. H of SA & SB • Calculate the p. H of the following solutions. 1) 1. 0 mol/L HI 2) 0. 050 mol/L HNO 3 3) 1. 0 M KOH 4) 2. 4 x 10 -5 M Mg(OH)2 p. H = 0. 00 p. H = 1. 30 p. H = 14. 00 p. H = 9. 68
![p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p. p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p.](http://slidetodoc.com/presentation_image_h/7e0291c43dd97caff8327f78bb2a9ca6/image-44.jpg)
p. H + p. OH = 14 p. H p. OH p. H=-log[H+] p. OH=-log[OH-] [H+]=10 -p. H [H+] [OH-]=10 -p. OH 1. 0 x 10 -14 = [H+][OH-]

Fin de la ème 3 journée • We will correct and turn in Ch 19 practice problems 13 -26 tomorrow at 1: 40 • Web. Assign Ch 19 is due Thursday, 17 May • On Web. Assign Ch 19, SKIP 3, 11, 18, 20, 21

Table of Contents Chapter 19: Acids and Bases 19. 4: Neutralization

Acids and Bases: Basic Concepts Types of Acid-Base Reactions • The reaction of an acid and a base is called a neutralization reaction. Strong acid + strong base → salt + water

Acids and Bases: Additional Concepts Acid-Base Titrations • Acid-base titration: The process of determining the molarity of an acid or a base through the use of an acid-base reaction. • Reactant: known molarity + unknown molarity → • Standard solutions: Solutions of known molarity used in titrations. • The known reactant molarity is used to find the unknown molarity of the other solution.

Acids and Bases: Additional Concepts Acid-Base Titrations • Indicator: A substance used to “locate” the equivalence point of a titration. (endpoint – color change) • Equivalence point: the point in the reaction where stoichiometrically equal amounts of acid and base have been added • For weak base-strong acid reaction, mixing equal moles of acid and base does not produce a neutral solution.
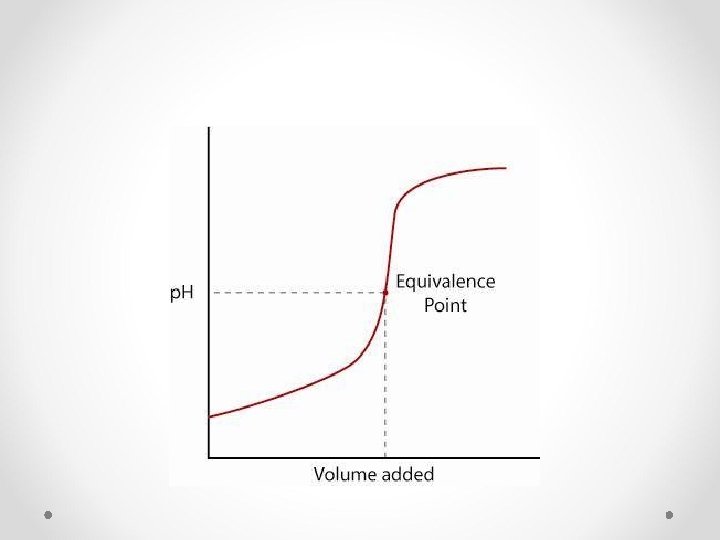

Acids and Bases: Additional Concepts Acid-Base Titrations • Titration/Neutralization calculations Mbase. Vbase=Macid. Vacid Plug and Chug!

Acids and Bases: Additional Concepts Determining Concentration: Using Stoichiometry • EXAMPLE: A 0. 100 M Li. OH solution was used to titrate an HBr solution of unknown concentration. At the endpoint, 21. 0 m. L of Li. OH solution had neutralized 10. 0 m. L of HBr. What is the molarity of the HBr solution? Mb. Vb = Ma. Va (0. 100 M)(21. 0 m. L) = (? M)(10. 0 m. L) 0. 210 M HBr

Practice Titrations 1. A 0. 350 M Li. OH solution was used to titrate an HBr solution of unknown concentration. At the endpoint, 75. 0 m. L of Li. OH solution had neutralized 25. 0 m. L of HBr. What is the molarity of the HBr solution? 1. 05 M HBr 2. 100 m. L of 0. 75 M HCl is used to titrate 150 m. L Na. OH. What is the concentration of Na. OH? 0. 5 M Na. OH

Acids and Bases: Additional Concepts Buffers Click box to view movie clip. 1. How was the buffer made? 2. Which turned basic first, the acid or the buffer? 3. What is the definition of a buffer?

Acids and Bases: Additional Concepts Buffers Defined • Buffer: a solution that resists changes in p. H. • Buffer solutions are prepared by using a weak acid or base with one of its salts (its conjugate base/conjugate acid) • When you add base to an acidic buffer, the base will react with the salt – not the acid, maintaining p. H.

End Ch. 19 • We will correct and turn in Ch 19 practice problems TODAY at 1: 40. • Web. Assign Ch 19 is due Thursday, 17 May

Universal Indicator

Acids and Bases: Basic Concepts Types of Acid-Base Reactions • The reaction of an acid and a base is called a neutralization reaction. Click box to view movie clip.

Acids and Bases: Basic Concepts Neutralization Reactions Strong acid + strong base → salt + water Click box to view movie clip.

Acids and Bases: Additional Concepts Acid-Base Titrations • Acid-base titration: The process of determining the molarity of an acid or a base through the use of an acid-base reaction. Click box to view movie clip.
- Slides: 60