Chapters 14 15 Acids and Bases AcidBase Equilibrium

Chapters 14 & 15 Acids and Bases Acid-Base Equilibrium

History of Acids and Bases In the early days of chemistry chemists were organizing physical and chemical properties of substances. They discovered that many substances could be placed in two different property categories: Substance B Substance A 1. Bitter taste 1. Sour taste 2. Reacts with carbonates to make CO 2 2. 3. Reacts with metals to produce H 2 3. Do not react with metals 4. Turns blue litmus pink 4. Turns red litmus blue 5. Reacts with B substances to make salt water 5. Reacts with A substances make salt and water Reacts with fats to make soaps Arrhenius was the first person to suggest a reason why substances are in A or B due to their ionization in water.

Arrhenius Theory The Swedish chemist Svante Arrhenius proposed the first definition of acids and bases. (Substances A and B became known as acids and bases) According to the Arrhenius model: “acids are substances that dissociate in water to produce H+ ions and bases are substances that dissociate in water to produce OH- ions” Na. OH (aq) Na+ (aq) + OH- (aq) HCl (aq) H+ (aq) + Cl- (aq) Base Acid
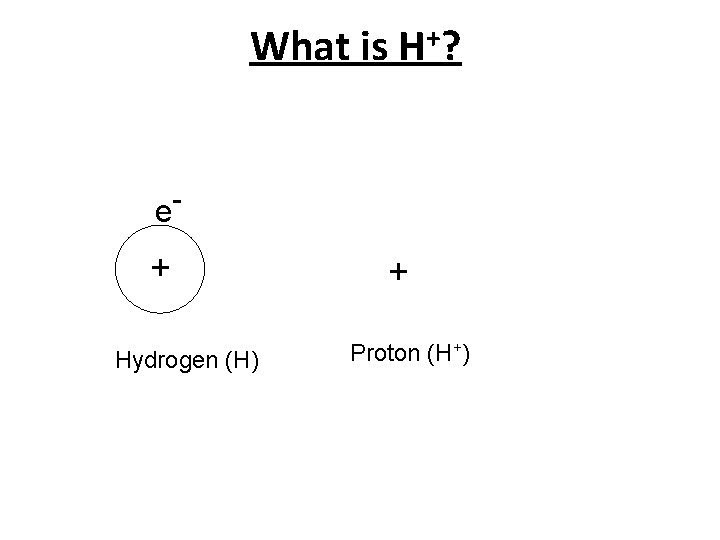
What is H+? e- + Hydrogen (H) + Proton (H+)

Hydronium Ion Unknown to Arrhenius free H+ ions do not exist in water. They covalently react with water to produce hydronium ions, H 3 O+. or: H+ (aq) + H 2 O (l) H 3 O+ (aq) This new bond is called a coordinate covalent bond since both new bonding electrons come from the same atom

Hydronium Ion Hydronium ion is the name for H 3 O+ and is often times abbreviated as H+ (aq) they both mean the same thing. What is the difference between a strong acid and a weak acid? Strong acids ionize 100% and weak ones do not! A single arrow is used to represent the ionization of a strong acid. Double arrows (Equilibrium) are used to represent weak acids. For example: HCl (g) H+ (aq) + Cl - (aq) HF (g) H+ (aq) + F - According to Arrhenius, is water an acid or base? HOH (l) H+ (aq) + OH – (aq) Neither, he called it Neutral (same amount of OH- and H+

Strong Acids and Bases How can we identify strong acids or bases? Easy, memorize them! Memorized Strong Acids 1. HCl. O 4 2. H 2 SO 4 3. HI 4. HBr 5. HCl 6. HNO 3 Memorized Strong Bases Hydroxides of group 1 and 2 metals, excluding Be and Mg

Bronsted Lowry Theory Johannes Brønsted and Thomas Lowry revised Arrhenius’s acid-base theory to include this behavior. They defined acids and bases as follows: Bronsted Lowry “An acid is a hydrogen containing species that donates a proton. A base is any substance that accepts a proton” HCl (aq) + H 2 O (l) Cl- (aq) + H 3 O+ (aq) In the above example what is the Brønsted acid? What is the Brønsted base?

Bronsted Lowry Theory In reality, the reaction of HCl with H 2 O is an equilibrium and occurs in both directions, although in this case the equilibrium lies far to the right. HCl (aq) + H 2 O (l) Cl - ( aq) + H 3 O+ (aq) For the reverse reaction Cl - behaves as a Brønsted base and H 3 O+ behaves as a Brønsted acid. The Cl- is called the conjugate base of HCl. Brønsted acids and bases always exist as conjugate acid-base pairs. Their formulas differ by only one proton.
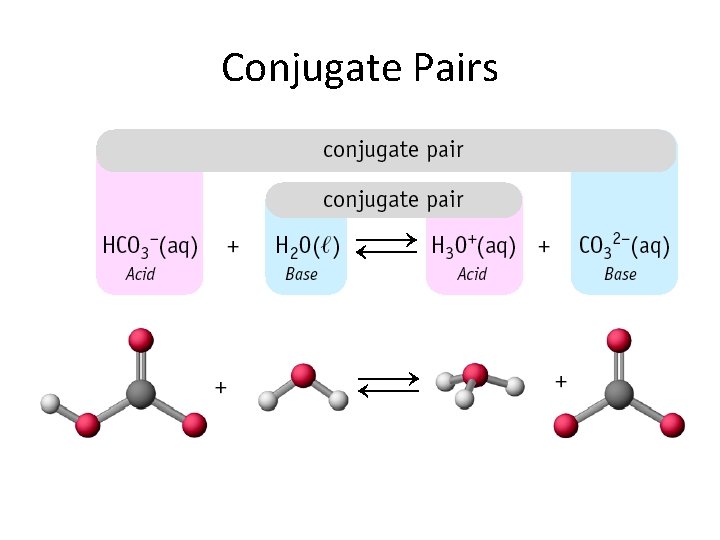
Conjugate Pairs

Give it a Try ! Label the acid, base, conjugate acid, and conjugate base in each reaction: HCl + OH- → Cl- + H 2 O + H 2 SO 4 → HSO 4 - + H 3 O+

Autoionization of Water In pure water (no solute) water molecules behave as both an acid and base!! It is called amphoteric meaning it will act as either an acid or a base depending on the situation. e. g. H 2 O (l) + H 2 O (l) H 3 O+ (aq) + OH- (aq) This is called the self-ionization (autoionizaion) of water. Although the equilibrium lies far to the left it is very important to take into consideration, especially for living systems.

Autoionization of Water The auto-ionization of water is described by the equation: H 2 O (l) + H 2 O (l) H 3 O+ (aq) + OH- (aq) • This equilibrium lies very much to the left i. e. mostly water. • For pure water [OH-] = [H+]

Autoionization of Water Kw is called the ion product of water. What is the value for the ion product of water? Kw = [H+][OH-] =(1 x 10 -7 M) = 1. 0 x 10 -14 M
![Autoionization of Water We define an aqueous solution as being neutral when the [H+] Autoionization of Water We define an aqueous solution as being neutral when the [H+]](http://slidetodoc.com/presentation_image/1e3c470f7acecf7942385078d990b942/image-15.jpg)
Autoionization of Water We define an aqueous solution as being neutral when the [H+] = [OH-]. We define an aqueous solution as being acidic when [H+] > [OH-]. We define an aqueous solution as being basic when [H+] < [OH-]. However, in each case Kw = 1 x 10 -14 M 2 [H+] = 0. 0000001 = 10 -7 (how can this be abbreviated further? ) By just describing the power Called the power of H, or p. H = 7
![Calculations To calculate p. H or p. OH p. H = -log [H+], or Calculations To calculate p. H or p. OH p. H = -log [H+], or](http://slidetodoc.com/presentation_image/1e3c470f7acecf7942385078d990b942/image-16.jpg)
Calculations To calculate p. H or p. OH p. H = -log [H+], or p. OH = -log [OH-] p. H + p. OH = 14 for water solutions To find the Molarity Concentrations 2 nd log = 10 – p. H or 2 nd log = 10 –p. OH The number of decimal places in the log answer is equal to the number of sig figs in the original Molar [ ]

Find the p. H of these: 1) 0. 15 M solution of Hydrochloric acid 2) 3. 00 X 10 -7 M solution of Nitric acid

Practice calculating Molarity A solution has a p. H of 8. 5. What is the Molarity of hydrogen ions in the solution?

A p. H Number line The p. H scale is a way of expressing the strength of acids and bases. [H+] =10 -14 p. H = 14 [OH -] = 100 basic [H+] < [OH -] basic p. H = 12 [H+] =10 -12 [OH -] = 10 -2 p. H = 7 [H+] = 10 -7 [OH -] = 10 -7 [H+] = [OH -] neutral p. H = 2 [H+] = 10 -2 [OH -] = 10 -12 [H+] > [OH -] acidic [H+] < [OH -] basic

Equilibrium Involving Acids and Bases • Strong Acids and Strong Bases completely dissociate in water. Therefore they DO NOT create an equilibrium system. • Weak Acids or weak bases disassociate to a far lesser degree in an aqueous solution. They will create an equilibrium system. • They have their own Kc constants.

Equilibria Involving Weak Acids and Bases Consider acetic acid, HC 2 H 3 O 2 + H 2 O → H 3 O+ + C 2 H 3 O 2 Acid Conj. Base Ka = [H 30+] [ C 2 H 3 O 2] = 1. 85 x 10 -5 [HC 2 H 3 O 2] K is designated Ka ACID Dissociation constant K gives the ratio of ions to molecules The[H 20] is not shown in the Ka constant The greater the dissociation, the larger the Ka will be.

Equilibrium Constants for Weak Acids Weak acid has Ka < 1 Leads to small [H 3 O+] and a p. H of 2 - 7


Equilibrium Constants for Weak Bases Weak base has Kb < 1 Leads to small [OH-] and a p. H of 12 - 7
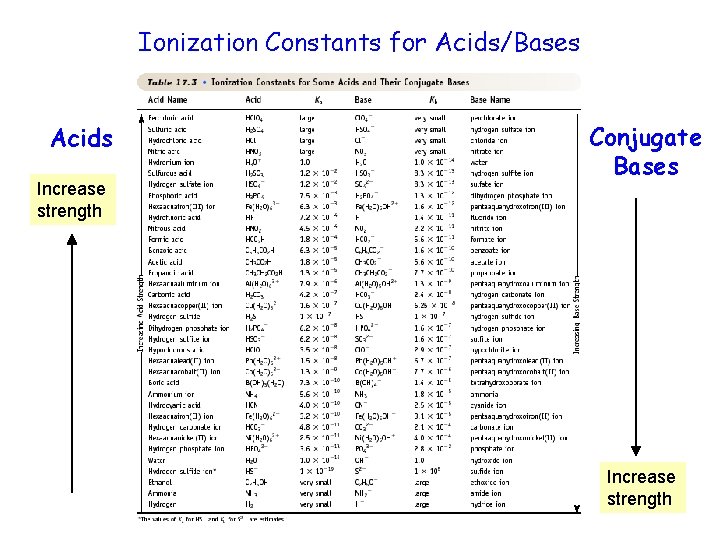
Ionization Constants for Acids/Bases Acids Increase strength Conjugate Bases Increase strength
![Relation of Ka, Kb, [H 3 O+] and p. H Relation of Ka, Kb, [H 3 O+] and p. H](http://slidetodoc.com/presentation_image/1e3c470f7acecf7942385078d990b942/image-26.jpg)
Relation of Ka, Kb, [H 3 O+] and p. H

Solving Weak Acid Equilibrium Problems 1. List the major species in the solution. 2. Choose the species that can produce H+, and write balanced equations for the reactions producing H+. 3. Using the values of the equilibrium constants for the reactions you have written, decide which equilibrium will dominate in producing H+. 4. Write the equilibrium expression for the dominant equilibrium. Copyright © Cengage Learning. All rights reserved 27

Solving Weak Acid Equilibrium Problems 5. List the initial concentrations of the species participating in the dominant equilibrium. 6. Define the change needed to achieve equilibrium; that is, define x. 7. Write the equilibrium concentrations in terms of x. 8. Substitute the equilibrium concentrations into the equilibrium expression. Copyright © Cengage Learning. All rights reserved 28

Solving Weak Acid Equilibrium Problems 9. Solve for x the “easy” way, that is, by assuming that [HA]0 – x about equals [HA]0. 10. Use the 5% rule to verify whether the approximation is valid. 11. Calculate [H+] and p. H. Copyright © Cengage Learning. All rights reserved 29

Section 14. 5 Calculating the p. H of Weak Acid Solutions EXERCISE! Calculate the p. H of a 0. 50 M aqueous solution of the weak acid HF. Calculate the % dissociation (Ka = 7. 2 × 10– 4) Copyright © Cengage Learning. All rights reserved 30

Section 14. 5 Calculating the p. H of Weak Acid Solutions Steps Toward Solving for p. H HF(aq) + H 2 O Initial H 3 O+(aq) + F–(aq) 0. 50 M ~0 ~0 –x +x +x 0. 50–x x x Change Equilibrium Copyright © Cengage Learning. All rights reserved 31

Section 14. 5 Calculating the p. H of Weak Acid Solutions Percent Dissociation (Ionization) ▪ For a given weak acid, the percent dissociation increases as the acid becomes more dilute. Copyright © Cengage Learning. All rights reserved 32

Neutralization Reactions ● When an acid and a base mix, the acid will donate protons to the base in what is called a neutralization reaction. ● There are four different ways that this can occur and it depends on the relative strengths of the acids and bases involved. ● The final p. H of the solution will depend on whether the excess ions at equilibrium are due to a weak acid or base combination or a strong acid and base combination.

Mechanisms for Acid/Base Neutralizations # 1 STRONG ACID AND STRONG BASE ● The only important ions are the H+ and OH-. All others are spectators. ● Water and a salt will always be created. ● H 3 O+ (aq) + OH- (aq) ● Reaction proceeds far to the right. ● p. H = 7 H 2 O(l) + Salt

# 2 STRONG ACID AND WEAK BASE ● The strong acid dissociates completely. ● The product will be the conjugate acid of the weak base. ● Net ionic equation involves the proton transfer from the strong acid to the weak base. ● H 3 O+ (aq) + B (aq) ● Kn = Kb X 1_ Kw H 2 O(l) + BH+ (aq)

#3 WEAK ACID AND STRONG BASE ● The net ionic equation involves proton transfer from HA (weak acid) to the strong base, OH● The products are the conjugate base of the weak acid and water. ● HA(aq) + OH- (aq) ● Kn = Ka X 1_ Kw H 2 O + A- (aq)

# 4 WEAK ACID AND WEAK BASE ● The solution contains both cations and anions and the p. H is dependent on the relative numbers of each produced. ● Less tendency to proceed to completion than neutralizations involving strong acids and strong bases. ● HA (aq) + B (aq) ● Kn = (Ka)(Kb) (1 / Kw) BH+ (aq) + A- (aq)

Summary of Steps for Strong Acid and Strong Base Titrations 1. Write out the equation 2. Determine the moles of each reactant 3. Create an ICE table and determine the limiting reactant and the excess reactant. 4. Determine the [H 3 O+] of the excess reactant 5. Use the formula: p. H = -log[H 3 O+] to determine p. H

Calculate the p. H of a solution whenever weak bases or weak acids are involved Use the Henderson-Hasselbalch Equation for acids ● p. Ka is found by taking the - log of Ka ● A- is the conjugate base of the corresponding weak acid HA

Calculate the p. H of a solution whenever weak bases or weak acids are involved Use the Henderson-Hasselbalch Equation for bases ● p. Kb is found by taking the - log of Kb ● BH+ is the conjugate acid of the corresponding weak base B

The Henderson-Hasselbalch Equation Find the p. H of a mixture of : 100 ml of 0. 3 NH 3 and 180 ml of 0. 1 M HCl Kb NH 3 = 1. 8 x 10 -5

The Henderson-Hasselbalch Equation 25. 0 ml of 1. 0 M HCl is mixed with 60. 0 ml of 0. 50 M pyridine C 5 H 5 N, a weak base, Kb = 1. 5 x 10 -9, determine the p. H of the solution.

Summary of Steps for Weak Acid and Weak Base Titrations 1. Write out the equation 1. Determine the moles of each reactant 1. Create an ICE table and determine the limiting reactant and the excess reactant. 4. Determine the [ ] of any excess reactants and the [ ] of any conjugate acid or conjugate base products. 5. Calculate either the p. Ka or the p. Kb 6. Use the appropriate HH Equation

Buffers ● A buffer is a solution with a very stable p. H ● You can add acid or base to the buffer without greatly affecting the p. H of the solution ● The p. H of the buffer will remain unchanged if the solution is diluted with water or water is lost to evaporation. ● A buffer is made by placing a large amount of a weak acid into a solution along with its conjugate in the form of salt. ● The two species will remain in solution together without neutralizing each other. ● This is called. THE COMMON ION EFFECT.

Titration • In this technique, an acid (or base) solution of known concentration is slowly added to a base (or acid) solution of unknown concentration. • A p. H meter or indicators are used to determine when the solution has reached the equivalence point: The point at which stoichiometrically equivalent quantities of acid and base have been brought together. © 2015 Pearson Education, Inc.

Titration • Two reasons to run a titration: 1. To determine the concentration of the unknown reactant 2. To determine the equilibrium constant for the reaction. (p. H=p. Ka at the ½ equivalence point, buffer region) © 2015 Pearson Education, Inc.

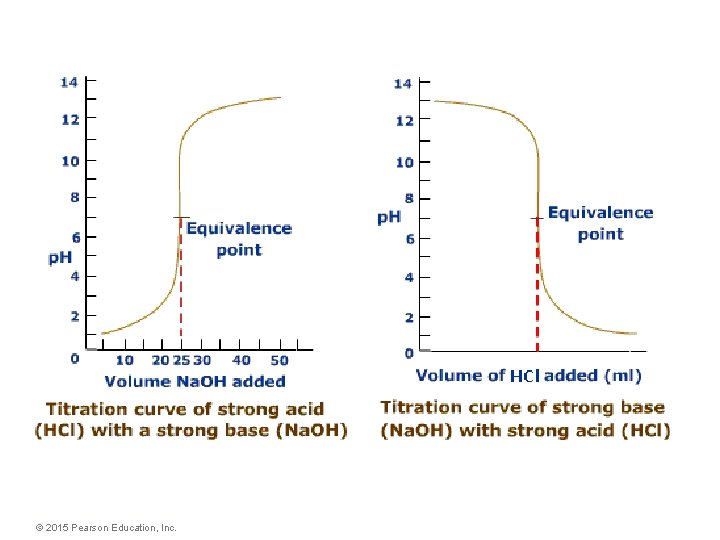
Titration of a Strong Acid with a Strong Base • From the start of the titration to near the equivalence point, the p. H goes up slowly. • Just before (and after) the equivalence point, the p. H rises rapidly. • At the equivalence point, p. H = 7. • As more base is added, the p. H again levels off. © 2015 Pearson Education, Inc.
© 2015 Pearson Education, Inc.

Ways That a Weak Acid Titration Differs from a Strong Acid Titration 1) A solution of weak acid has a higher initial p. H than a strong acid. 2) The p. H change near the equivalence point is smaller for a weak acid. (This is at least partly due to the buffer region. ) 3) The p. H at the equivalence point is greater than 7 for a weak acid.

Ways That a Weak Acid Titration Differs from a Strong Acid Titration Note: It is the amount of acid not its strength that determines the equivalence point. It is the strength of the acid that determines the p. H value at the equivalence point.
- Slides: 50