Nuclear Reactions FISSION FUSION Introduction Nuclear reactions deal












- Slides: 14

Nuclear Reactions: FISSION & FUSION

Introduction ã Nuclear reactions deal with interactions between the nuclei of atoms ã Both fission and fusion processes deal with matter and energy

Matter and Energy ã Previous studies have taught us that “matter and energy cannot be created nor destroyed” ã We now need to understand that Matter and Energy are two forms of the same thing

E = mc 2 ã Remember that matter can be changed into Energy ã This tells us that a small amount of mass can be converted into a very large amount of energy because the speed of light (c) is an extremely large number Energy Mass Light Speed

Fission ã Fission may be defined as the process of splitting an atomic nucleus into fission fragments ã The fission fragments are generally in the form of smaller atomic nuclei and neutrons ã Large amounts of energy are produced by the fission process

Fission ã Fissile nuclei are generally atoms with more neutrons than protons ã The nuclei of such heavy atoms are struck by neutrons initiating the fission process ã Fission occurs when the strong nuclear force is disrupted by an incoming projectile (in this case a neutron) ã When the strong nuclear force is disrupted electrostatic repulsion splits the nuclei

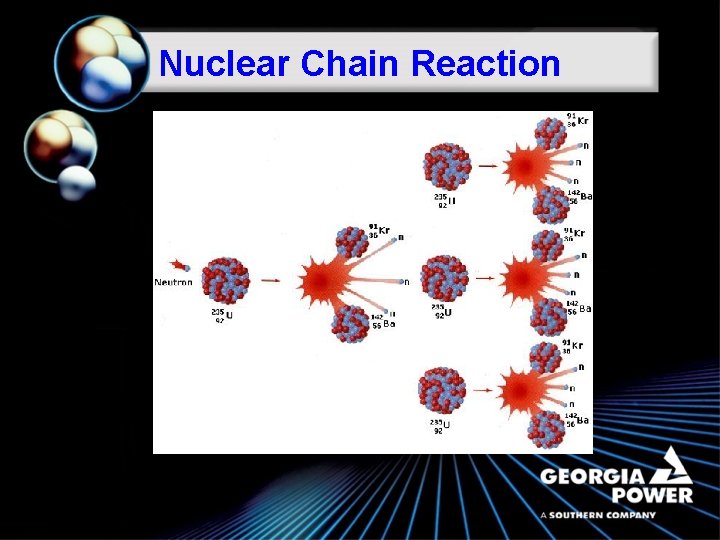
Fission ã A classic example of a fission reaction is that of U-235: U-235 + 1 Neutron 3 Neutrons + Kr-91 + Ba-142 + Energy ã In this example, a stray neutron strikes an atom of U-235. It absorbs the neutron and becomes an unstable atom of U-236. It then undergoes fission. Notice that more neutrons are released in the reaction. These neutrons can strike other U-235 atoms to initiate their fission.

Nuclear Chain Reaction

Fission ã Fission produces large amounts of heat energy and it is this heat that is captured by nuclear power plants to produce electricity.

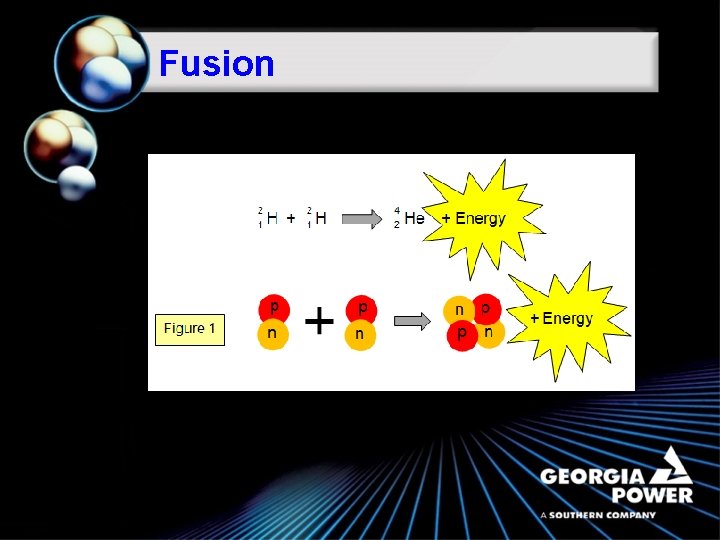
Fusion ã Fusion is a nuclear reaction whereby two light atomic nuclei fuse or combine to form a single larger nuclei which is lighter than the sum of the two that fuse. ã The lost mass is converted to energy. (E ( = mc 2 ) ã For fusion to occur, a large amount of energy is needed to overcome the electrical charges of the nuclei and fuse them together

Fusion

Fusion ã Fusion reactions do not occur naturally on our planet but are the principal type of reaction found in stars ã The large masses, densities, and high temperatures of stars provide the initial energies needed to fuel fusion reactions ã The sun fuses hydrogen atoms to produce helium, subatomic particles, and vast amounts of energy

Energy Comparison ã Because of the large binding energies involved in a nucleus, both fission and fusion involve energy changes of more than a million times larger than those energy changes associated with chemical reactions.

Review ã Mass and Energy are two forms of the same thing; neither can be created nor destroyed but mass can be converted into energy (E = mc 2) ã Fission is a nuclear reaction in which a heavy atomic nucleus is split into lighter atomic nuclei ã Fusion is a nuclear reaction in which 2 light atomic nuclei are combined into a single, heavier atomic nucleus