Nuclear Chemistry Nuclear Reactions Chemical reactions w What















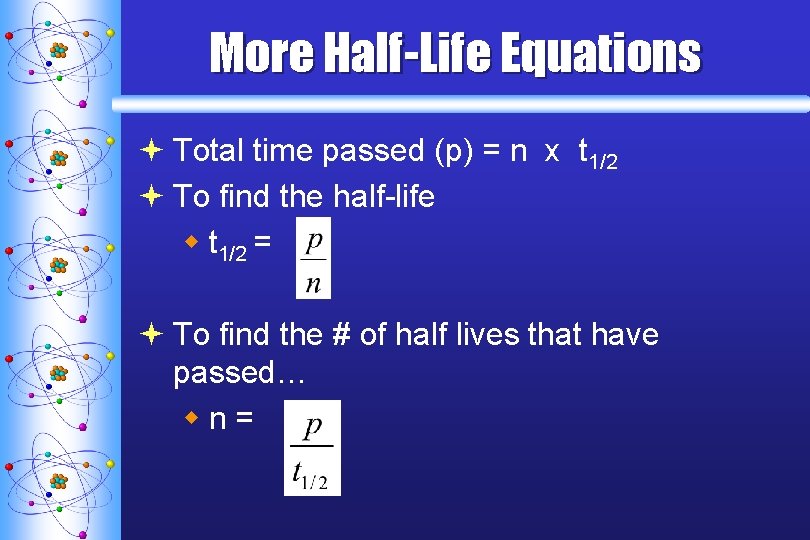





- Slides: 24

Nuclear Chemistry

Nuclear Reactions ª Chemical reactions w What can NOT change in a chemical reaction? ª Nuclear Reactions: changes that occur in the nucleus of an atom w The nucleus is unstable!! ª Most atoms have unstable nuclei.

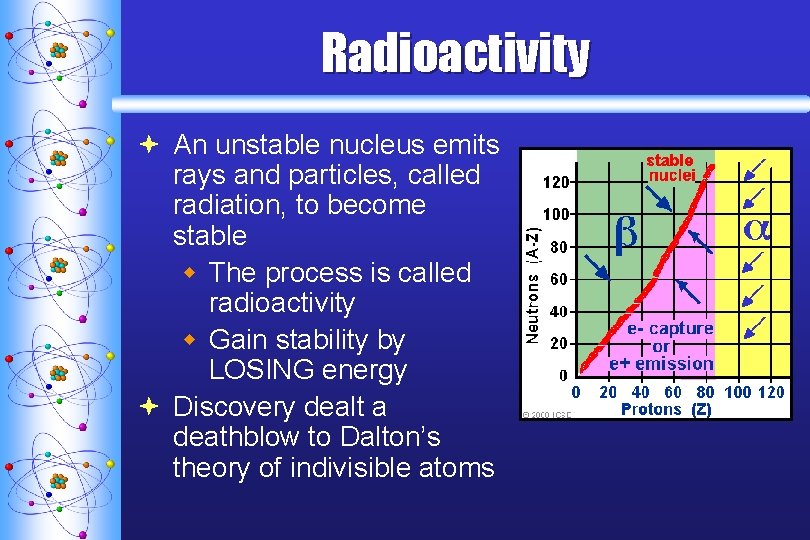
Why are some stable, while others are not? ª Primary Reason: w ratio of the neutrons to the protons (n/p) ª An atom is most stable when ratio is 1: 1 ª The maximum ratio of stability is around 1. 5 : 1

Radioactivity ª An unstable nucleus emits rays and particles, called radiation, to become stable w The process is called radioactivity w Gain stability by LOSING energy ª Discovery dealt a deathblow to Dalton’s theory of indivisible atoms

Types of Radiation ª Alpha particle ( ) w helium nucleus ª Beta particle ( -) w electron ª Gamma ( ) w high-energy photon 2+ paper 10 lead


3 Radioactive Particles ª 1. Alpha Particle (+) w It travels about 1/10 the speed of light (slowest) • It is the largest, most massive particle w It is the most dangerous if ingested w It has the least penetrating ability paper can stop this particle

3 Radioactive Particles ª 2. Beta Particle (-) w Fast accelerated electron w Ejected when a neutron is converted to a proton in the nucleus w Travels 1/4 the speed of light. w It is lighter and faster than the alpha particle. w Average penetrating ability - can be stopped by heavy clothing

3 Radioactive Particles ª 3. Gamma Ray w Not a particle; it is high energy electromagnetic radiation w Has no mass and no charge. w Always accompanies either beta or alpha radiation. w Travels at the speed of light. w Highest penetrating ability - can be stopped by heavy shielding such as lead.

Radioactive Particles Summary

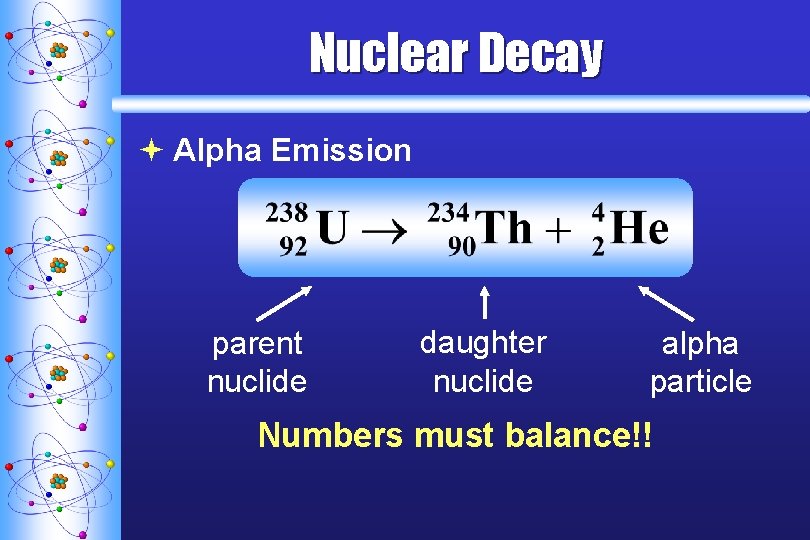
Nuclear Decay ª Alpha Emission parent nuclide daughter nuclide alpha particle Numbers must balance!!

Nuclear Decay ª Beta Emission ª A neutron is converted in the nucleus of an atom ª A new atom is formed whose atomic number is increased by 1

Nuclear Decay ª Gamma Emission w Always accompanies alpha and beta emissions. ª Transmutation w One element becomes another.


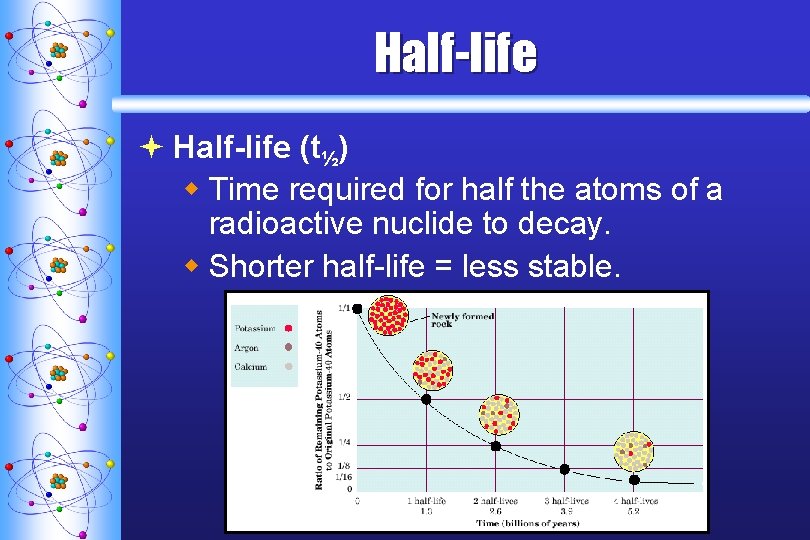
Half-life ª Half-life (t½) w Time required for half the atoms of a radioactive nuclide to decay. w Shorter half-life = less stable.

So, what does this mean? ª Let’s say you have 100 g of radioactive C 14. The half-life of C-14 is 5730 years. w How many grams are left after one halflife? 50 g w How many grams are left after two halflives? 25 g

Common Radioisotopes Isotope Half-Life Radiation Emitted Carbon-14 5, 730 years b Radon-222 3. 8 days b, g Uranium-235 7. 0 x 108 years a, g Uranium-238 4. 46 x 109 years a

Half-life ª To find the remaining amount… r : final mass i : initial mass n : # of half-lives
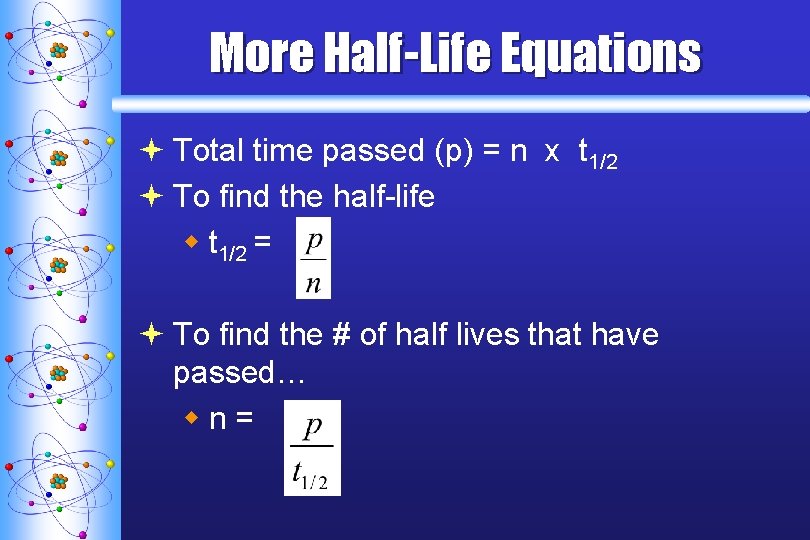
More Half-Life Equations ª Total time passed (p) = n x t 1/2 ª To find the half-life w t 1/2 = ª To find the # of half lives that have passed… wn=

Half-life ª Fluorine-21 has a half-life of 5. 0 seconds. If you start with 25 g of fluorine-21, how many grams would remain after 60. 0 s? GIVEN: WORK: t½ = 5. 0 s r = i (0. 5 )n i = 25 g r = (25 g)(0. 5)12 r=? r = 0. 0061 g p = 60. 0 s n = 60. 0 s ÷ 5. 0 s =12

You try it! ª If the half life element A is 3 hours and you have 90 grams of it, how many grams would be left after 9 hours?

Fission ª splitting a nucleus into two or more smaller nuclei ª 1 g of 235 U = 3 tons of coal

Fission ª chain reaction - self-propagating reaction

Fusion ª combining of two nuclei to form one nucleus of larger mass ª thermonuclear reaction – requires temp of 40, 000 K to sustain ª 1 g of fusion fuel = 20 tons of coal ª occurs naturally in stars