TITRATION CURVE WEAK ACID WITH STRONG BASE MGKP
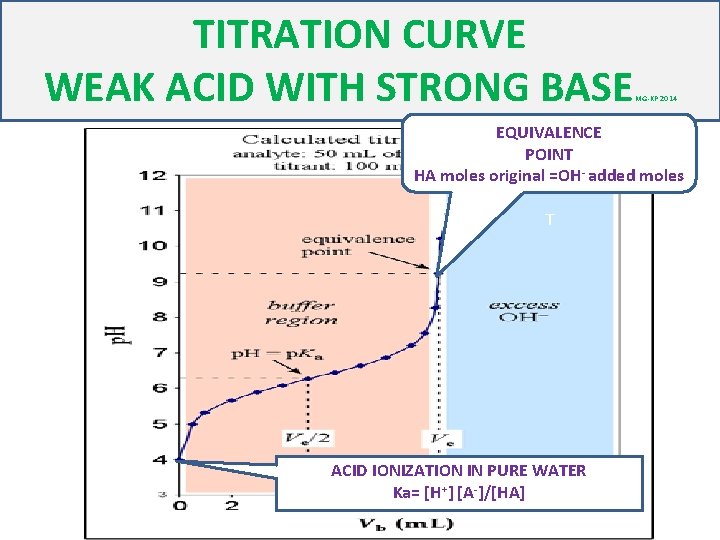
TITRATION CURVE WEAK ACID WITH STRONG BASE MG-KP 2014 EQUIVALENCE POINT HA moles original =OH- added moles T ACID IONIZATION IN PURE WATER Ka= [H+] [A-]/[HA]
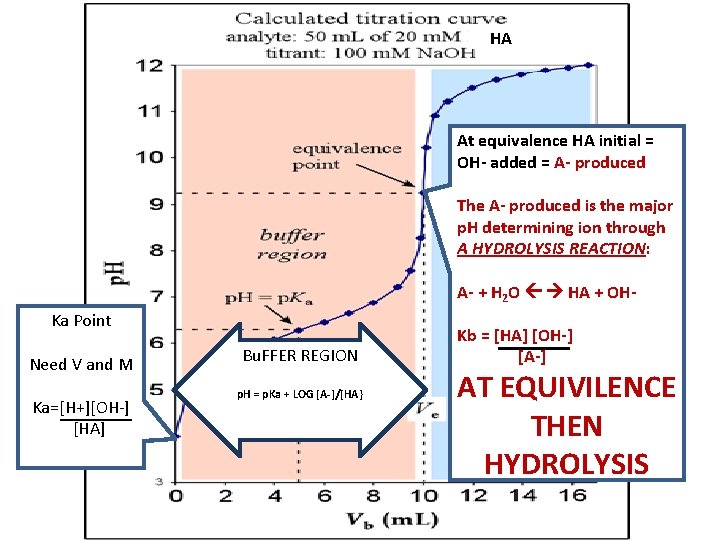
HA At equivalence HA initial = OH- added = A- produced The A- produced is the major p. H determining ion through A HYDROLYSIS REACTION: A- + H 2 O HA + OHKa Point Need V and M Ka=[H+][OH-] [HA] Bu. FFER REGION p. H = p. Ka + LOG [A-]/[HA} Kb = [HA] [OH-] [A-] AT EQUIVILENCE THEN HYDROLYSIS

RECOGNIZING AREAS OF THE TITRATION CURVE 1) THE a) b) c) 2) THE BUFFER ZONE a) SOME MOLES OF WEAK ACID (REMMAINNING) b) BEFORE EQUIVALENCE POINT c) THE BASE IS THE LIMMITING REAGENT IN NEUTRALIZATION ICE CHART. d) AFTER YOU FIND HA AND A- IN THE ICE CHART, USE THEM IN HENDERSON-HASSLEBACH. 3) EQUIVALENCE a) ALL OF THE ACID HAS BEEN NEUTRALIZED EXACTLY (STOICHIOMECTRIC ). b) MOLES OF HA NEUTRALIZED =MOLES OF BASE ADDED = MOLES OF CONJUGATE PRODUCED. Ka POINT: NO BASE HAS BEEN ADDED, BASE ADDED IS (0. 0 m. L). THE p. H CALCULATED WITH THE Ka MASS ACTION. ASSUMES HA IN PURE WATER. HA + OH- H 2 O + A- c) THE BASE AND ACID ARE 0. 0 IN THE END BAR OF THE ICE CHART. d) Ma. Va = Mb. Vb 4) HYDROLYSIS OF THE CONJUGAT E BASE. a) AFTER NEUTRALIZATION THEN HYDROLYSIS b) USE THE Kb: Kb * Ka = Kw c) Kb = [OH-] [HA]/[A-] A- + H 20 OH- + HA

EX) Calculate the p. H of a solution of 100. 0 m. L of 1. 0 M HA (p. Ka = 6. 2)that has had 0. 0 m. L of 1 M Na. OH added in a titration. STEP ONE: - you are at the Ka point, the titration has not begun and no base (OH-) has been added as of now. Simply do a Ka ice chart working in molarities. HA A- 1 molar -x 1 M -x 0. 0 M Approximate this x, 5% rule STEP TWO: Ka = 6. 3 * 10 -7 Ka = [A- ][H+]/[HA] Ka = 6. 3 * 10 -7 = x 2 /1. 0 M X = [H+] = 0. 00079 M p. H = - LOG 0. 00079 = 3. 1 = answer H+ 0. 0 M x x
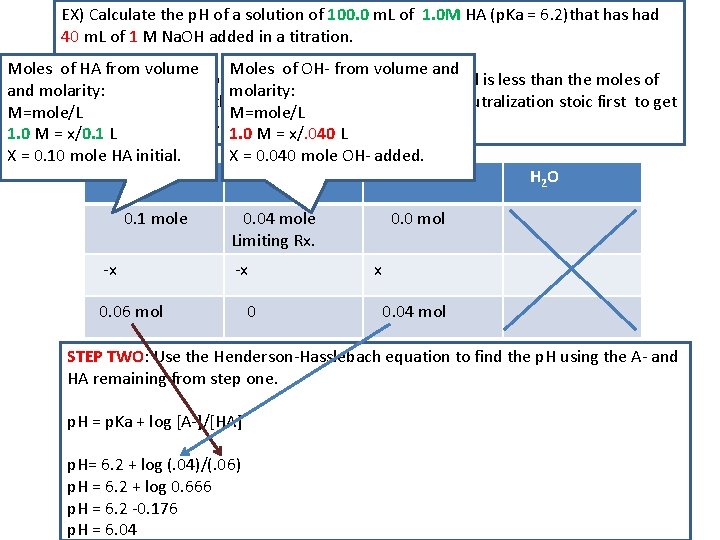
EX) Calculate the p. H of a solution of 100. 0 m. L of 1. 0 M HA (p. Ka = 6. 2)that has had 40 m. L of 1 M Na. OH added in a titration. Moles of HA from volume Moles of OH- from volume and STEP ONE: - you are below equivalence as the OH- added is less than the moles of and molarity: HA initial. You are in the buffer range so evaluate the neutralization stoic first to get M=mole/L A- and HA remaining. 1. 0 M = x/0. 1 L 1. 0 M = x/. 040 L X = 0. 10 mole HA initial. /#L X = 0. 040 mole OH- added. /#L HA OHAH 2 O 0. 1 mole -x 0. 04 mole Limiting Rx. -x 0. 06 mol 0 0. 0 mol x 0. 04 mol STEP TWO: Use the Henderson-Hasslebach equation to find the p. H using the A- and HA remaining from step one. p. H = p. Ka + log [A-]/[HA] p. H= 6. 2 + log (. 04)/(. 06) p. H = 6. 2 + log 0. 666 p. H = 6. 2 -0. 176 p. H = 6. 04

EX) Calculate the p. H of a solution of 100. 0 m. L of 1. 0 M HA (p. Ka = 6. 2)that has had Moles 100 ofm. L HAm. L from of a. OHfrom volume and of volume 1 M Na. OH Moles added in titration. and molarity: M=mole/L STEP ONE: - you are below equivalence as the OH- added is less than the moles of 1. 0 MHA = x/0. 1 M =range x/0. 10 initial. L You are in the 1. 0 buffer so. Levaluate the neutralization stoic first to get X = 0. 10 mole. HA HAremaining. initial. /#L X = 0. 10 mole OH- added. /#L A- and HA OH- 0. 01 mole -x 0 A- 0. 01 mole -x 0 Equivalence…all consumed H 2 O 0. 0 mol x 0. 01 mole Convert moles of A- -to molarity for the next step which is the hydrolysis equilibrium, remember you should work in molarity for any equilibrium calculation. : M=0. 010 mole / 0. 2 L (total volume HA + OH-)=. 050 molar ASEE NEXT SLIDE FOR HYDROLYSIS Eq

The molarity of the conjugate from last slide is 0. 05 molar A- H 2 O 0. 050 M OH- HA 0. 0 -x x x 0. 050 M -x x M x. M Now solve for [OH-] in the hydrolysis (Kb) mass action. Kb = [OH-] [ HA] / [A-] 5. 9 x 10 -11 = (x)(x)/0. 05, x = [OH-] = 1. 7 x 10 -6 , - LOG(1. 7 x 10 -6 ) = p. OH- = 5. 7 14=p. H + p. OH , 14 = p. H + 5. 7, p. H 8. 3 Kb = Kw/Ka Kb=1 x 10 -14/1. 8 x 10 -4 Kb= 5. 9 x 10 -11
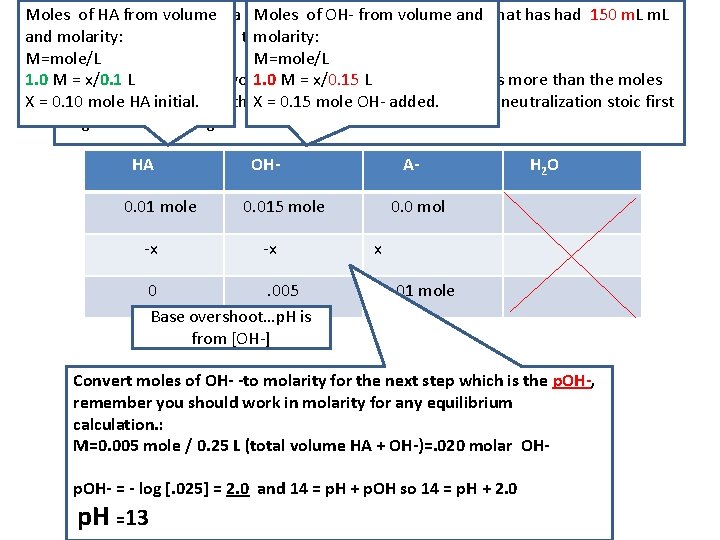
Moles. EX)of. Calculate HA from the volume p. H of a solution Moles of of OH 100. 0 from m. L volume of 1. 0 Mand HA that has had 150 m. L and molarity: of 1 M Na. OH added in a titration. molarity: M=mole/L 1. 0 MSTEP = x/0. 1 ONE: L - you are beyond 1. 0 equivalence M = x/0. 15 as L the OH- added is more than the moles X = 0. 10 of HA mole initial. HA initial. /#L You are in the. Xovershoot = 0. 15 mole range OH-so added. /#L evaluate the neutralization stoic first to get OH- remaining. HA 0. 01 mole -x OH- A- 0. 015 mole -x 0. 005 Base overshoot…p. H is from [OH-] H 2 O 0. 0 mol x 0. 01 mole Convert moles of OH- -to molarity for the next step which is the p. OH-, remember you should work in molarity for any equilibrium calculation. : M=0. 005 mole / 0. 25 L (total volume HA + OH-)=. 020 molar OHp. OH- = - log [. 025] = 2. 0 and 14 = p. H + p. OH so 14 = p. H + 2. 0 p. H =13
- Slides: 8