Matter and Change Matter Phys v Chem Periodic


















- Slides: 28

Matter and Change!

Matter Phys v. Chem Periodic Tbl Sig Figs Problems 10 10 10 20 20 20 30 30 30 40 40 40 50 50 50

Topic 1 – 10 Points QUESTION: • Define matter. (at least 2 of the 3) ANSWER: • mass, volume, exists as a solid, liquid, gas, or plasma

Topic 1 – 20 Points QUESTION: Which state of matter has the lowest amount of energy? • ANSWER: • Solid

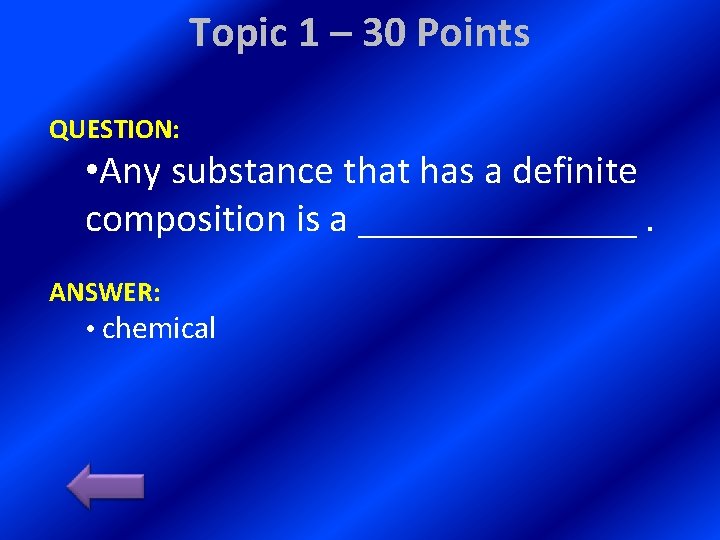
Topic 1 – 30 Points QUESTION: • Any substance that has a definite composition is a _______. ANSWER: • chemical

Topic 1 – 40 Points QUESTION: • Is oxygen gas (O 2) a compound, element, homogeneous mixture, or heterogeneous mixture? ANSWER: • element

Topic 1 – 50 Points QUESTION: • The smallest unit of an element. ANSWER: • an atom

Topic 2 – 10 Points QUESTION: • Changing matter without changing its chemical identity. ANSWER: • Physical

Topic 2 – 20 Points QUESTION: • Burning wood is an example of _____. ANSWER: • Chemical change

Topic 2 – 30 Points QUESTION: • List the five things that indicate a chemical change has occurred ANSWER: • smell, color, gas production, heat production, precipitate formation

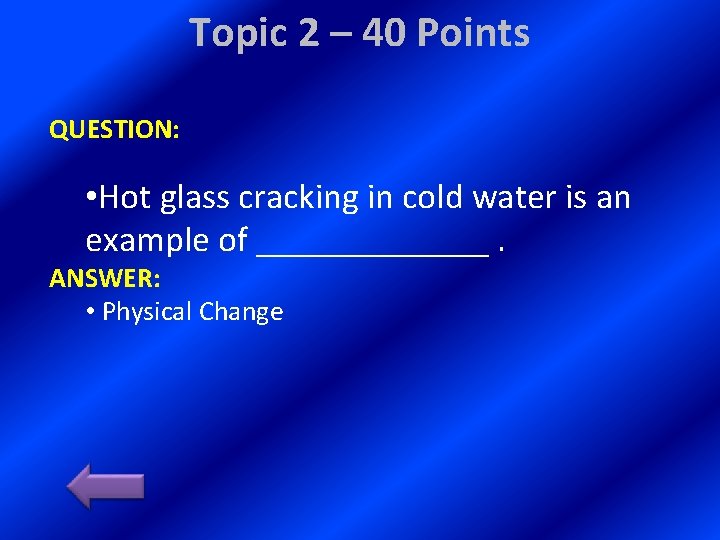
Topic 2 – 40 Points QUESTION: • Hot glass cracking in cold water is an example of _______. ANSWER: • Physical Change

Topic 2 – 50 Points QUESTION: • The physical change from a solid to a gas is called _______. ANSWER: • sublimation

Topic 3 – 10 Points QUESTION: • The vertical columns are also known as _______. ANSWER: • groups or families

Topic 3 – 20 Points QUESTION: • Column 1 is known as the _________. ANSWER: • Alkali metals

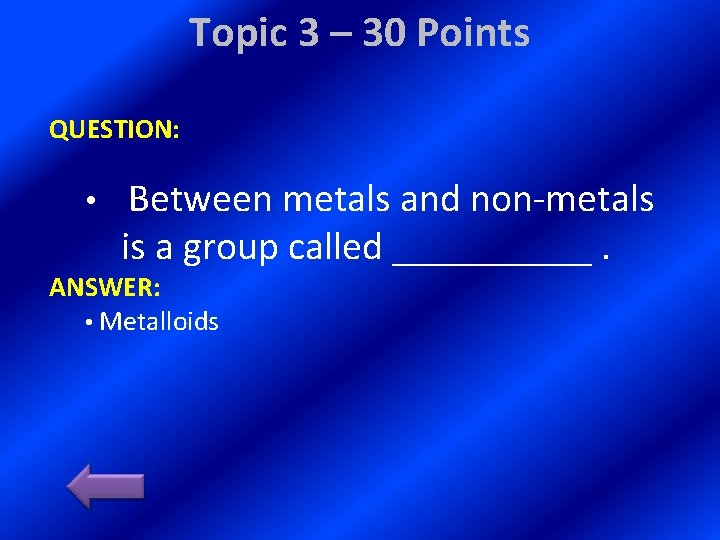
Topic 3 – 30 Points QUESTION: • Between metals and non-metals is a group called _____. ANSWER: • Metalloids

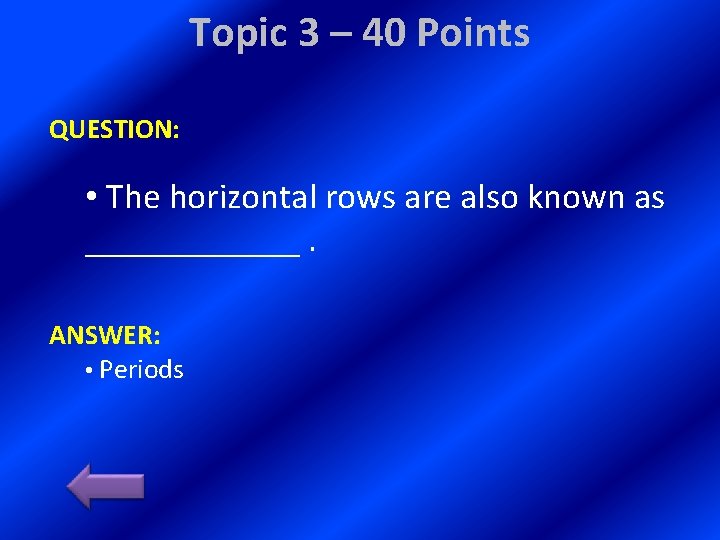
Topic 3 – 40 Points QUESTION: • The horizontal rows are also known as ______. ANSWER: • Periods

Topic 3 – 50 Points QUESTION: • List four general properties of metals. ANSWER: • Luster, heat and electrical conductors, ductile, malleable

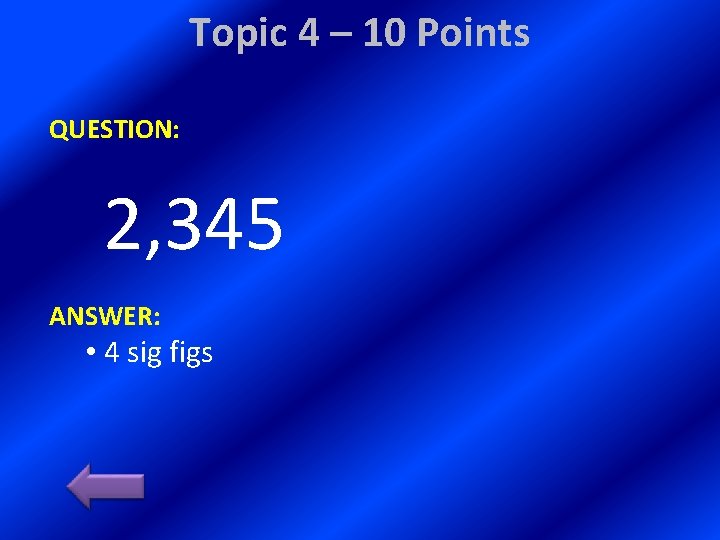
Topic 4 – 10 Points QUESTION: 2, 345 ANSWER: • 4 sig figs

Topic 4 – 20 Points QUESTION: 67, 000 ANSWER: • 2 sig figs

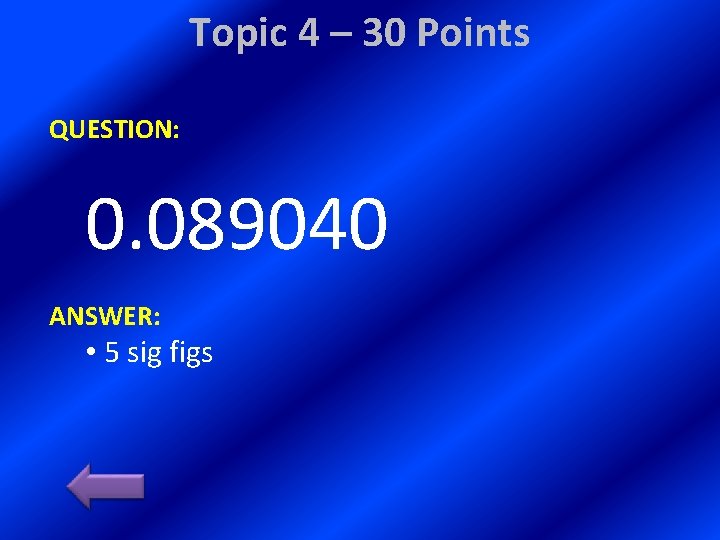
Topic 4 – 30 Points QUESTION: 0. 089040 ANSWER: • 5 sig figs

Topic 4 – 40 Points QUESTION: Problem: 1818. 2 lb x 3. 23 ft 5872. 786 lbft Daily Double Calc says: What is the answer? ANSWER: • 5870 lbft

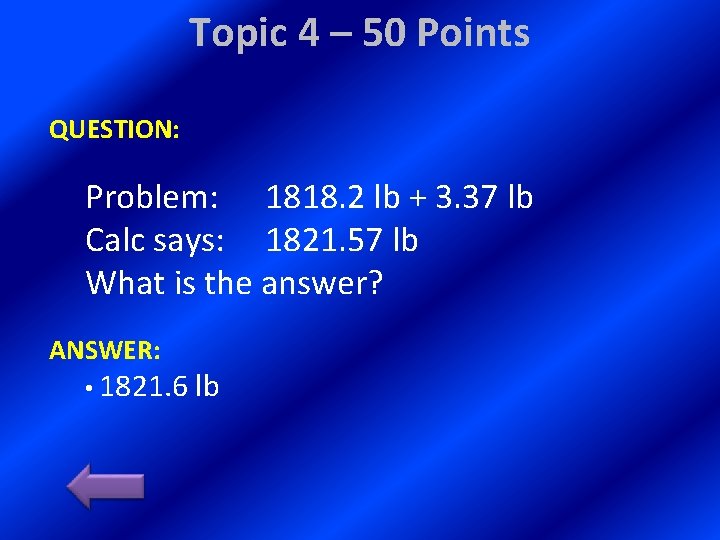
Topic 4 – 50 Points QUESTION: Problem: 1818. 2 lb + 3. 37 lb Calc says: 1821. 57 lb What is the answer? ANSWER: • 1821. 6 lb

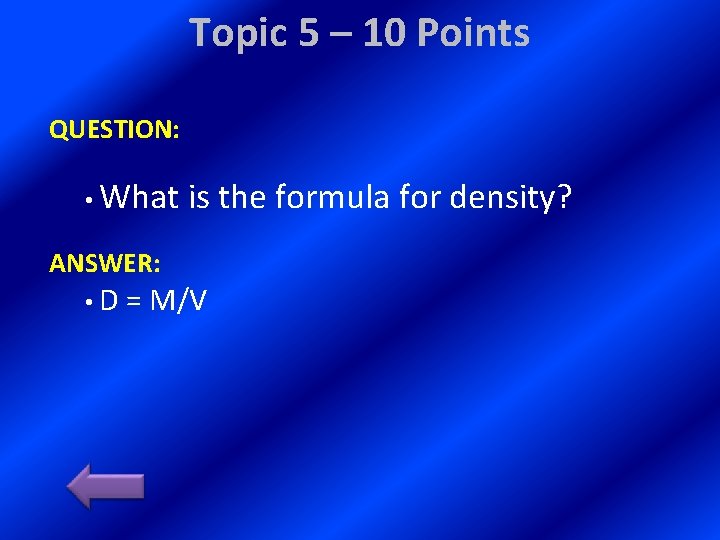
Topic 5 – 10 Points QUESTION: • What ANSWER: is the formula for density? • D = M/V

Topic 5 – 20 Points QUESTION: • If there is a sample of gold with a volume of 10 cm 3 and a mass of 100 g, what is the density? ANSWER: • 10 g/cm 3

Topic 5 – 30 Points QUESTION: • I have 35 m. L of water and the density of water is 1. 0 g/m. L, what is the mass of the water? ANSWER: • 35 g

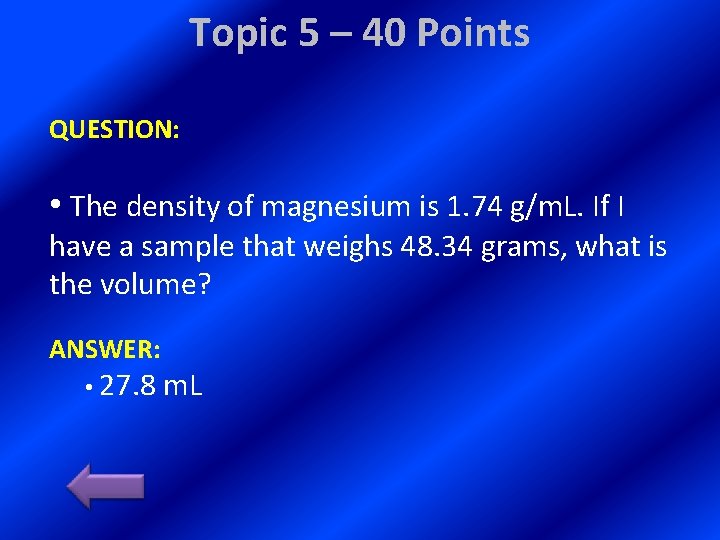
Topic 5 – 40 Points QUESTION: • The density of magnesium is 1. 74 g/m. L. If I have a sample that weighs 48. 34 grams, what is the volume? ANSWER: • 27. 8 m. L

Topic 5 – 50 Points QUESTION: • Convert 2, 780 μm into km and express the answer in scientific notation. ANSWER: • 2. 78 x 10 -6

Final Jeopardy Question: You are given a piece of gold foil with a length of 7. 32 cm and a width of 14. 9 mm. You weigh the foil at. 30 g. The density of gold is 19. 3 g/m. L. What is the thickness of the foil? Answer: . 30 / 19. 3 = 0. 016 cm 3 0. 016 / (7. 32 x 1. 49) = 0. 0015 cm