Periodic Table of Elements Periodic Table The periodic

Periodic Table of Elements

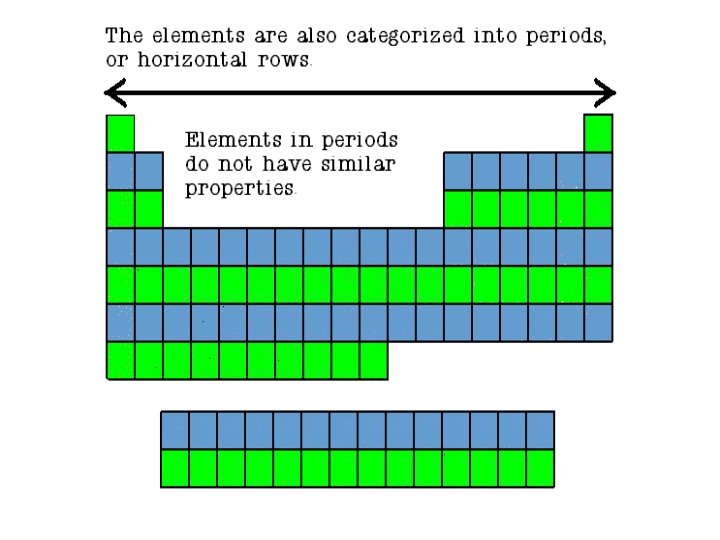
Periodic Table � The periodic table organizes the elements in a particular way. A great deal of information about an element can be gathered from its position in the period table. � For example, you can predict with reasonably good accuracy the physical and chemical properties of the element. You can also predict what other elements a particular element will react with chemically. � Understanding the organization and plan of the periodic table will help you obtain basic information about each of the 118 known elements.

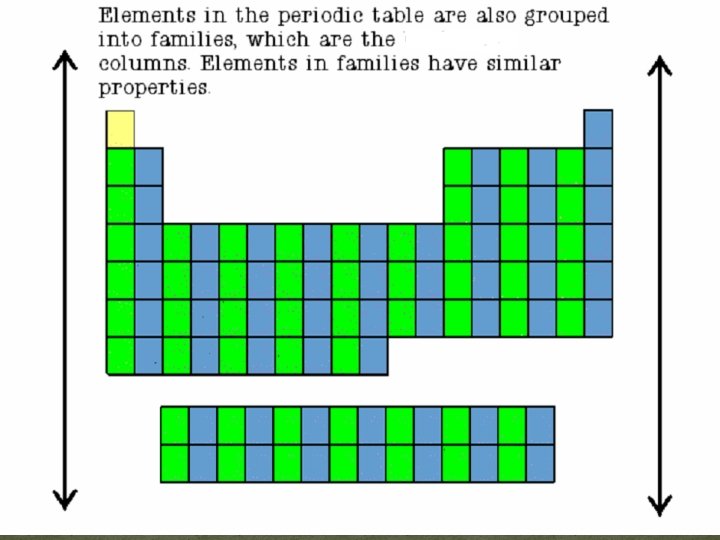
Periodic Table Families
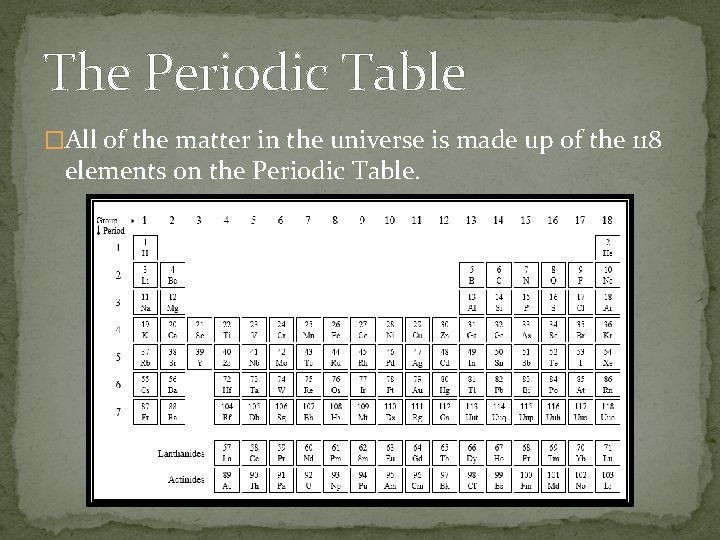
The Periodic Table �All of the matter in the universe is made up of the 118 elements on the Periodic Table.
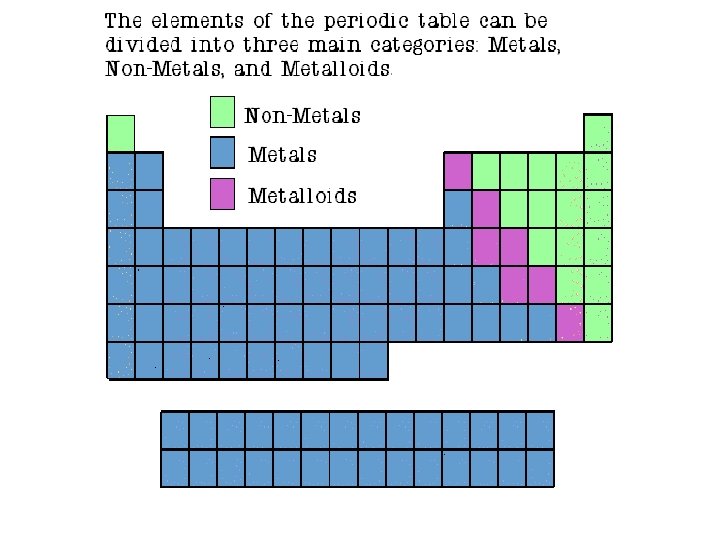

Properties of Metals � Physical Properties: � Shiny � Good conductors of heat and electricity � High melting point � High density � Malleable (can be hammered) � Ductile (can be drawn into wires) � Usually solid at room temperature (an exception is mercury) � Chemical Properties: � Corrode easily � Have lower electronegativities

Properties of Non-Metals �Physical Properties: � Dull � Poor conductors of heat and electricity � Not ductile or malleable � Brittle � May be solids, liquids or gases at room temperature � Many are gases Sulfur �Chemical Properties: � Readily gain or share valence electrons � Have higher electronegativities

Properties of Metalloids � Metalloids (metal-like) shares properties of both metals and non-metals �Solid at room temperature �Brittle, hard �Somewhat reactive �Conducts electricity �They are ductile and malleable. Silicon

Unknown Element #1 Melting Point = 357٥ F (considered low) Boiling Point = 2, 426٥ F Floats on water Solid at room temperature Very reactive metal 1 Valence electron Good conductor of heat

Unknown Element #2 Melting Point = 6, 332 *F Boiling Point = 8, 720 *F Solid at room temperature Unreactive with most elements 4 Valence electrons Poor conductor of heat

Unknown Element #3 Melting Point = -458 *F Boiling Point = -452٥ F 2 nd lightest element Gas at room temperature Unreactive nonmetal 8 Valence electrons Good conductor of heat

Unknown Element #4 Melting Point = 145*F Boiling Point = 1, 420*F Floats on water Soft enough to cut with a knife Very reactive metal 1 Valence electron Good conductor of heat

Unknown Element #5 Melting Point = 246٥ F Boiling Point = 832٥ F Solid at room temperature Reactive nonmetal 6 Valence electrons Poor conductor of heat

Unknown Element #6 Melting Point = -415٥ F Boiling Point = -410٥ F Gas at room temperature Unreactive nonmetal 8 Valence electrons Poor conductor of heat

Unknown Element #7 Melting Point = 208*F Boiling Point = 1, 621*F Floats on water Soft enough to be cut with a knife Very reactive metal 1 Valence electron Good conductor of electricity

Unknown Element #8 Melting Point = -308٥ F Boiling Point = -302٥ F Gas at room temperature Unreactive nonmetal 8 Valence electron Poor conductor of heat

Unknown Element #9 Melting Point = -361٥ F Boiling Point = -297٥ F Gas at room temperature Very reactive nonmetal 6 Valence electrons Poor conductor of heat

Elements 1. 2. 3. 4. 5. 6. 7. 8. 9. Lithium Carbon Helium Potassium Sulfur Neon Sodium Argon Oxygen
- Slides: 20