Constructing the periodic table Periodic Table The Periodic

Constructing the periodic table Periodic Table

The Periodic Table Arrangement The modern periodic table is arranged by orbital diagrams. Schrodinger gave us an equation that describes atomic orbitals. The values we get from solving that equation are quantum numbers. We used quantum numbers to build orbital diagrams, we used orbital diagrams to build the periodic table

Using the periodic table to determine orbital diagrams The period relates to the energy level of electrons Therefore H and He only have electrons in the 1 st energy level Li, Be, B, C , N , O , F and Ne have electrons up to the 2 nd energy level
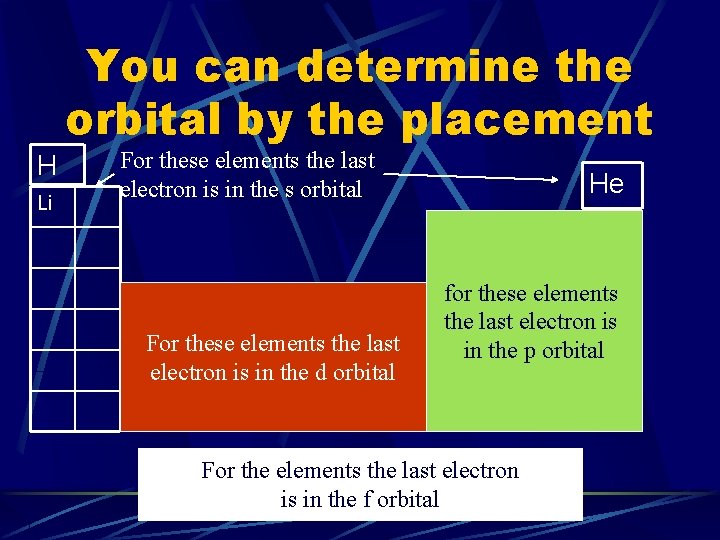
You can determine the orbital by the placement H Li For these elements the last electron is in the s orbital For these elements the last electron is in the d orbital He for these elements the last electron is in the p orbital For the elements the last electron is in the f orbital
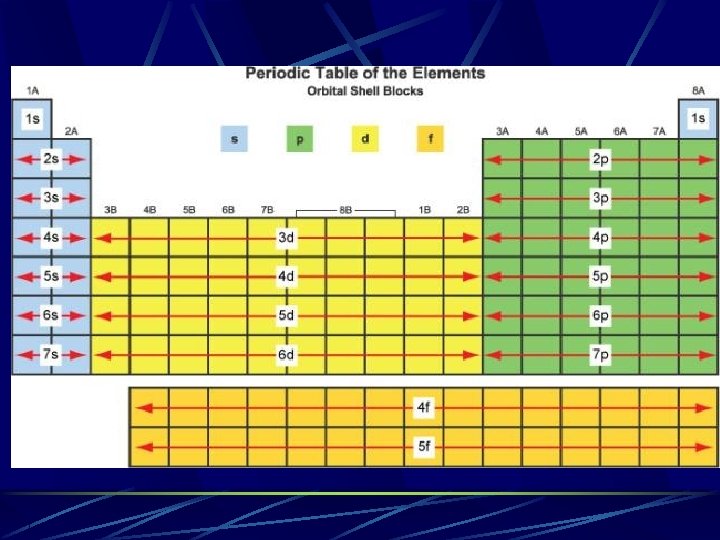

Determining the number of electrons in the last energy level count the number of elements to the left edge of the “section” For example Si It is the second element from the left in the 2 nd row of p (3 p) filling elements The last electron for silicon will be the second electron possible in 3 p

Continuing… Si The last electron will be the second possible in the 2 nd p • 1 s __ • 2 s __ p __ __ __ • 3 s __ p __ __ __ d __ __ __

Question Determine where the last electron will be in Nickel, Bromine, Cesium and Bismuth. Ni 3 d __ __ __ Br 4 p __ __ __ Cs 6 s __ Bi 6 p __ __ __

Noble Gas (condensed) Electron Configuration long electron configurations are not normally written out because it gets rather repetitive Instead it is condensed, by starting from the previous noble gas and adding shells to it. For example K 1 s 22 s 2 p 63 s 2 p 64 s 1 Instead becomes [Ar]4 s 1 This is the same as saying everything that is in Ar (1 s 22 s 2 p 63 s 2 p 6) +4 s 1

To determine the noble gas electron configuration Start at the previous noble gas (lower atomic number) Count out every element in order of atomic number to your given Determine which shell all elements are filling. The number of elements in that shell is your superscript number.

For Iron Previous Noble Gas----Ar Count all elements K Ca Sc Ti V Cr Mn Fe These fill 4 s These fill 3 d So it is [Ar] 4 s 23 d 6 but we should put the energy levels in order [Ar]3 d 64 s 2
![Question Determine noble gas electron configuration for Nickel, Bromine, Cesium and Bismuth Ni [Ar] Question Determine noble gas electron configuration for Nickel, Bromine, Cesium and Bismuth Ni [Ar]](http://slidetodoc.com/presentation_image_h2/0632305893134c55c2a79329ccd05ce3/image-15.jpg)
Question Determine noble gas electron configuration for Nickel, Bromine, Cesium and Bismuth Ni [Ar] 3 d 8 4 s 2 Br [Ar] 3 d 10 4 s 2 p 5 Cs [Xe] 6 s 1 Bi [Xe]4 f 14 5 d 10 6 s 2 p 3

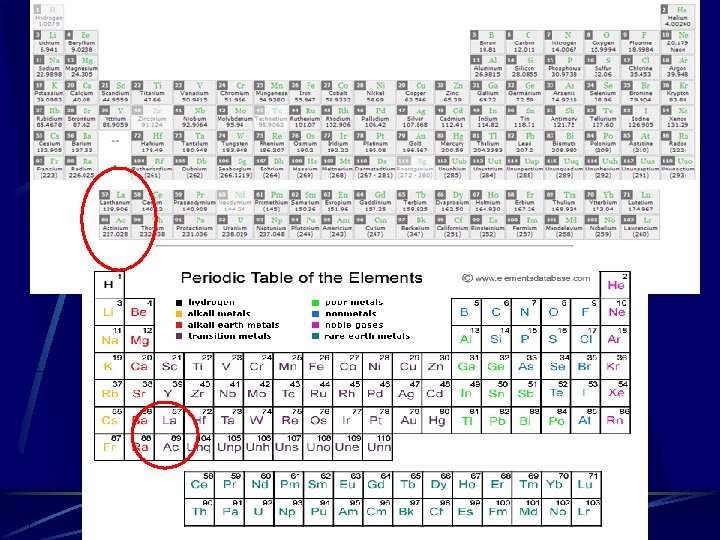
Lone electron in the 5 d or 6 d Odd exception Lanthanum’s last electron fills the 5 d orbital. Ce has its last electron fill in the 4 f, and the one from 5 d (La) jumps to 4 f. The next elements all fill up the 4 f until Gadolinium which fills up the 5 d again. A similar “jumping” of one electron occurs with the actinide series




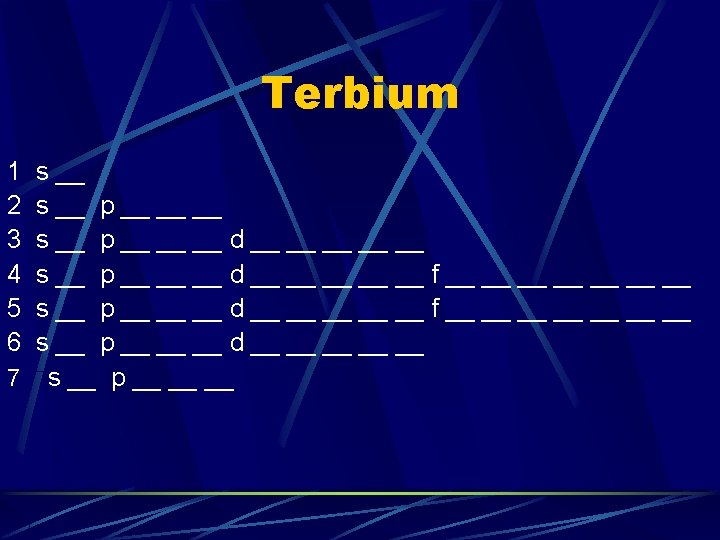
Praseodymium 1 2 3 4 5 6 s __ p __ __ __ d __ __ __ f __ __ __ __ s __ p __ __ __ d __ __ __ 7 s __ p __ __ __ Nd, Pm, Sm Eu fill the next 4 orbitals in 4 f


Aufbau Principle exceptions You don’t need to memorize these

Different Periodic Tables The electron “jumping” is why there are two forms of the periodic table in common usage. One has lanthanum and actinium in with the d filling elements. The other has lanthanum and actinium with the f filling elements

Exceptions There are several exceptions to the Aufbau principal, the “normal order” that we follow for orbital diagrams. The first occur with Chromium and Copper. Cr = [Ar]3 d 54 s 1, Cu= [Ar]3 d 104 s 1 You don’t need to memorize the exceptions (at this level of chemistry), just know that there are exceptions

Homework For the following elements Ignore the exception if you run into it. fill in the last shell only of the orbital diagram and do the noble gas electron configuration Ti, Cl, Sr, Sn and Au
- Slides: 27