PERIODIC TABLE Organization of Periodic Table The Periodic

PERIODIC TABLE

Organization of Periodic Table � The Periodic Table organizes information about all known Elements according to the properties � Elements are arranged by atomic number � Patterns exist in the table that adds additional information about how the Elements will react or will not react

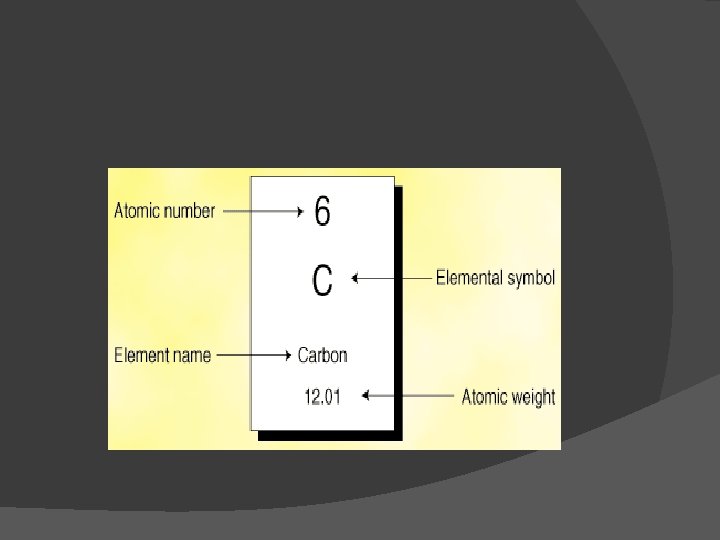
Reading the Periodic Table � Elements are substances that cannot be broken down into other substances and are made up of only one kind of atom. � As of know there are 117 Elements. � Each block on the table denotes one Element � Periodic Tables usually include chemical symbol, element name, atomic number, and atomic mass for each element

Reading continued � Usually the chemical symbol is easy to figure out, such as “Si” for Silicon and “C” for Carbon. � Other like gold have chemical symbols like “Au”. This chemical symbol is based on the Latin word for gold (aurum).

Element Block � The atomic number is equal to the number of protons. � The atomic mass of an element is equal to the sum of the protons and neutrons in one atom of an element.

Organizing the Elements � The Elements increase in atomic number across rows and down columns. � The horizontal rows are called periods. � The vertical columns are called groups or families � The Groups or families have similar physical and chemical properties � If we look at Be, Mg, Ca, they will have similar properties

Metals � Metals can be found to the left of the bold staircase that runs through the table � Metals can conduct electricity easily and are called conductors � Metals are malleable, meaning they can be hammered into thin sheets � Metals are also ductile which means they can be stretched into thin wire

Nonmetals Found to the right of the bold staircase � They can be solids, liquids, or gases � Poor conductors � Nitrogen and Oxygen are two nonmetals that make up a large portion of what we breathe � Graphite is another example and if you think about your pencil lead you see how its properties are different from metal �

Properties of Groups or Families � The Alkali Family is Group 1 - They are the most reactive metals, if placed in water will react violently � The alkaline earth family is group 2 harder than the alkali metals � The halogen family is group 17, most reactive nonmetals � The noble (inert) gas family is group 18, the last group on the right side. They are least reactive of all elements.

Sodium in Water � http: //www. teachertube. com/view. Video. php? video_id=28174&title=Sodium_in_ Water___FHMS

Metalloids � Metalloids carry some properties of metals and some of nonmetals � They are along that jagged line on the periodic table

Transition Elements � Majority of the metals � Groups 3 -12 � Also include elements 57 -71 and 89 -103 � Transition metals tend to be less reactive that the alkali and alkaline earth families

� http: //www. youtube. com/watch? v=3 p. RT Kx. AJK 7 I
- Slides: 14