THE PERIODIC TABLE INTRODUCING THE PERIODIC TABLE Periodic

THE PERIODIC TABLE
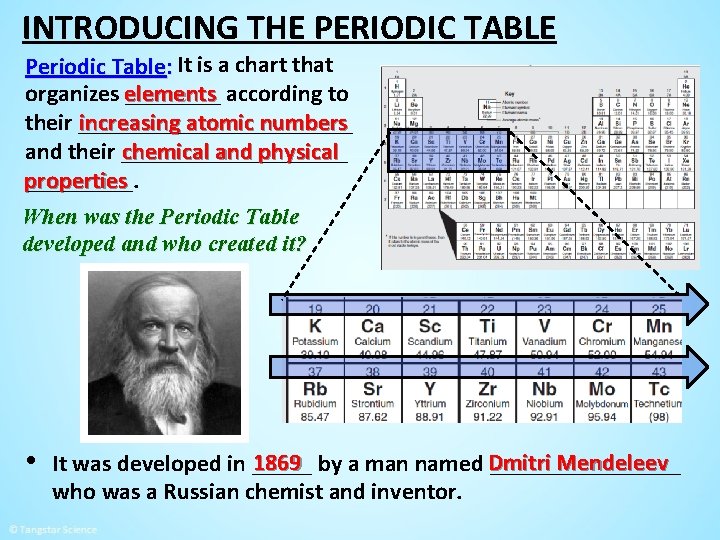
INTRODUCING THE PERIODIC TABLE Periodic Table: It is a chart that organizes elements ____ according to increasing atomic numbers their ____________ and their __________ chemical and physical properties _____. When was the Periodic Table developed and who created it? • 1869 by a man named Dmitri Mendeleev It was developed in ___________ who was a Russian chemist and inventor.
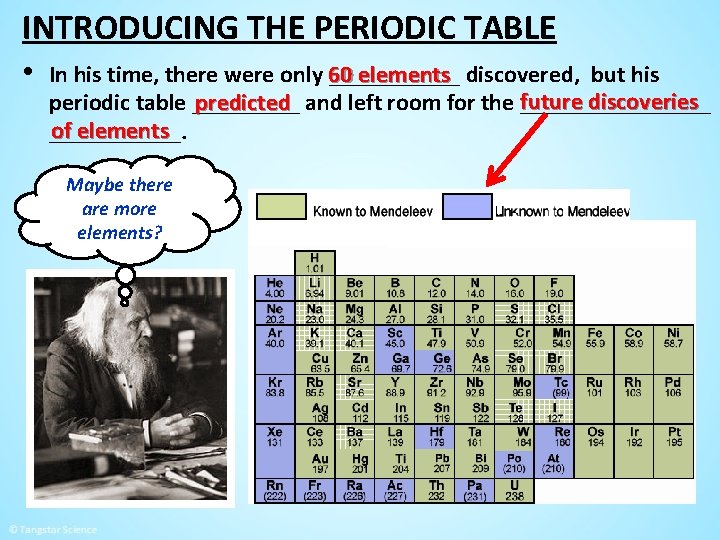
INTRODUCING THE PERIODIC TABLE • In his time, there were only 60 ______ elements discovered, but his future discoveries predicted and left room for the ________ periodic table _____ of elements ______. Maybe there are more elements?

INTRODUCING THE PERIODIC TABLE • Each element on the periodic table is represented within an element box which contains the following basic information. ______ atomic number (A whole number with no decimals. ) 2+ oxidation number (It has a + or – sign. ) element symbol (Only the first letter is capitalized. ) element name atomic mass a) Draw the box for the element that has an atomic mass of 107. 87. 1+ (It is larger than atomic number and always has decimals. ) b) Draw the box for the element that has the element symbol Pt. 4++ 2
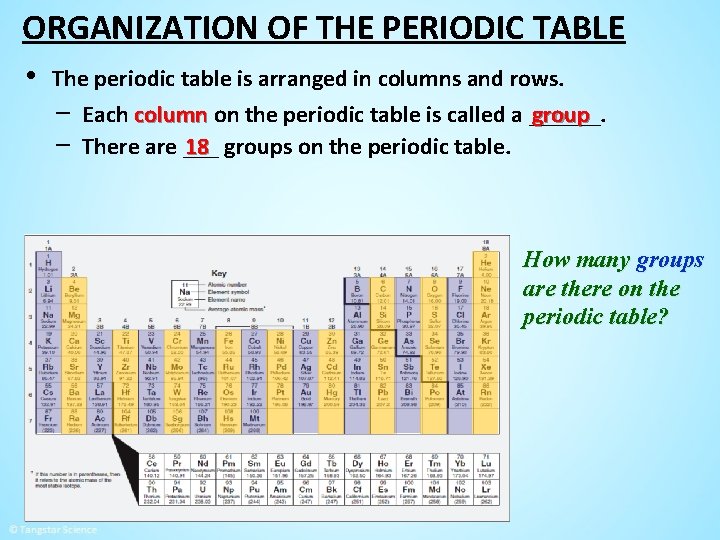
ORGANIZATION OF THE PERIODIC TABLE • The periodic table is arranged in columns and rows. − Each column on the periodic table is called a ______. group − There are ___ 18 groups on the periodic table. How many groups are there on the periodic table?
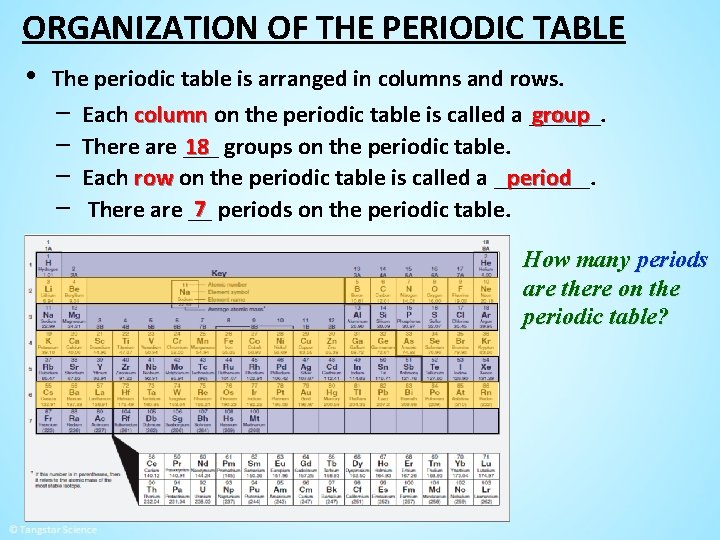
ORGANIZATION OF THE PERIODIC TABLE • The periodic table is arranged in columns and rows. − Each column on the periodic table is called a ______. group − There are ___ 18 groups on the periodic table. period − Each row on the periodic table is called a ____. 7 periods on the periodic table. − There are __ How many periods are there on the periodic table?

ORGANIZATION OF THE PERIODIC TABLE • There is a zigzag that separates the periodic table into two sides. This zigzag is also called the staircase ____. Can you find the zigzag on the periodic table?
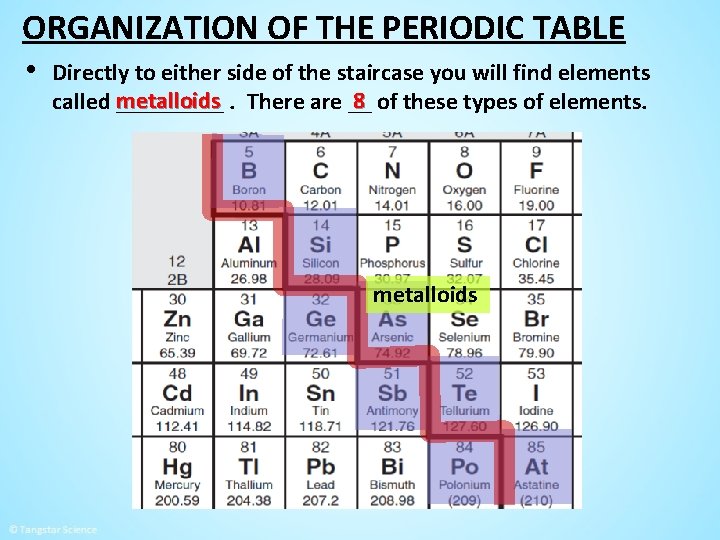
ORGANIZATION OF THE PERIODIC TABLE • Directly to either side of the staircase you will find elements 8 of these types of elements. called metalloids _____. There are __ metalloids

ORGANIZATION OF THE PERIODIC TABLE − − To the left of the staircase you will find all the metals ______. To the right of the staircase you will find all the non-metals _____ , hydrogen , which is a non-metal except for the element _____ found on the left side. non-metals

METALS, NON-METALS & METALLOIDS Metals: • • All metals are solids _____ at room temperature except for mercury which is a liquid _____. Many metals are lustrous _______ and silvery ______ in color, ductile ______ , malleable _____ , conductive _____ and some are magnetic ____. Hg Cu

METALS, NON-METALS & METALLOIDS Non-Metals: gases , except for bromine • Most non-metals are ________ which is a liquid _____ , and five non-metals which ______ , phosphorus _____ , are solids _____. These ones are: carbon sulfur iodine _____ , selenium ____ , and ______. I Br • colors Non-metal elements are either colorless ____ or have various ______. malleable , _____ not ductile , ______ brittle when solid, They are not ______ nonmagnetic poor conductors and _______

METALS, NON-METALS & METALLOIDS Metalloids: all solids at room temperature. • Metalloids are ____ • They have some properties of both ____ metals and non-metals. − They are mostly metallic _______ in appearance but they are fair electrical conductors. brittle They are ______. B Si Ge As Sb Te

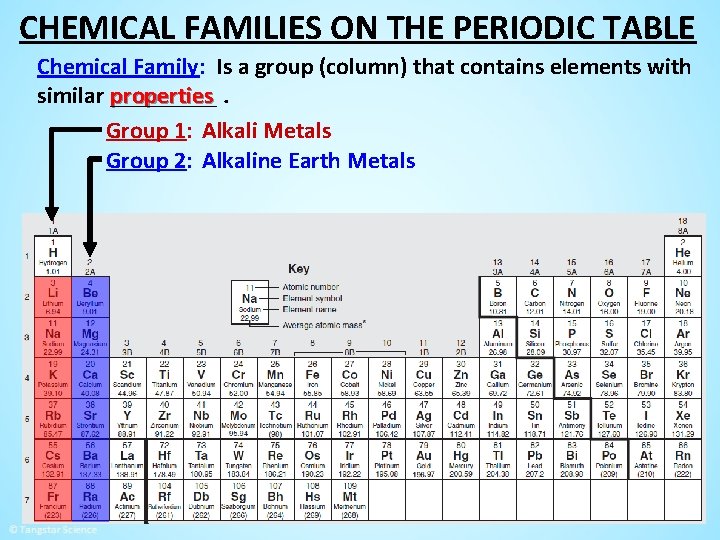
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____
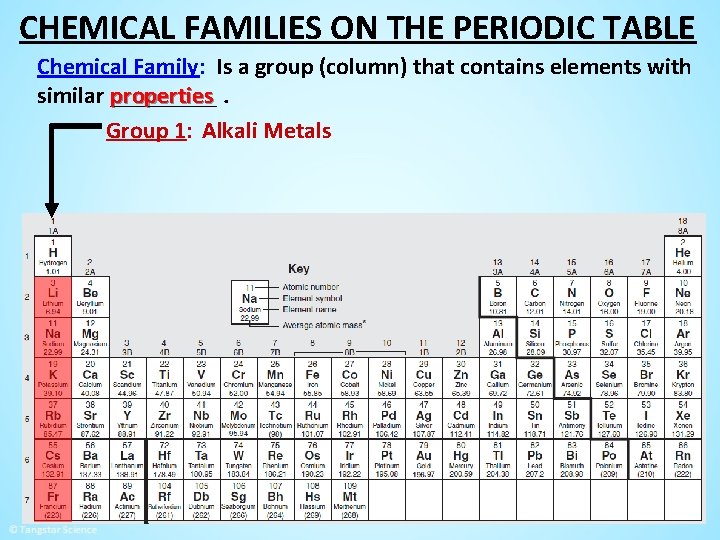
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____ Group 1: Alkali Metals
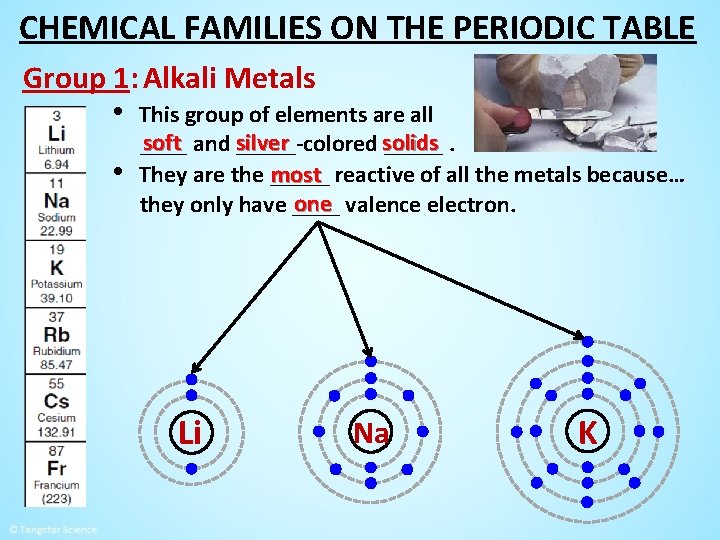
CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 1: Alkali Metals • This group of elements are all • soft and silver _____-colored solids _____. most reactive of all the metals because… They are the _____ one valence electron. they only have ____ Li Na K
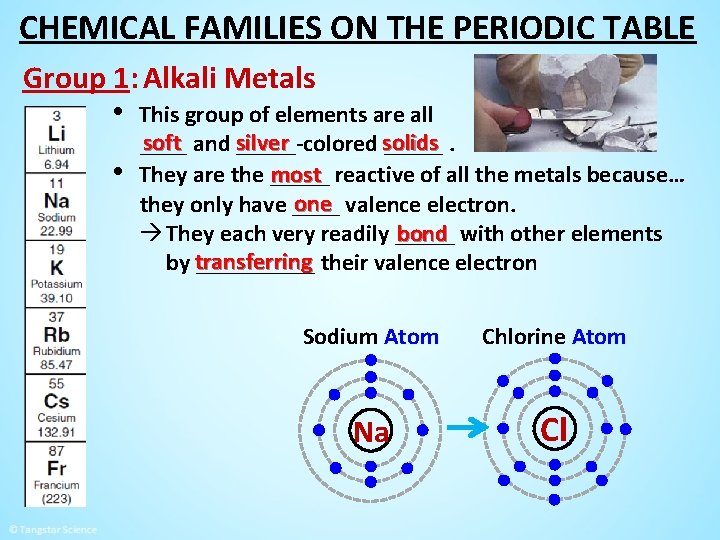
CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 1: Alkali Metals • This group of elements are all • soft and silver _____-colored solids _____. most reactive of all the metals because… They are the _____ one valence electron. they only have ____ bond with other elements à They each very readily _____ by transferring _____ their valence electron Sodium Atom Chlorine Atom Na Cl

CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 1: Alkali Metals • This group of elements are all • soft and silver _____-colored solids _____. most reactive of all the metals because… They are the _____ one valence electron. they only have ____ bond with other elements à They each very readily _____ by transferring _____ their valence electron to each become ion meaning they have a full a stable ___, ___ outer orbital. à The bond Chlorine Ion Atom Sodium Ion between the ions creates new a ____ Cl Na substance called a Na. Cl _____. compound table salt

CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 1: Alkali Metals • They are all reactive with water _____. • vapor in à To prevent contact with water ______ Fr oil the air, they are stored inmineral _____. more reactive as you go Elements get _____ down _____ the group. Francium ____ is the most reactive. . and it’s… water mineral oil
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____ Group 1: Alkali Metals Group 2: Alkaline Earth Metals

CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 2: Alkaline Earth Metals • These elements are also all silver _____-colored solids _____. less so than • They are also reactive with water _____ but ____ group 1 elements They each have two ___ valence electrons. Be Mg Ca Sr Ba
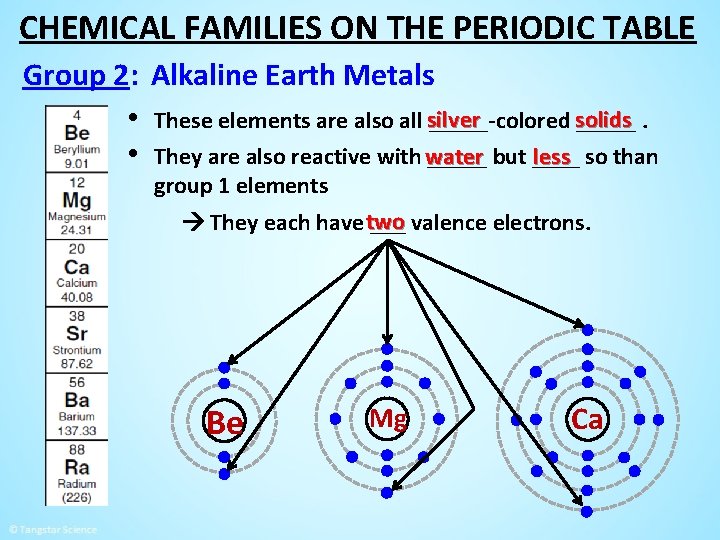
CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 2: Alkaline Earth Metals • These elements are also all silver _____-colored solids _____. less so than • They are also reactive with water _____ but ____ group 1 elements They each have two ___ valence electrons. Be Mg Ca

CHEMICAL FAMILIES ON THE PERIODIC TABLE Group 2: Alkaline Earth Metals • These elements are also all silver _____-colored solids _____. less so than • They are also reactive with water _____ but ____ group 1 elements. • They each have two ___ valence electrons. à Similar to group 1 elements, they readily bond with other elements. However, in this case they two valence electrons to become transfer their ____ stable ions with full outer orbitals. more reactive as you go down Elements get _____ the group. Radium is the most reactive. _______ Ra . . . and it’s also…
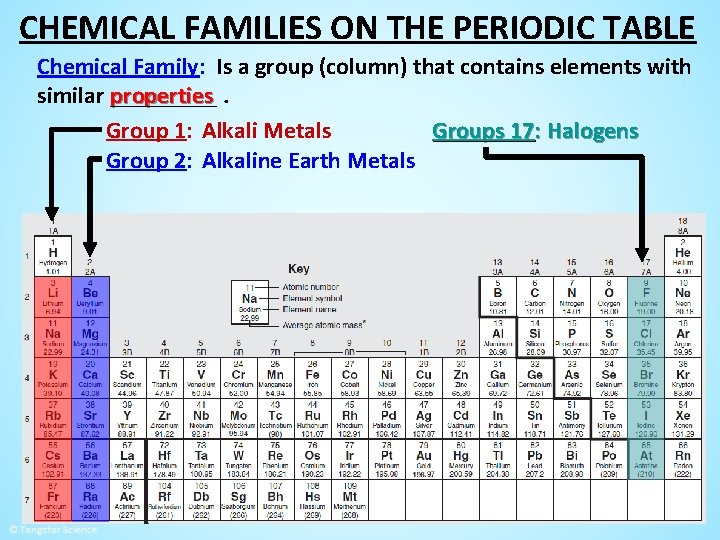
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____ Group 1: Alkali Metals Groups 17: Halogens Group 2: Alkaline Earth Metals
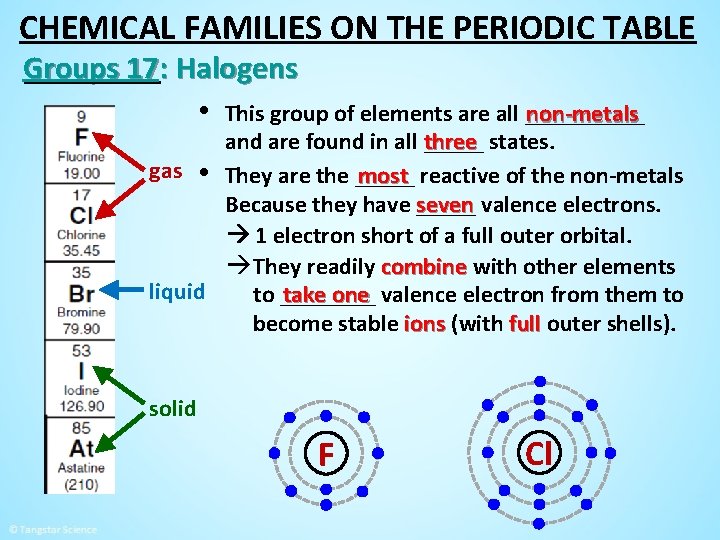
CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 17: Halogens • This group of elements are all _____ non-metals and are found in all three _____ states. gas • They are the _____ most reactive of the non-metals seven valence electrons. Because they have _____ 1 electron short of a full outer orbital. àThey readily combine with other elements liquid take one valence electron from them to to ____ become stable ions (with full outer shells). solid F Cl
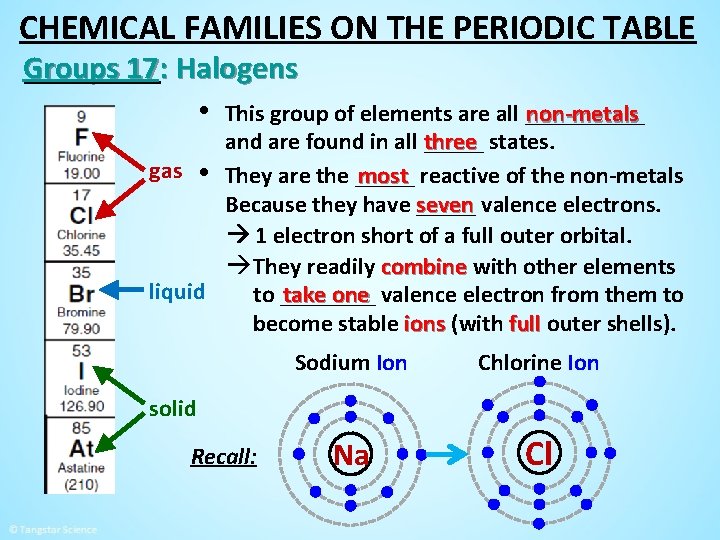
CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 17: Halogens • This group of elements are all _____ non-metals and are found in all three _____ states. gas • They are the _____ most reactive of the non-metals seven valence electrons. Because they have _____ 1 electron short of a full outer orbital. àThey readily combine with other elements liquid take one valence electron from them to to ____ become stable ions (with full outer shells). Sodium Ion Chlorine Ion Na Cl solid Recall:
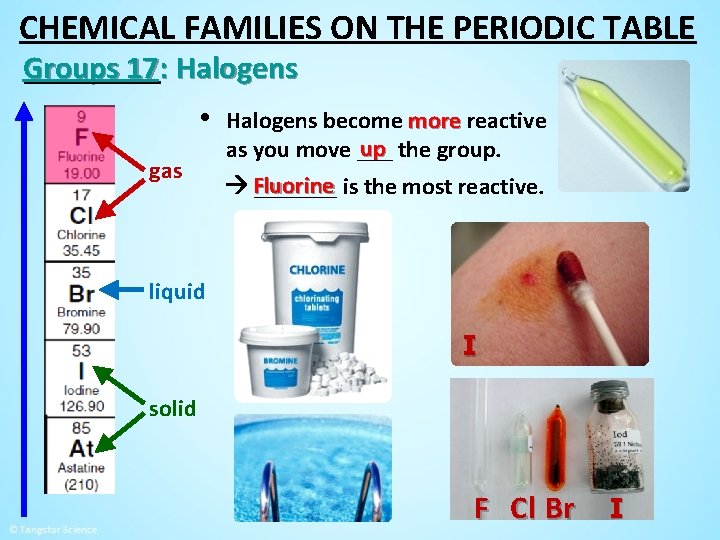
CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 17: Halogens • gas Halogens become more reactive up the group. as you move ___ Fluorine _______ is the most reactive. liquid I solid F Cl Br I
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____ Group 1: Alkali Metals Groups 17: Halogens Group 2: Alkaline Earth Metals Groups 18: Noble Gases

CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 18: Noble Gases • These non-metal elements are non-reactive ______ gases. He Ne Ar Kr Xe
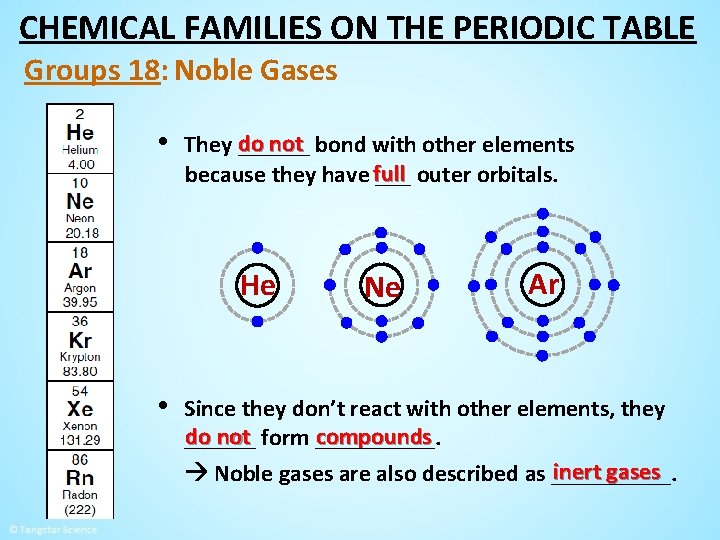
CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 18: Noble Gases • do not bond with other elements They ______ because they have full ___ outer orbitals. He • Ne Ar Since they don’t react with other elements, they do not form _____. compounds ______ inert gases Noble gases are also described as _____.
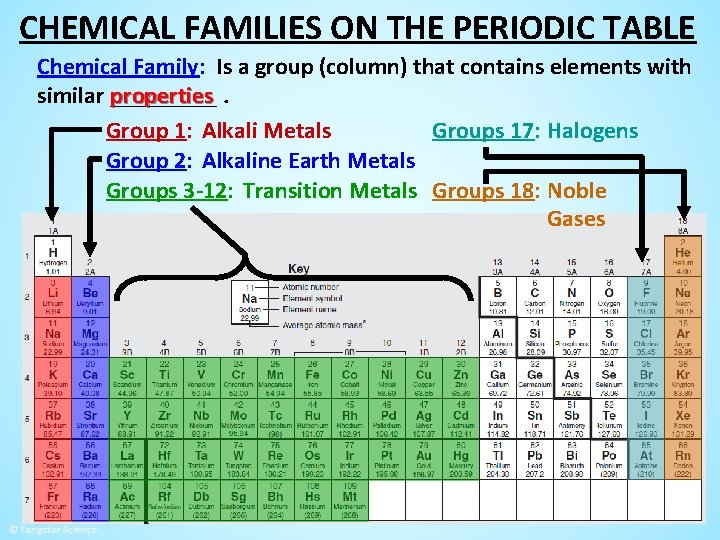
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with properties. similar _____ Group 1: Alkali Metals Groups 17: Halogens Group 2: Alkaline Earth Metals Groups 3 -12: Transition Metals Groups 18: Noble Gases

CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 3 -12: Transition Metals • These groups contain metals that have the usual properties of • metals (Lustrous, malleable, ductile, electrically conductive, etc. ) These metals are all found in the solid state except for mercury _______ which is a liquid _____ at room temperature and pressure.

CHEMICAL FAMILIES ON THE PERIODIC TABLE Groups 3 -12: Transition Metals • The transition metals contain some of the most commonly known metals platinum ring iron skillet titanium aircraft silver goblet copper pipes copper penny tungsten light bulb gold necklace mercury thermometer
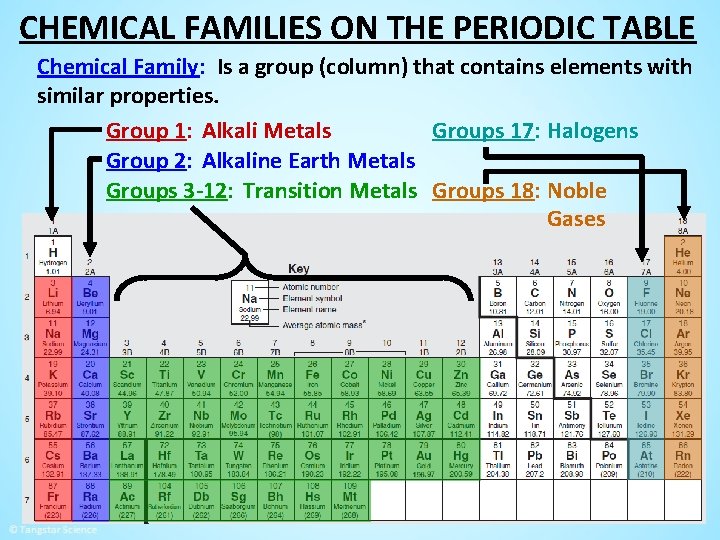
CHEMICAL FAMILIES ON THE PERIODIC TABLE Chemical Family: Is a group (column) that contains elements with similar properties. Group 1: Alkali Metals Groups 17: Halogens Group 2: Alkaline Earth Metals Groups 3 -12: Transition Metals Groups 18: Noble Gases
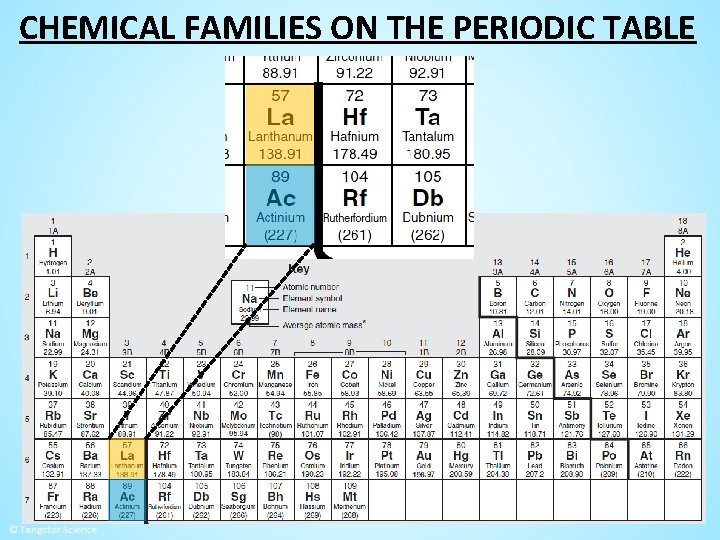
CHEMICAL FAMILIES ON THE PERIODIC TABLE
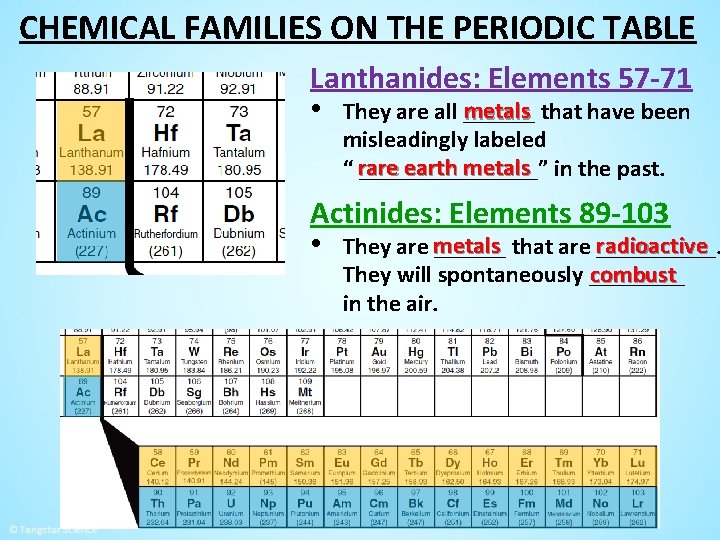
CHEMICAL FAMILIES ON THE PERIODIC TABLE Lanthanides: Elements 57 -71 metals that have been • They are all ______ misleadingly labeled are earth metals in the past. “ r________” Actinides: Elements 89 -103 radioactive • They are metals ______ that are _____. combust They will spontaneously ____ in the air.

The End!

Created by Anh-Thi Tang – Tangstar Science Copyright © April 2013 Anh-Thi Tang (a. k. a. Tangstar Science) All rights reserved by author. This document is for personal classroom use only. This entire document, or any parts within, may not be electronically distributed or posted to any website.
- Slides: 37