Periodic Trends the Periodic Table Periodic Table Periodic

Periodic Trends & the Periodic Table

Periodic Table • Periodic Table – arrangement of elements in order of increasing atomic number with elements having similar properties in vertical columns – Groups – vertical columns – Periods – horizontal rows
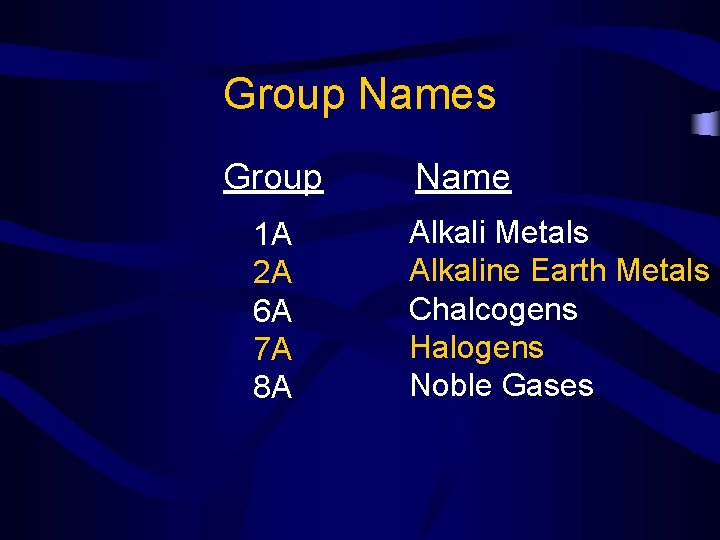
Group Names Group 1 A 2 A 6 A 7 A 8 A Name Alkali Metals Alkaline Earth Metals Chalcogens Halogens Noble Gases

Groups • Representative elements – group A elements • Transition elements – group B elements

Groups • The group tell you the number of valence electrons that the element has • Valence electrons are electrons in the outermost shell of the atom • All group 1 A elements have 1 valence electron. Likewise, all group 8 A elements have 8 valence electrons.

Characteristics • Elements in the same group exhibit similar chemical characteristics due to the fact that they all have the same number of valence electrons. • The most stable number of valence electrons is 8 • This is called an octet

Charges • Every element wants 8 valence electrons to become stable. They will gain or lose valence electrons to form an octet • For example…Group 1 A elements have 1 valence electron. They can either gain 7 electrons to have an octet or lose 1. • What is easier? Lose 1 • If an element loses 1 electron (1 negative charge) what charge will the resulting ion have? +1

Charges • Let’s go to group 7 A. This group has 7 valence electrons • It can either loose 7 or gain 1 • What is the easiest? Gain 1 • What will be the resulting charge if the element gain 1 electron (1 negative charge)? -1

Physical States and Classes of the Elements • The majority of the elements are metals. They occupy the entire left side and center of the periodic table. • Nonmetals occupy the upper-right-hand corner. • Metalloids are located along the boundary between metals and nonmetals

Metals • Metals are elements that have luster, conduct heat and electricity, and usually bend without breaking.

Transition Metals • The elements in Groups 3 through 12 of the periodic table are called the transition elements. • All transition elements are metals. • Many transition metals can have more than one charge

Inner Transition Metals • In the periodic table, two series of elements, atomic numbers 58 -71 and 90 -103, are placed below the main body of the table. • These elements are separated from the main table because putting them in their proper position would make the table very wide. • The elements in these two series are known as the inner transition elements.

Inner Transition Metals • The first series of inner transition elements is called the lanthanides because they follow element number 57, lanthanum. • The second series of inner transition elements, the actinides, have atomic numbers ranging from 90 (thorium, Th) to 103 (lawrencium, Lr).

Non Metals • Although the majority of the elements in the periodic table are metals, many nonmetals are abundant in nature • Most nonmetals don’t conduct electricity, are much poorer conductors of heat than metals, and are brittle when solid. • Many are gases at room temperature; those that are solids lack the luster of metals.

Properties of Metals and Nonmetals

Metalloids • Metalloids have some chemical and physical properties of metals and other properties of nonmetals. • In the periodic table, the metalloids lie along the border between metals and nonmetals.

Electron Dot Structures • An electron dot structure consists of the elemental symbol surrounded by dots which represent valence electrons

Examples • Draw the electron dot structure for Na • Draw the electron dot structure for Al • Draw the electron dot structure for Br

Periodic Trends • Periodic Trends are trends that occur across the periodic table and down the periodic table • They include: atomic radius, Ionization energy, electro negativity, metallic character, and ionic radius
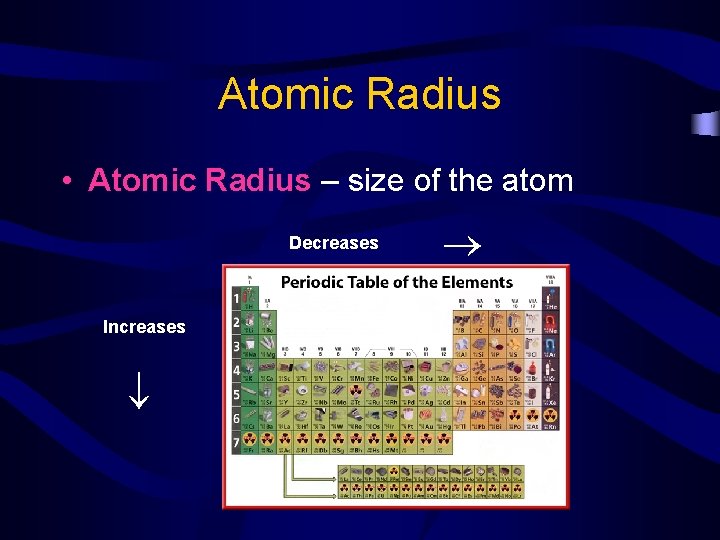
Atomic Radius Decreases Increases • Atomic Radius – size of the atom
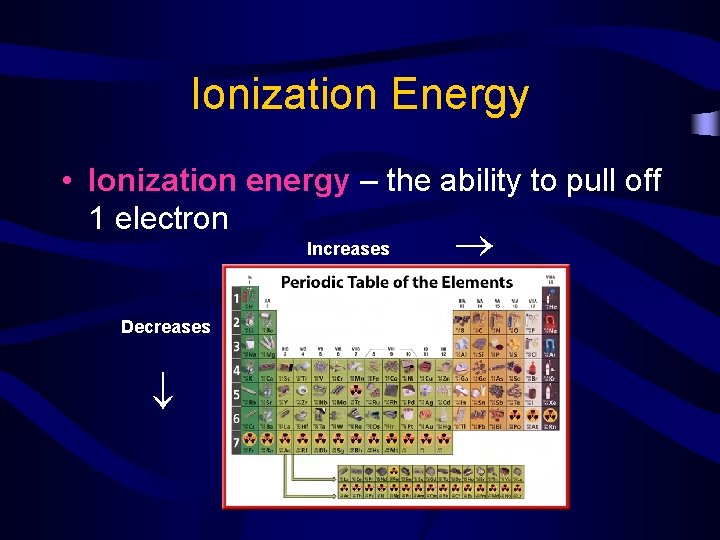
Ionization Energy Increases Decreases • Ionization energy – the ability to pull off 1 electron

Electro negativity Increases Decreases • Electro negativity – the ability of an atom to attract another atom
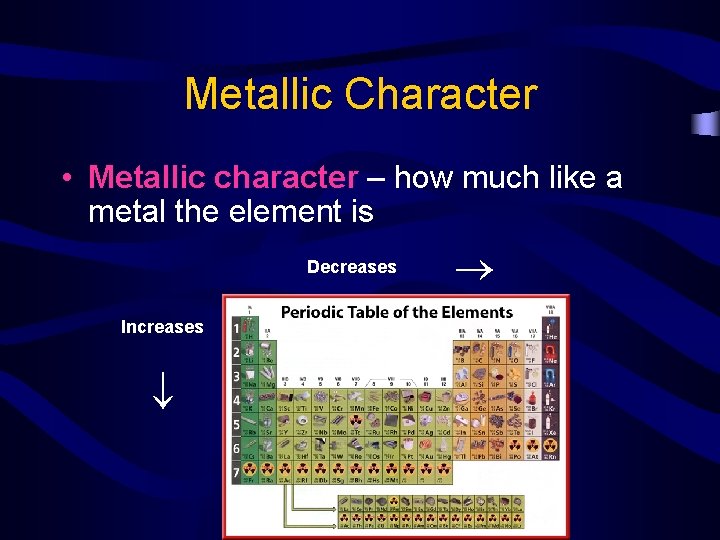
Metallic Character Decreases Increases • Metallic character – how much like a metal the element is

Ionic Radius • When you talk about ionic radius, you are comparing an atom and its ion • When an atom has a negative charge, you have added electrons • Which makes it bigger • For example, which will be larger: • Cl or Cl-1

Ionic Radius • When an atom has a positive charge, you have taken away electrons • Which makes it smaller • For example, which will be larger: • Na or Na+1
- Slides: 25