Chapter 6 The Periodic Table and Periodic Law

Chapter 6: The Periodic Table and Periodic Law

Development of the Periodic Table • 1790 s – Antoine Lavoisier composed a list of the 23 known elements – Included gold, silver, carbon, and oxygen • Electricity which is used to break down compounds into elements led to an “explosion” in chemistry as did the spectrophotometer and the industrial revolution.

• 1864 – John Newlands proposed an organization scheme for the elements – Arranged by increasing atomic mass and noticed that the properties of the elements repeated after every 8 th element (PERIODIC) – See fig 6. 2 page 153 for picture

Meyer, Mendeleev, and Moseley, OH MY!!! • Lothar Meyer and Dmitri Mendeleev showed a connection between atomic mass and elemental properties • Mendeleev published first!!! • Left spaces on the Periodic Table for the unknown elements • By noting trends in the periodic table, he was able to predict the properties of yet to be known elements. • Mendeleev organized the periodic table by atomic mass

Mendeleev’s Periodic Table • http: //z. about. com/d/chemistry/1/0/0/W/mendeleevperiodic. jpg

• Mendeleev was not completely correct – More accurate measurements of atomic mass • Mosely (1913)- arranged elements in order of increasing atomic number – Resulted in clearer patterns of properties • PERIODIC LAW: There is a periodic repetition of chemical and physical properties of the elements when they are arranged by increasing atomic number

The Modern Periodic Table • Groups - the columns of the periodic table (Sometimes called families) • Periods – the rows of the periodic table • SEE PAGE 154 Fig. 6. 4 • Representative Elements (labeled 1 A-8 A) • Transition Elements (labeled 3 B-12 B)

Classifying the Elements (3 Types) 1. Metals (solid, shiny, good conductors) • Group 1 A: Alkali Metals • • Group 2 A: Alkaline Earth Metals • • Most reactive of all metals Also very chemically reactive but not as much as the alkali metals Group 3 A: Transition Metals (main part of table) and Inner Transition Metals (bottom two rows, lanthanide and actinide series)

2. Nonmetals – generally gases, dull, brittle, poor conductors – Group 7 A is called the halogens and are very reactive – Group 8 A is called the Noble Gases and are unreactive 3. Metalloids – Phys. and chem. properties of both metals and nonmetals – Border on the stair-step line – Silicon and Germanium are two most important (comp. chips)


6. 2: Classification of the Elements

• Valence electrons • Found in highest principle energy level • All elements of group 1 A have the same number of valence electrons; therefore, have same chemical properties • Valence electrons by period • The energy level by the valence electrons are found reveals the period • Valence electrons by group • The group number corresponds the number of valence electrons

The s, p, d, and f elements • Review pages 160 -161 in case you had trouble or may be a little confused

6. 3 PERIODIC TRENDS This is a very important section!!!!!

• Many properties on the periodic table change in a very predictable manner • Includes: – Atomic Radius – Ionization Energy – Electronegativity • YOU MUST MEMORIZE THESE!!!!!!

1. Atomic Radius • Atomic size is based on how closely an atom is to it’s neighboring atom • Because the neighboring atom can vary from one substance to another, the size itself tends to vary • For sodium, The atomic radius is defined as half the distance between adjacent nuclei in a crystal of an element

Atomic Radius CONT’D • Trends within the periods – As you move left-right, atomic size decreases – Caused by the increasing positive charge in a nucleus – Each successive element increases in number of electrons and protons – Remain in same principal energy level – The increased nuclear charge pulls the outermost electrons in closer to the nucleus

Atomic Radius CONT’D • Trends within groups – Increase as you move down a group – The nuclear charge increases and electrons are added to higher principal energy levels – Outer electrons are farther from the nucleus

2. Ionic Radius • Atoms can gain or lose electrons to form ions • Because electrons are negatively charged the change in quantity causes there to be a change in the net charge • ION- an atom or bonded group of atoms that has a positive or negative charge – When atoms lose electrons, they become positive and, therefore, are smaller – When atoms gain electrons, they become negative and, therefore, are larger
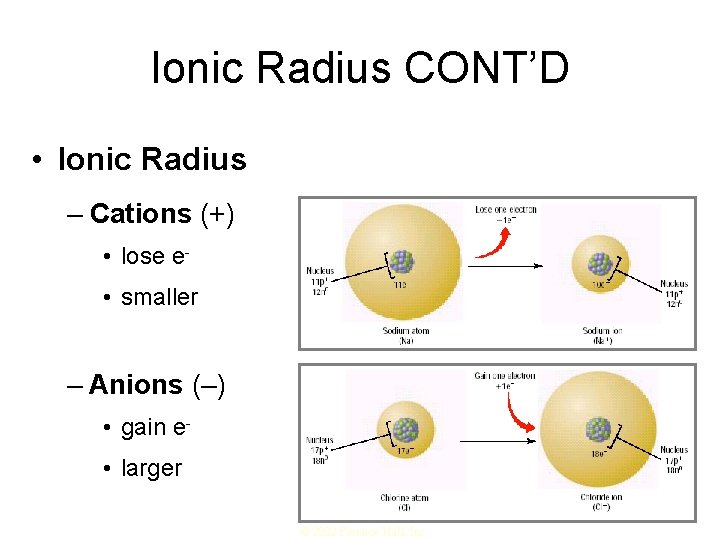
Ionic Radius CONT’D • Ionic Radius – Cations (+) • lose e • smaller – Anions (–) • gain e • larger © 2002 Prentice-Hall, Inc.
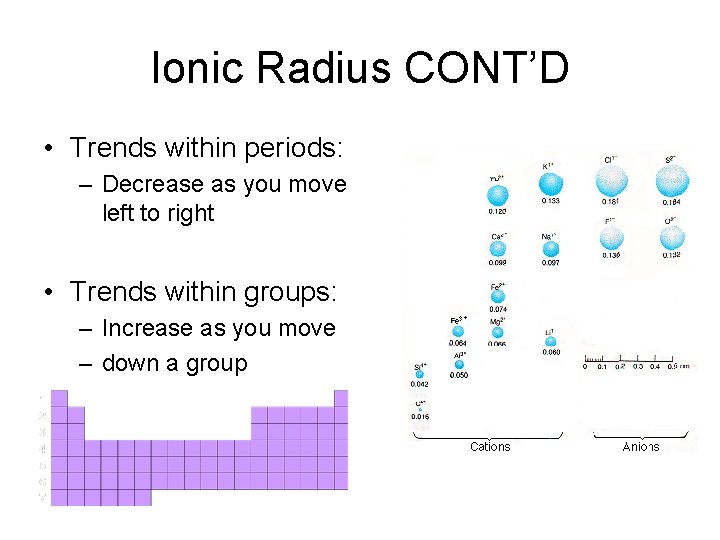
Ionic Radius CONT’D • Trends within periods: – Decrease as you move left to right • Trends within groups: – Increase as you move – down a group

3. Ionization Energy • Defined as the energy required to remove an electron from a gaseous atom • A high ionization value indicates that the atom has a strong hold on its electrons therefore, tend to not form positive ions • Trends within periods: – Increase left to right • Trends within groups: – Decrease down a group • OCTET RULE: – States that atoms tend to lose or gain electrons in order to achieve a set of 8 valence electrons

4. Electronegativity • Indicates the relative ability of atoms to attract electrons in a chemical bond • Calculated based on many factors and are expresses in terms of a value of 4. 0 or less • Units are called Paulings • Fluorine is the most electronegative with a value of 3. 98 and Francium is the least electronegative with a value of 0. 70.
Electronegativity CONT’D • The greater the electronegativity, the more strongly it attracts the bond’s electrons • Trends within periods: – Increases left to right • Trends within groups: – Decreases down the group
- Slides: 24