Eleanor Roosevelt High School ChinSung Lin LESSON 16























































































- Slides: 121

Eleanor Roosevelt High School Chin-Sung Lin LESSON 16 ELECTROSTATICS

Electrostatics is the electricity at rest It involves electric charges, the force between them, and their behavior in the materials

History The word electricity comes from the Greek “elektron” which means “amber” The “amber effect” is what we call static electricity

History Based on the experiment situations, there must be two types of charges. Benjamin Franklin arbitrarily call one positive and another one negative He also argued that when a certain amount of charge is produced on one body, an equal amount of the opposite charge is produced on the other body…

ELECTRIC CHARGE

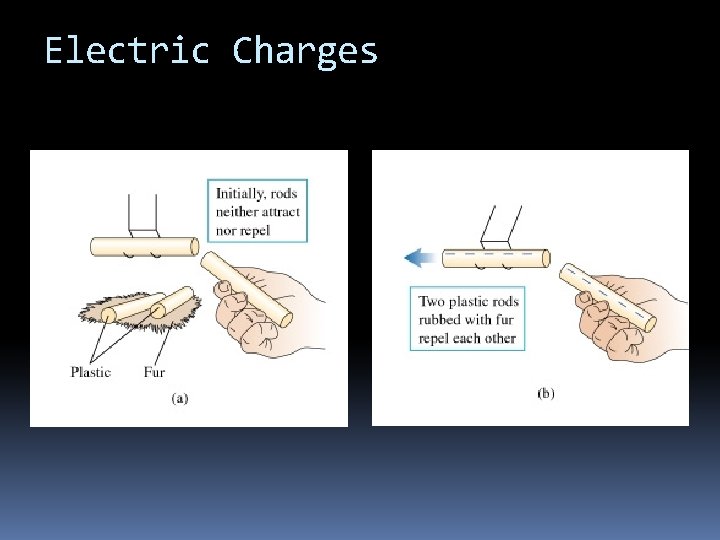
Electric Charges

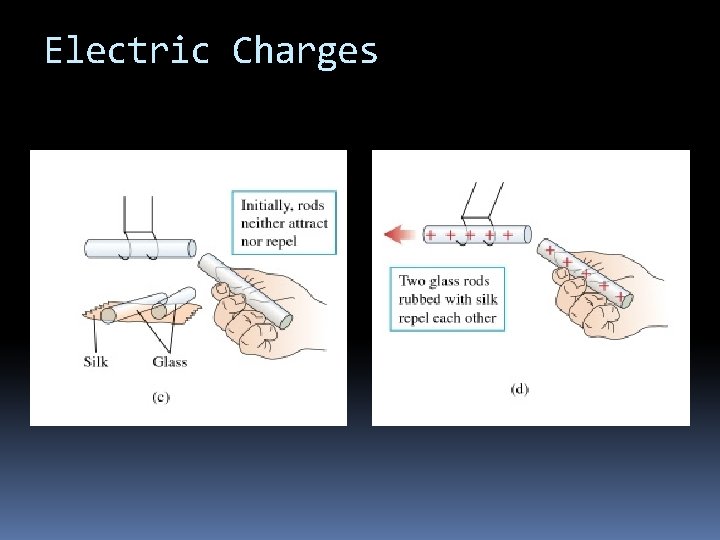
Electric Charges

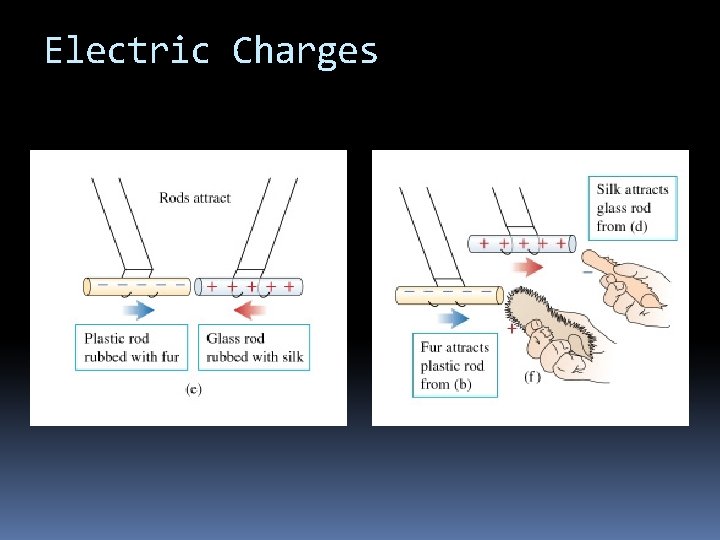
Electric Charges

Electric Charges The fundamental rule of all electrical phenomena is Like charges repel opposite charges attract

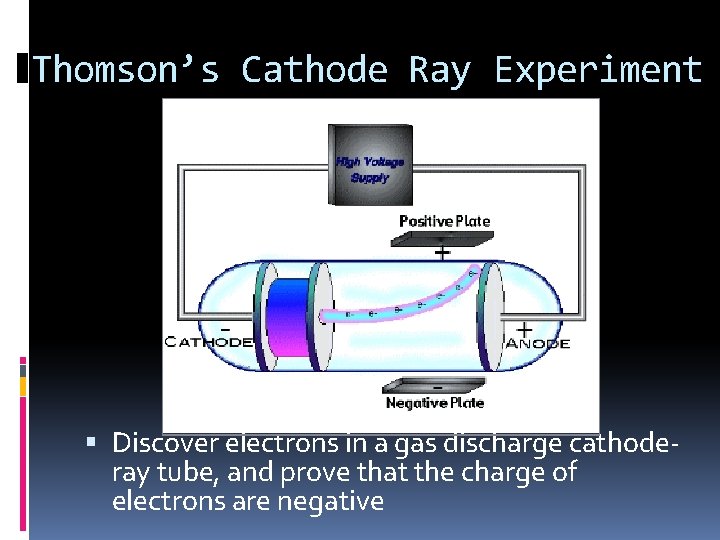
Thomson’s Cathode Ray Experiment Discover electrons in a gas discharge cathoderay tube, and prove that the charge of electrons are negative

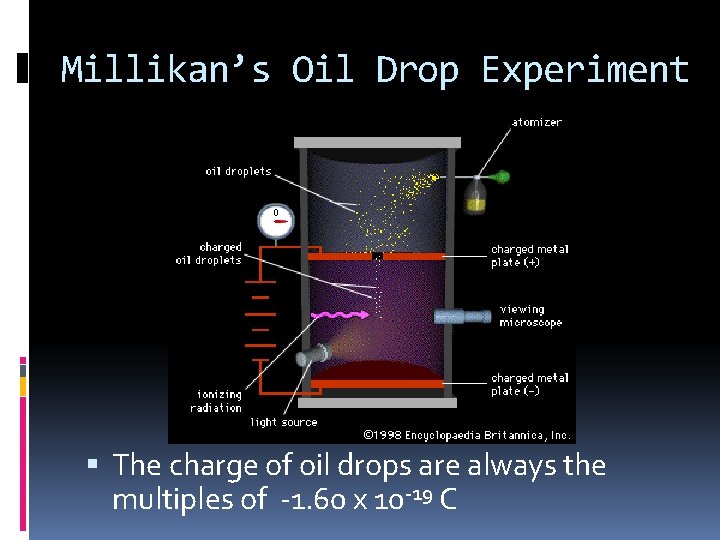
Millikan’s Oil Drop Experiment The charge of oil drops are always the multiples of -1. 60 x 10 -19 C

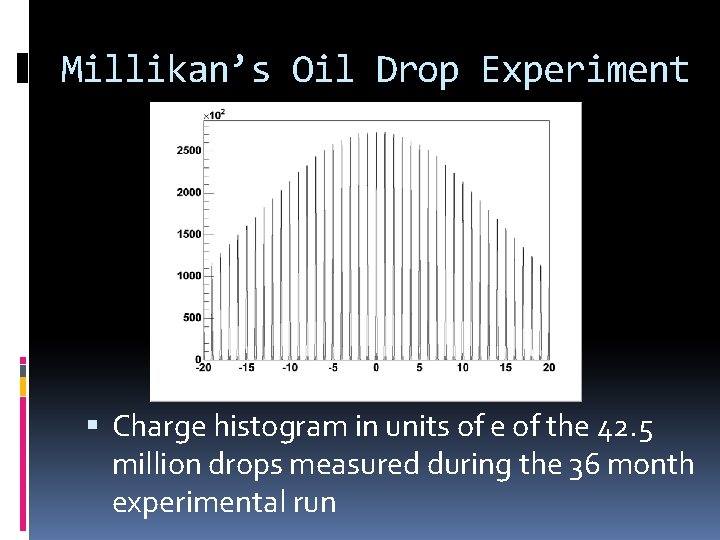
Millikan’s Oil Drop Experiment Charge histogram in units of e of the 42. 5 million drops measured during the 36 month experimental run

Millikan’s Oil Drop Experiment Summary as of January 2007 Total mass throughput for all experiments 351. 4 milligrams of fluid Total drops measured all experiments 105. 6 million

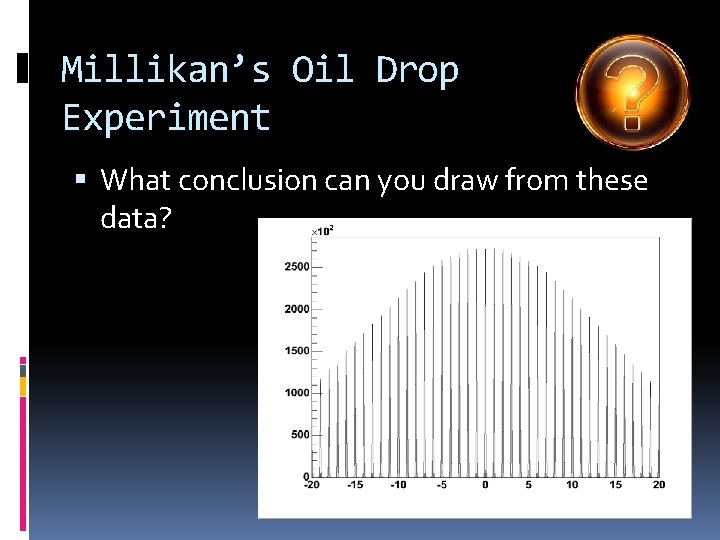
Millikan’s Oil Drop Experiment What conclusion can you draw from these data?

Millikan’s Oil Drop Experiment Electric charges are quantized (discrete) Charges are always multiple of fixed charges No fractional charge particles was found Prove the existence of unit charge

Electric Charges The SI unit of charge is the coulomb (C) The charge of an electron is -1. 60 x 10 -19 C 1 C is the charge of 6. 24 x 1018 electrons Symbol: q, Q

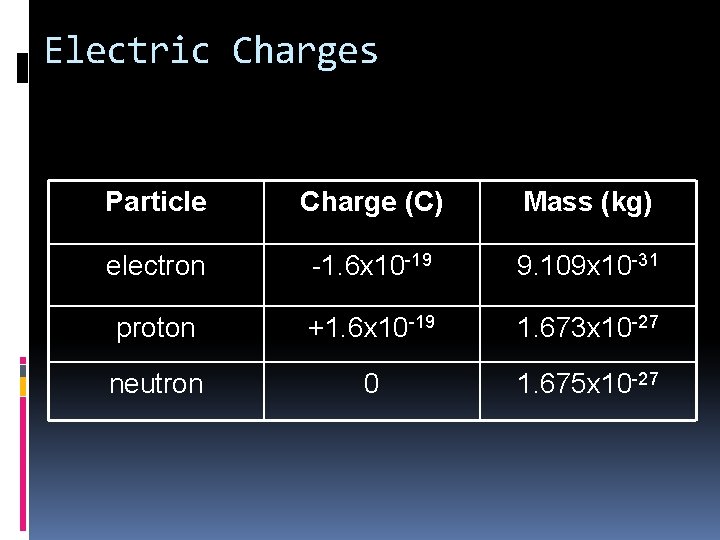
Electric Charges By convention, electrons are negatively charged protons are positively charged neutrons have no charge All electrons are identical All protons are identical All neutrons are identical A proton’s charge is equal in magnitude to the negative charge of the electron

Electric Charges Particle Charge (C) Mass (kg) electron -1. 6 x 10 -19 9. 109 x 10 -31 proton +1. 6 x 10 -19 1. 673 x 10 -27 neutron 0 1. 675 x 10 -27

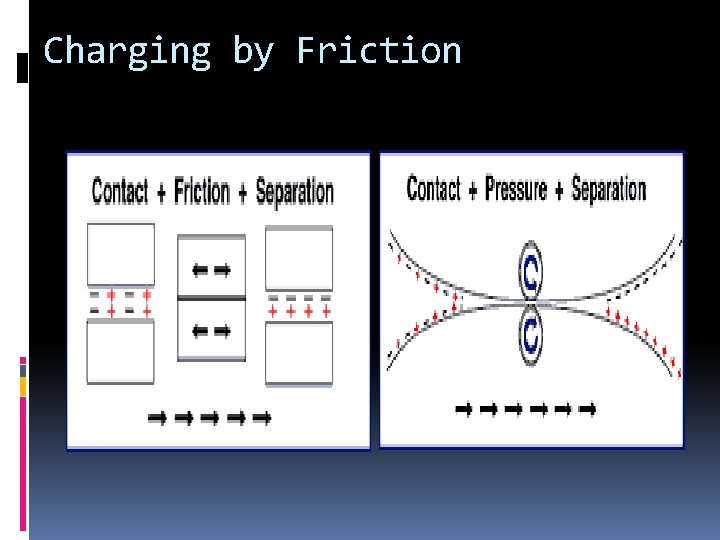
CHARGE BY FRICTION

Charge by Friction Why is the rod got charged simply by rubbing it with fur?

Charge by Friction What decides which will gain or loss electrons during friction?

Charge by Friction What happens when electrons are excited to such an extent that they leave the atoms?

Charge by Friction Why won’t protons move?

Charge by Friction Electrons are being transferred by friction when one material rubs against another The one that gains electrons becomes negative. The one that lose electrons becomes positive Different materials have different tendency to gain or lose electrons

Triboelectric Series The triboelectric series is a list that ranks various materials according to their tendency to gain or lose electrons The tendency of a material to become positive or negative after triboelectric charging has nothing to do with the level of conductivity of the material The greater the relative position, the larger the expected electric charge

Triboelectric Series

Triboelectric Series

Triboelectric Series

Charging by Friction

CHARGE BY CONTACT

Charge by Contact Electrons can be transferred from one material to another by simply touching

Charge by Contact A positively charged metal ball with charge Q contacts with an identical electrically neutral metal ball The charges equally redistribute to both balls Separate these two balls and each one has charge Q/2

Electric Charges Distribution If the object is good conductor, the charges will spread to all part of its surface because the like charges repel each other. Net charge inside is zero

Electric Charges Distribution If we dump a bunch of electrons to the statue of liberty, and the statue is insulated from the ground, where will those electrons go?

Electric Charges Distribution On symmetric objects, charge distributes uniformly, on non-symmetric objects, charges stay at sharp corners

Electric Charges Distribution Why do electrons stay at sharp corners?

Electric Charges Distribution If the object is a poor conductor, charge cannot move, so it remains localized around the contact region

LAW OF CONSERVATION OF CHARGE

Law of Conservation of Charge The net amount of electric charge produced in any process is zero In an isolated system the total charge is conserved Electrons are neither created nor destroyed but are simply transferred from one material to another

Law of Conservation of Charge An object that has unequal numbers of electrons and protons is electrically charged In a neutral atom, there as many electrons as protons, so there is no charge A charged atom is called an ion. An imbalance comes about by adding or removing electrons

CHARGE BY INDUCTION

Charge by Induction Why does the balloon stick to the wall?

Charge by Induction If we bring a charged object near a conducting surface, even without physical contact, electrons will move in the conducting surface

Charge by Induction The introduction of a charge into another body without contact being made Temporary charging of this type produces Temporary Polarity

Charge by Induction Two neutral metal balls A & B are placed next to each other and contact each other Approach a negatively charged rod to ball A and different charges will be induced on each ball

Charge by Induction Separate the two metal balls while the rod is still there Remove the rod and the charges will be redistributed evenly on both balls

AIM: Charge by Induction Do. Now: Started with a neutral ball with metal paint and a negatively charged rod, how can you charge the ball with positive charges by induction?

Charge by Induction Approach a negatively charged rod to a neutral ball with metal paint and different charges will be induced on each side of the ball

Charge by Induction While the rod is still there, ground the opposite side of the ball and electrons will flow to the ground

Charge by Induction A charged object is needed to charge an object by induction (without any contact) The object being charged ends up with a charge which is the opposite of the object being used to charge it A ground must be used to charge on the object. The ground allows for electron movement into or out of the object being charged

Charge by Induction Touch the negatively charged rod with the positive charged ball, then the excess electrons of the rod will flow to the ball Now like charges repel

Charge by Induction Charging by induction is not limited to conductors When a charged rod is brought near an insulator, 0 n side of the atom or molecular is more positive (or negative) than the other side

Charge by Induction This explains why neutral bits of paper are attracted to a charged object or the negatively charged balloon attached to the neutral wooden wall

Charge by Induction Many molecules (H 2 O for example) are electrically polarized in their normal states. The distribution of electric charge is not perfectly even. Such molecules are said to be electric dipoles

Charge by Induction Microwave oven uses oscillating electric field to make the water molecules oscillating. The flipflopping H 2 O molecules impart thermal motion to surrounding food molecules

Lightning The negatively charged bottoms of clouds induced a positive charge on the surface of Earth below Becomes very “negative” Becomes very “positive”

Lightning Rod A metal rod mounted on top of a building and electrically connected to the ground through a wire, to protect the building in the event of lightning

TYPES OF MATERIALS

Types of materials Conductor: a material that transfers charge easily (ex. Metals)

Types of materials Insulator: a material that does not transfer charge easily (ex. glass, water, air, ceramic, nonmetals)

Types of materials Semiconductors: somewhere between 1 & 2 (ex. Silicon, carbon, germanium)

Types of materials Superconductors: some metals become perfect conductors below certain temperatures

ELECTROSTATICS INSTRUMENTS

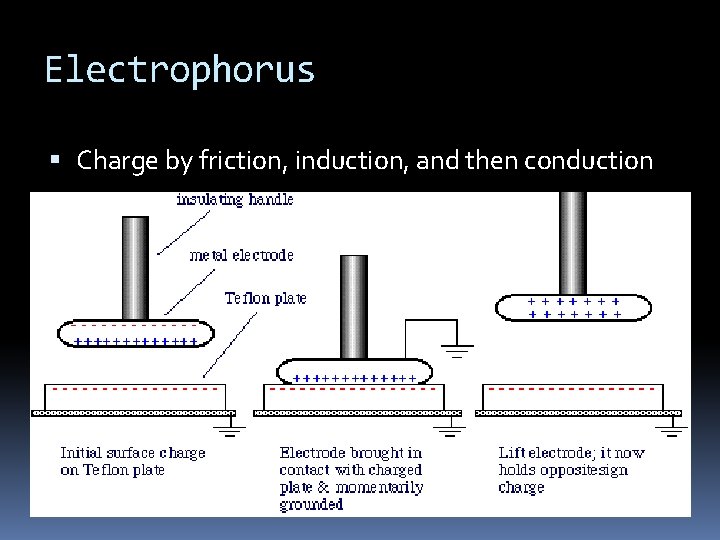
Electrophorus An electrophorus is used to produce electrostatic charge via the process of electrostatic induction It consists of a plastic plate and a metal disc with an insulating handle

Electrophorus Charge by friction, induction, and then conduction

Electroscope

Electroscope used to detect the presence and magnitude of electric charge on a body It consists of a vertical metal rod, from the end of which hang two strips of thin gold leaf A disk or ball terminal is attached to the top of the rod, where the charge to be tested is applied

Electroscope

Electroscope

Electroscope

Electroscope A B

ELECTRIC FORCE

Electric Force— The Coulomb’s Law Describing the electrostatic force between electrically charged particles Published in 1785 by French physicist Charles Augustin de Coulomb Torsion balance experiment

Electric Force— The Coulomb’s Law Torsion Balance Cylindrical Glass case & tube Torsion head & fiber Needle & spheres Removable spheres Scale

Electric Force— The Coulomb’s Law Torsion Balance Experiment (Video 4 minutes) Write down: What’s the purpose? Three major steps What’s the conclusion?

Electric Force— The Coulomb’s Law The electric force between two charges is proportional to the product of the two charges and Fe ~ q 1 q 2

Electric Force— The Coulomb’s Law The electric force between two charges is inversely proportional to the square of the distance between the charges Fe ~ 1/r 2

Electric Force— The Coulomb’s Law Fe ~ q 1 q 2

Electric Force— The Coulomb’s Law Fe ~ q 1 q 2 Fe ~ 2 1/r

Electric Force— The Coulomb’s Law Fe ~ q 1 q 2 Fe ~ 2 1/r q 1 q 2 2 r

Electric Force— The Coulomb’s Law Fe ~ q 1 q 2 Fe ~ 2 1/r q 1 q 2 Fe ~ 2 r q 1 q 2 Fe = k r 2

Electric Force— The Coulomb’s Law Fe = k Fe: k: q 1, q 2: r: q 1 q 2 2 r electric force (N) electrostatic constant (N m 2/C 2) charge (C) distance between charges (m)

Electric Force— The Coulomb’s Law Electrostatic constant k Fe = k q 1 q 2 r 2 k= k — A very large constant 8. 99 x 109 What’s the unit of k? (1 minute) Fe 2 r q 1 q 2

Electric Force— The Coulomb’s Law Electrostatic constant k Fe = k q 1 q 2 r 2 k= Fe 2 r q 1 q 2 k — A very large constant 8. 99 x 109 N m 2/C 2 What’s the unit of k? 2 Nm [k]=[ C 2 ]

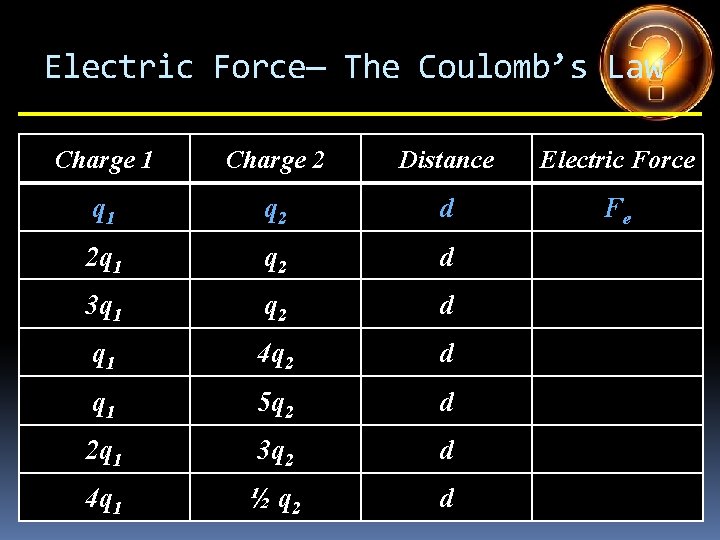
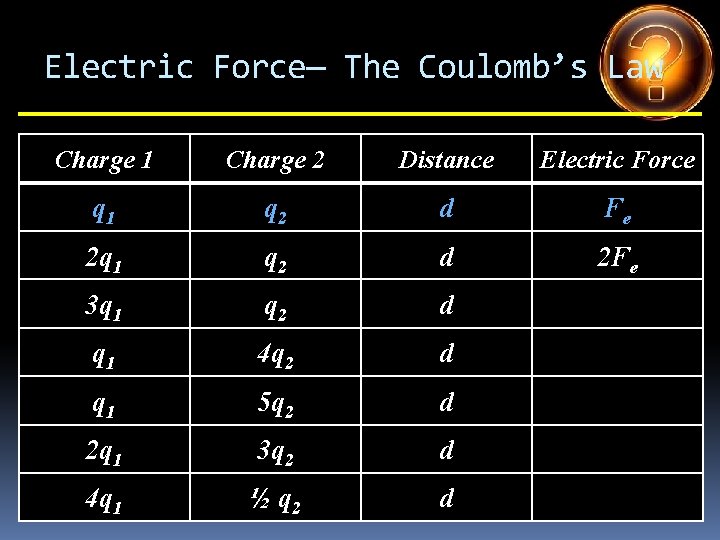
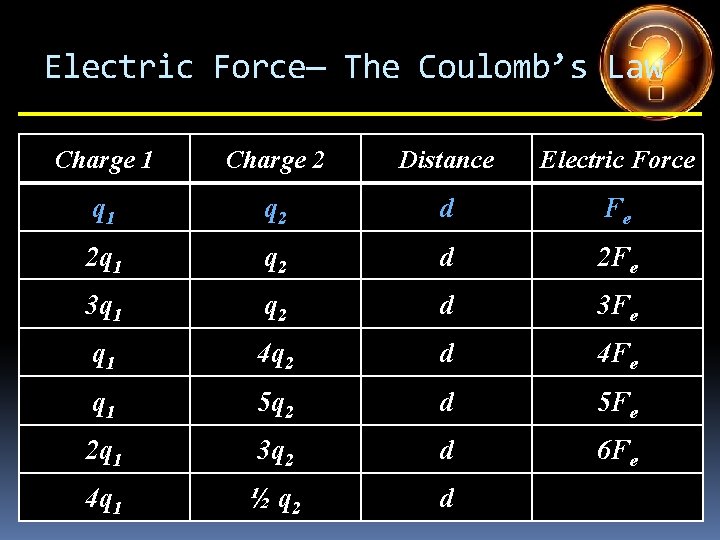
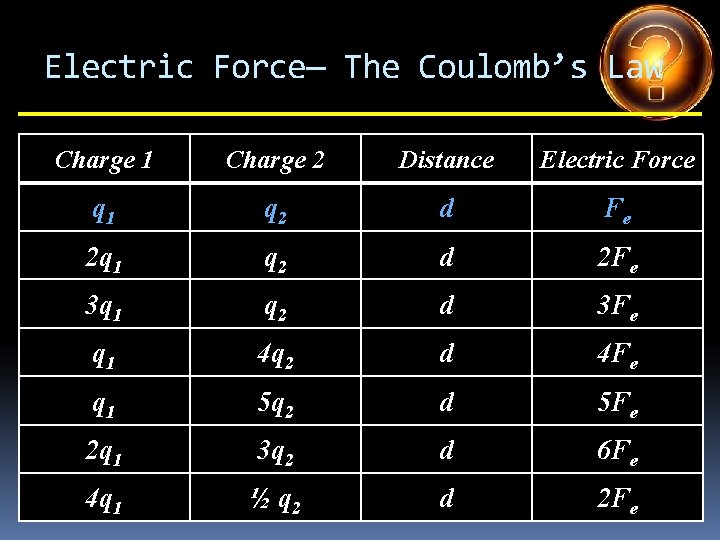
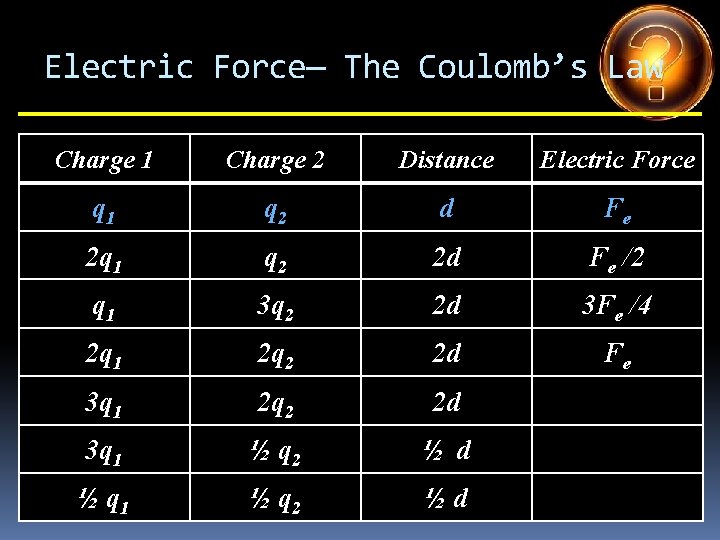
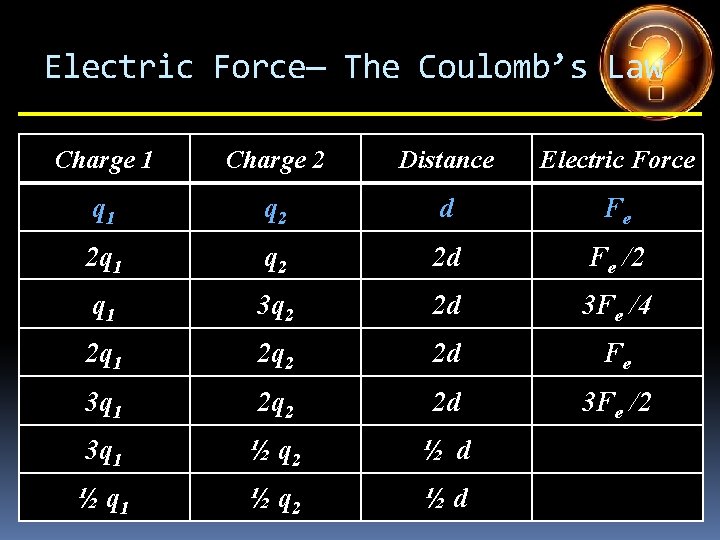
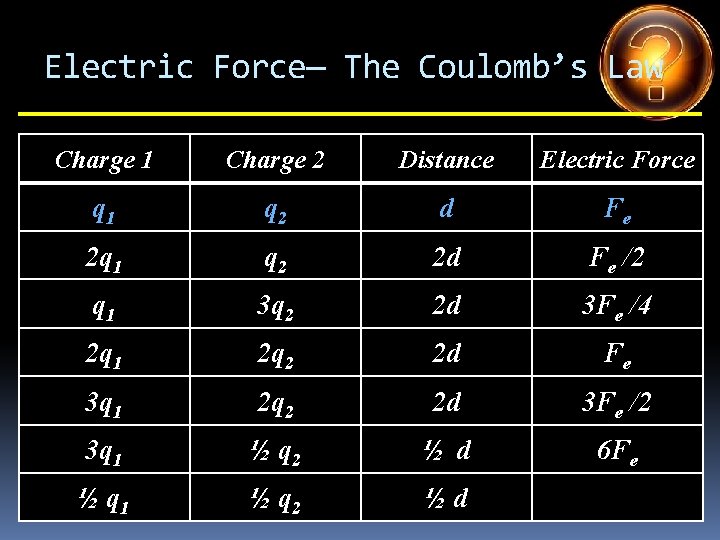
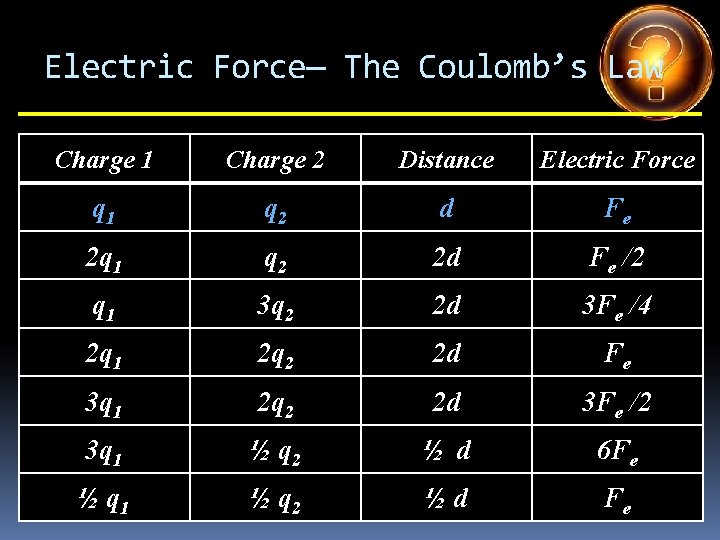
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 3 q 1 q 2 d q 1 4 q 2 d q 1 5 q 2 d 2 q 1 3 q 2 d 4 q 1 ½ q 2 d

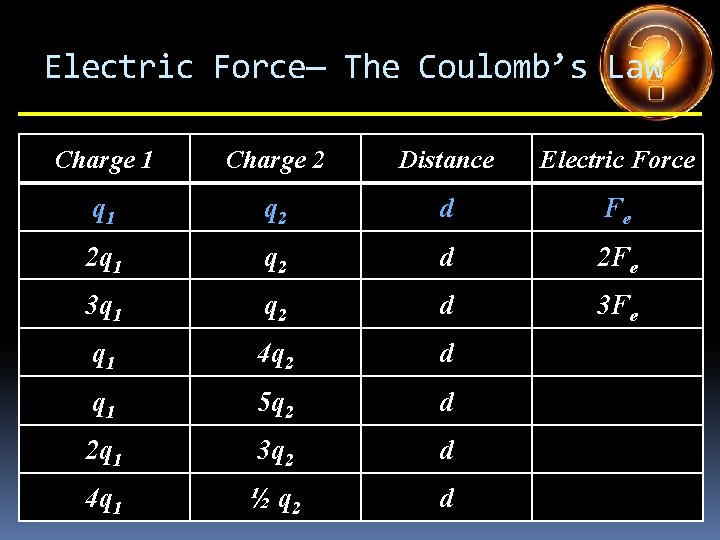
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d q 1 4 q 2 d q 1 5 q 2 d 2 q 1 3 q 2 d 4 q 1 ½ q 2 d

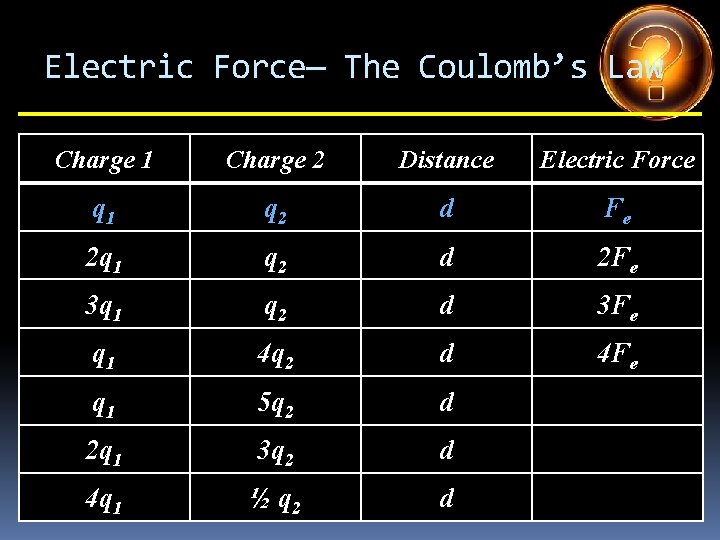
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d 3 Fe q 1 4 q 2 d q 1 5 q 2 d 2 q 1 3 q 2 d 4 q 1 ½ q 2 d

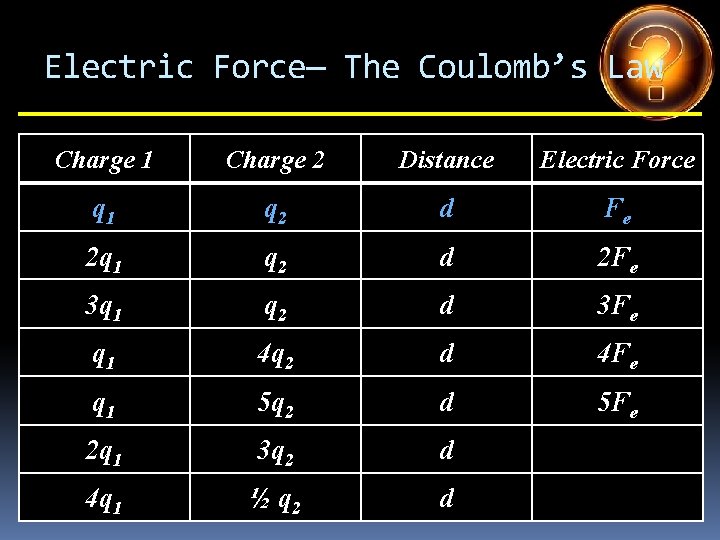
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d 3 Fe q 1 4 q 2 d 4 Fe q 1 5 q 2 d 2 q 1 3 q 2 d 4 q 1 ½ q 2 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d 3 Fe q 1 4 q 2 d 4 Fe q 1 5 q 2 d 5 Fe 2 q 1 3 q 2 d 4 q 1 ½ q 2 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d 3 Fe q 1 4 q 2 d 4 Fe q 1 5 q 2 d 5 Fe 2 q 1 3 q 2 d 6 Fe 4 q 1 ½ q 2 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 d 2 Fe 3 q 1 q 2 d 3 Fe q 1 4 q 2 d 4 Fe q 1 5 q 2 d 5 Fe 2 q 1 3 q 2 d 6 Fe 4 q 1 ½ q 2 d 2 Fe

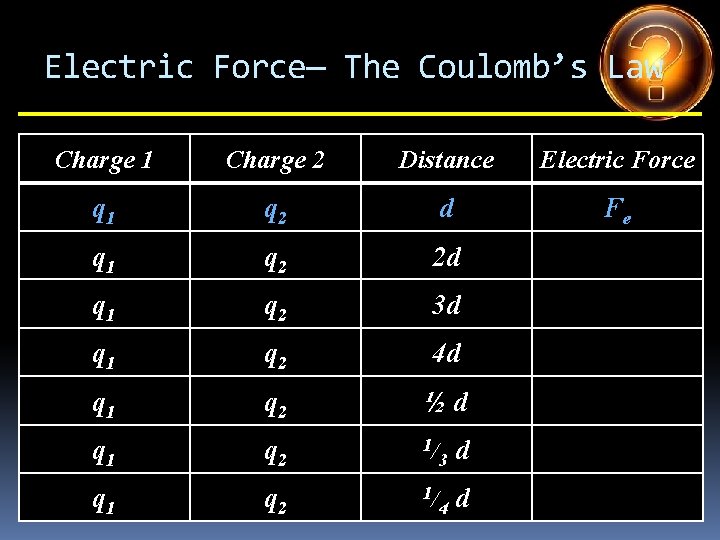
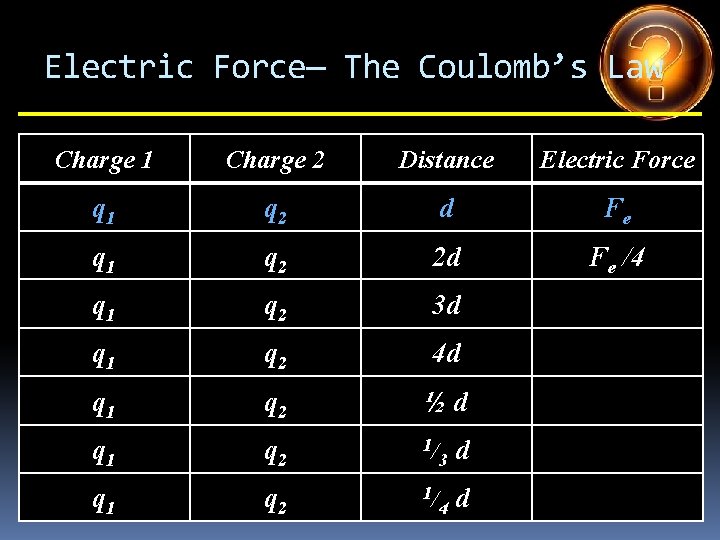
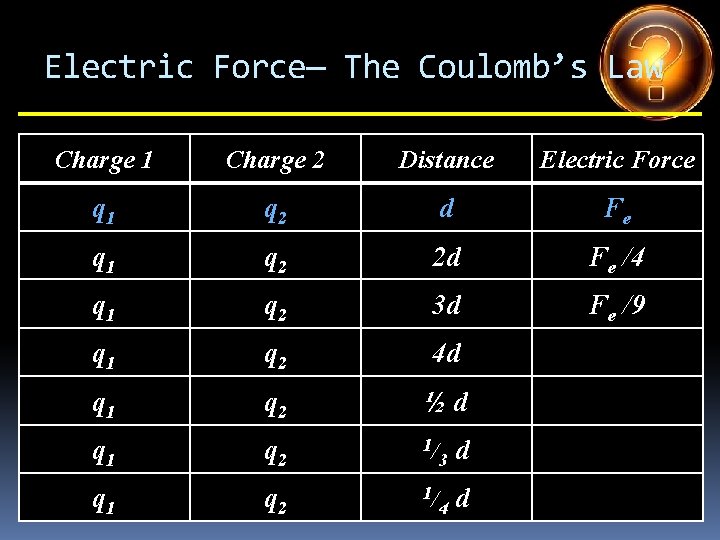
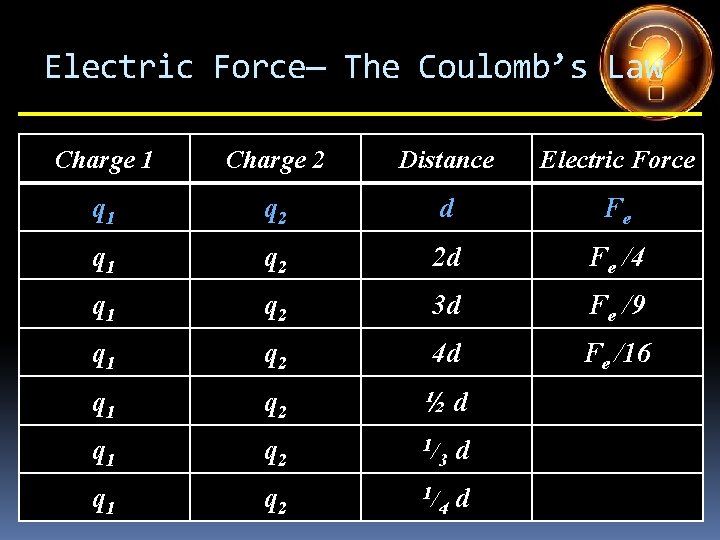
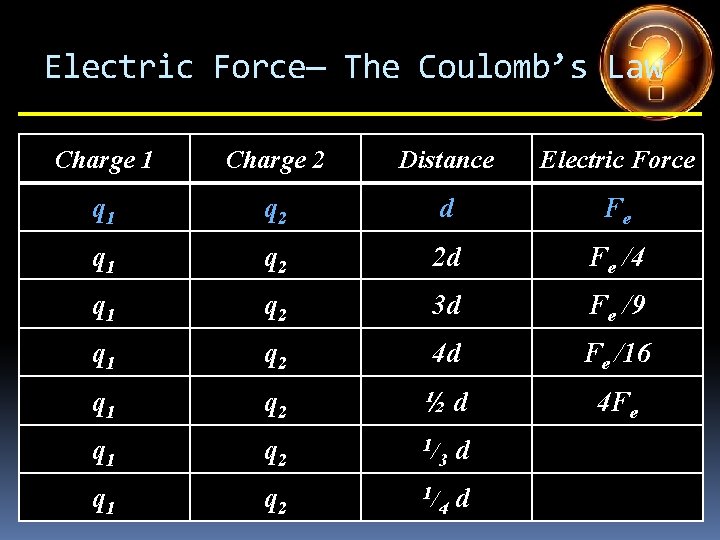
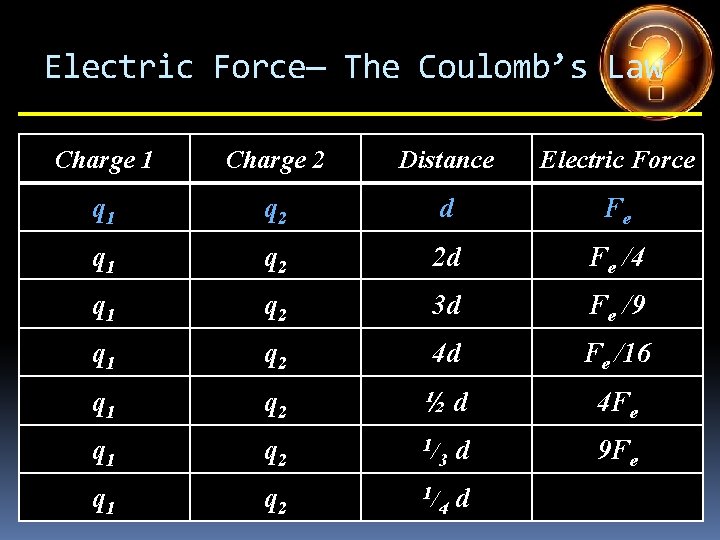
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d q 1 q 2 3 d q 1 q 2 4 d q 1 q 2 ½d q 1 q 2 1/ 3 d q 1 q 2 1/ 4 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d q 1 q 2 4 d q 1 q 2 ½d q 1 q 2 1/ 3 d q 1 q 2 1/ 4 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d Fe /9 q 1 q 2 4 d q 1 q 2 ½d q 1 q 2 1/ 3 d q 1 q 2 1/ 4 d

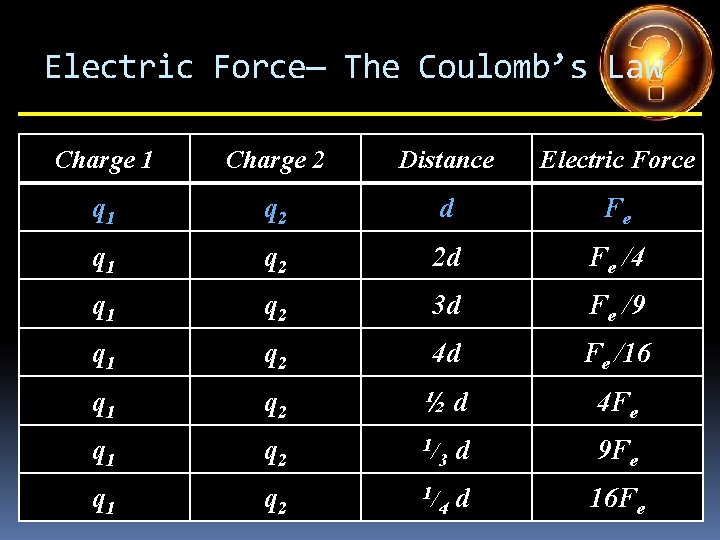
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d Fe /9 q 1 q 2 4 d Fe /16 q 1 q 2 ½d q 1 q 2 1/ 3 d q 1 q 2 1/ 4 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d Fe /9 q 1 q 2 4 d Fe /16 q 1 q 2 ½d 4 Fe q 1 q 2 1/ 3 d q 1 q 2 1/ 4 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d Fe /9 q 1 q 2 4 d Fe /16 q 1 q 2 ½d 4 Fe q 1 q 2 1/ 3 d 9 Fe q 1 q 2 1/ 4 d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe q 1 q 2 2 d Fe /4 q 1 q 2 3 d Fe /9 q 1 q 2 4 d Fe /16 q 1 q 2 ½d 4 Fe q 1 q 2 1/ 3 d 9 Fe q 1 q 2 1/ 4 d 16 Fe

AIM: Electric Force— The Coulomb’s Law Do. Now: (3 minutes) 1. Write down the “Coulomb’s Law”. 2. The particle A carries 1. 2 x 10 -4 C, the particle B carries 2. 4 x 10 -4 C, and the particle C carries 9. 7 x 10 -8 C. The three particles form a equilateral triangle with the measure of the side 0. 45 m. What’s the ratio of the repelling forces between A-C and B-C?

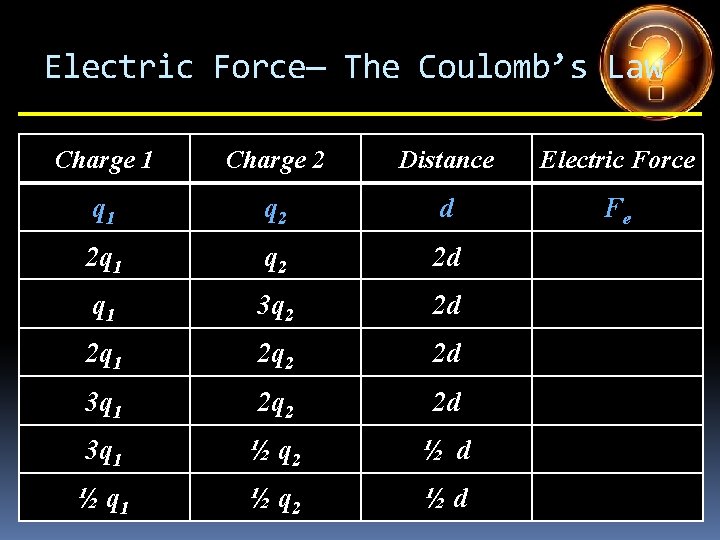
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d q 1 3 q 2 2 d 2 q 1 2 q 2 2 d 3 q 1 ½ q 2 ½ d ½ q 1 ½ q 2 ½d

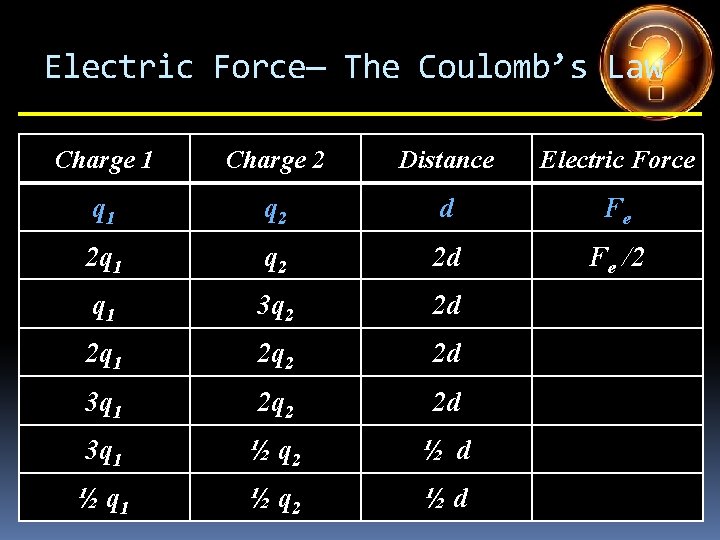
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 2 q 1 2 q 2 2 d 3 q 1 ½ q 2 ½ d ½ q 1 ½ q 2 ½d

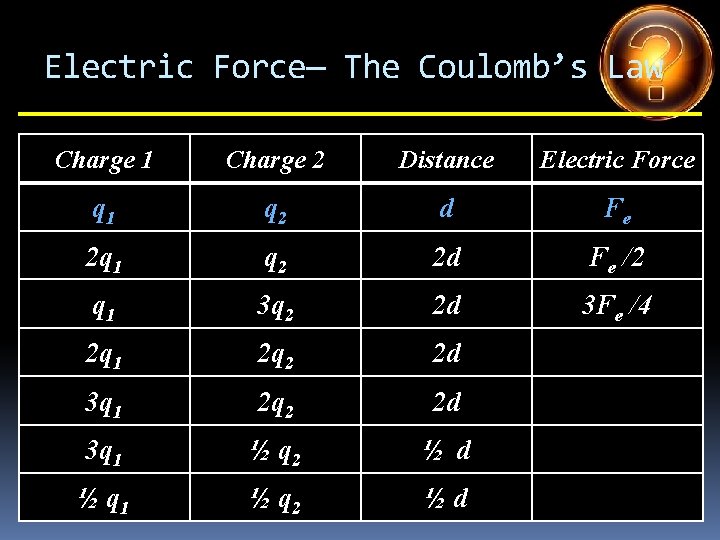
Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 3 Fe /4 2 q 1 2 q 2 2 d 3 q 1 ½ q 2 ½ d ½ q 1 ½ q 2 ½d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 3 Fe /4 2 q 1 2 q 2 2 d Fe 3 q 1 2 q 2 2 d 3 q 1 ½ q 2 ½ d ½ q 1 ½ q 2 ½d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 3 Fe /4 2 q 1 2 q 2 2 d Fe 3 q 1 2 q 2 2 d 3 Fe /2 3 q 1 ½ q 2 ½ d ½ q 1 ½ q 2 ½d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 3 Fe /4 2 q 1 2 q 2 2 d Fe 3 q 1 2 q 2 2 d 3 Fe /2 3 q 1 ½ q 2 ½ d 6 Fe ½ q 1 ½ q 2 ½d

Electric Force— The Coulomb’s Law Charge 1 Charge 2 Distance Electric Force q 1 q 2 d Fe 2 q 1 q 2 2 d Fe /2 q 1 3 q 2 2 d 3 Fe /4 2 q 1 2 q 2 2 d Fe 3 q 1 2 q 2 2 d 3 Fe /2 3 q 1 ½ q 2 ½ d 6 Fe ½ q 1 ½ q 2 ½d Fe

Electric Force— The Coulomb’s Law Determine the electrical force of attraction between two balloons with separate charges of 3. 5 x 10 -8 C and -2. 9 x 10 -8 C when separated a distance of 0. 65 m.

Electric Force— The Coulomb’s Law Determine the electrical force of attraction between two balloons with separate charges of 3. 5 x 10 -8 C and -2. 9 x 10 -8 C when separated a distance of 0. 65 m. Fe = k q 1 x q 2/r 2 Fe = 8. 99 x 109 Nm 2/C 2 x (3. 5 x 10 -8 C)(-2. 9 x 10 -8 C) / (0. 65 m)2 = -2. 16 x 10 -5 N

Electric Force Exercise Each of the two identical hot-air balloons acquires a charge of 3. 2 x 10 -6 C on its surface as it travels through the air. How far apart are the balloons if the electrostatic force between them is 4. 5 x 10 -2 N?

Electric Force Exercise Each of the two identical hot-air balloons acquires a charge of 3. 2 x 10 -6 C on its surface as it travels through the air. How far apart are the balloons if the electrostatic force between them is 4. 5 x 10 -2 N? Fe = k q 1 x q 2/r 2 4. 5 x 10 -2 N = 8. 99 x 109 Nm 2/C 2 x (3. 2 x 10 -6 C) / r 2 r = 1. 43 m

Electrical & Gravitational Forces Law of Universal Gravitation m 1 m 2 Fg = G 2 r Fg: gravitational force (N) G: Universal gravitational constant, 6. 77 x 10 -11 N m 2/kg 2 m 1, m 2: mass (kg) r: distance between masses (m)

Electrical & Gravitational Forces Electric Force vs. Gravitational Forces Force Electric Force Fe Gravitational Force Fg

Electrical & Gravitational Forces Law of Universal Gravitation Fg = G m 1 m 2 r 2 Coulomb’s Law Fe = k q 1 q 2 r 2

Electric Force Electrostatic constant (k) = 8. 99 x 109 Nm 2/C 2 k is a very, very large number! Compare to G = 6. 67 x 10 -11 N m 2/kg 2 Electrical force is much stronger than the gravity The reason why we don’t feel it most of the time is because that the attracting force and repelling force cancel each other

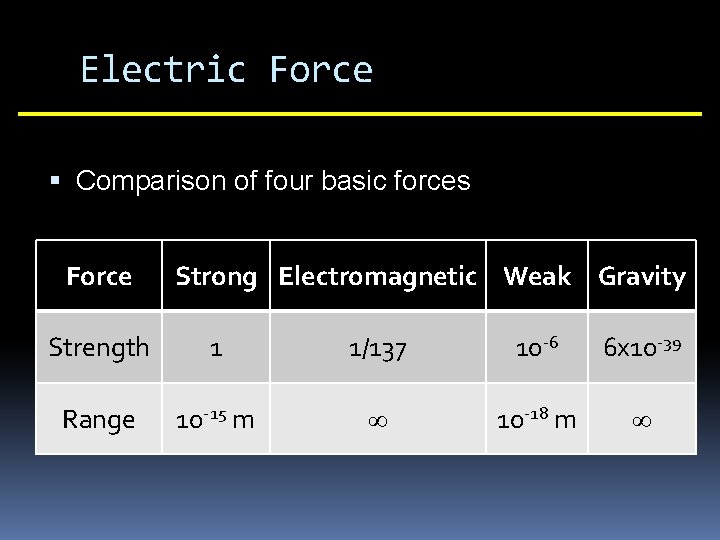
Electric Force Comparison of four basic forces Force Strong Electromagnetic Weak Gravity Strength 1 1/137 10 -6 6 x 10 -39 Range 10 -15 m ∞ 10 -18 m ∞

Electric Force Model of a helium atom: Based on the atomic model proposed by Rutherford and Bohr, a positively charged nucleus is surrounded by electrons Electrons are attracted to protons in the nucleus, but electrons repel other electrons Model of a Helium atom - + + -

Electric Force Why don’t protons pull the oppositely charged electrons into the nucleus? - + + - Model of a Helium atom

Electric Force The reason why don’t protons pull the oppositely charged electrons into the nucleus is in the domain of quantum physics. An electron behaves like a wave and has to occupy a certain amount of space related to its wavelength - + + - Model of a Helium atom

Electric Force Why the protons in the nucleus do not mutually repel and fly apart? - + + - Model of a Helium atom

Electric Force The reason why the protons in the nucleus do not mutually repel and fly apart is that in addition to electrical forces in the nucleus, there are even stronger forces (strong forces) that are non-electrical in nature - + + - Model of a Helium atom

The End