CHEMISTRY OF MATCHES P 4 S 3 KCl































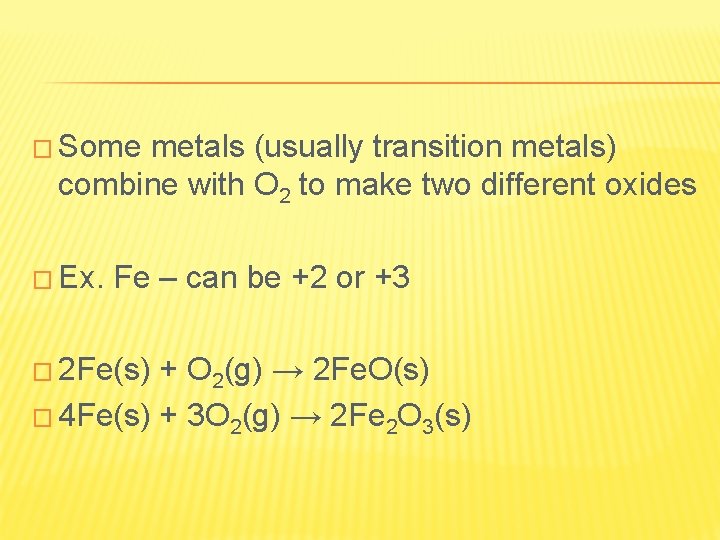



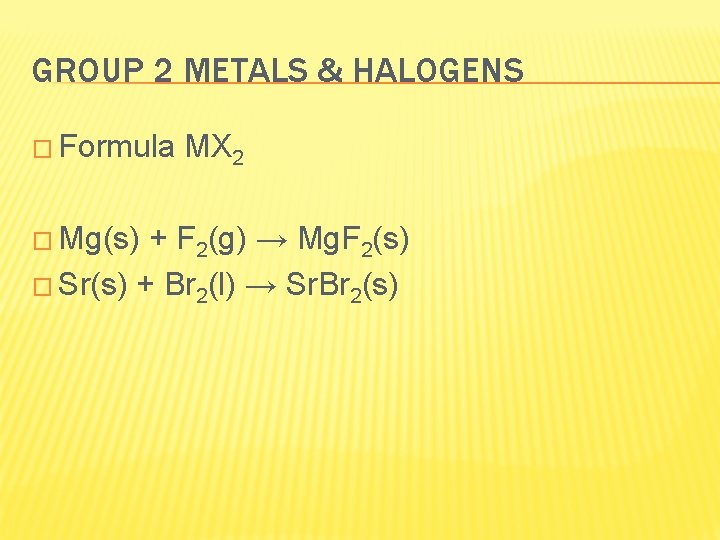





















- Slides: 71

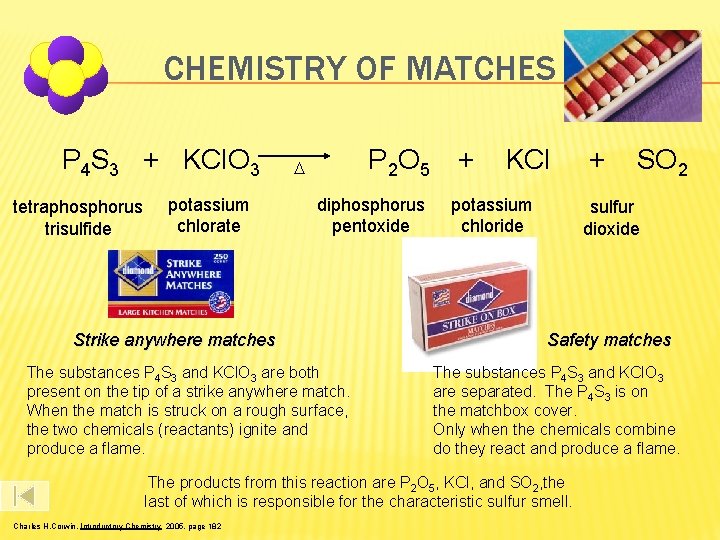
CHEMISTRY OF MATCHES P 4 S 3 + KCl. O 3 tetraphosphorus trisulfide potassium chlorate P 2 O 5 D diphosphorus pentoxide Strike anywhere matches The substances P 4 S 3 and KCl. O 3 are both present on the tip of a strike anywhere match. When the match is struck on a rough surface, the two chemicals (reactants) ignite and produce a flame. + KCl potassium chloride SO 2 sulfur dioxide Safety matches The substances P 4 S 3 and KCl. O 3 are separated. The P 4 S 3 is on the matchbox cover. Only when the chemicals combine do they react and produce a flame. The products from this reaction are P 2 O 5, KCl, and SO 2, the last of which is responsible for the characteristic sulfur smell. Charles H. Corwin, Introductory Chemistry 2005, page 182 +

Chemical Equations and Reactions CHAPTER 8

Describing Chemical Reactions SECTION 1

� Chemical reaction process by which one or more substances are changed into one or more different substances � Represented by chemical equations represents, with symbols and formulas, the identities and relative amounts of the reactants and products in a chemical reaction (NH 4)2 Cr 2 O 7 (s) N 2(g) + Cr 2 O 3(s) + 4 H 2 O(g)

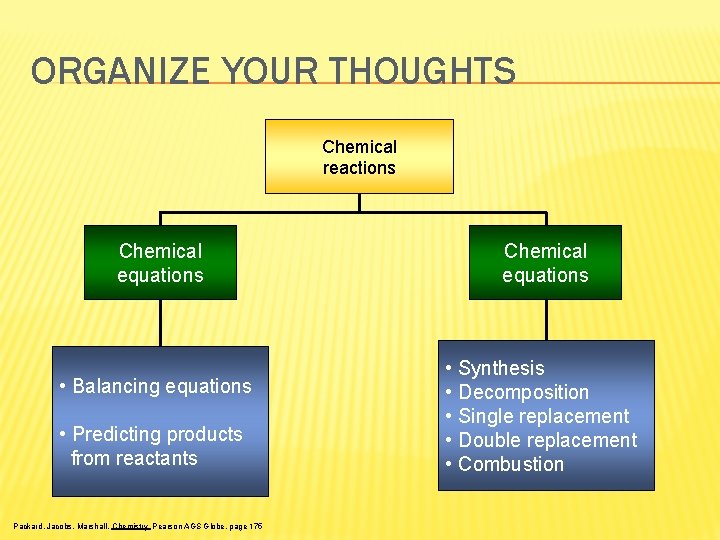
ORGANIZE YOUR THOUGHTS Chemical reactions Chemical equations • Balancing equations • Predicting products from reactants Packard, Jacobs, Marshall, Chemistry Pearson AGS Globe, page 175 Chemical equations • Synthesis • Decomposition • Single replacement • Double replacement • Combustion

CHEMICAL EQUATIONS aluminum oxide reactants Depict the kind of reactants and products product and their relative amounts in a reaction. 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) The letters (s), (g), and (l) are the physical states of compounds. The numbers in the front are called stoichiometric coefficients

CHEMICAL EQUATIONS 4 Al(s) + 3 O 2(g) aluminum oxide sandpaper 2 Al 2 O 3(s) 4 g Al + 3 g O 2 yield 2 g Al 2 O 3 This equation means: 4 Al atoms + 3 O 2 molecules yield 2 molecules of Al 2 O 3 or 4 Al moles + 3 O 2 moles yield 2 moles of Al 2 O 3 4 mol Al@27 g/mol 3 mol O 2@32 g/mol 108 g + 96 g 2 mol Al 2 O 3@102 g/mol = 204 g

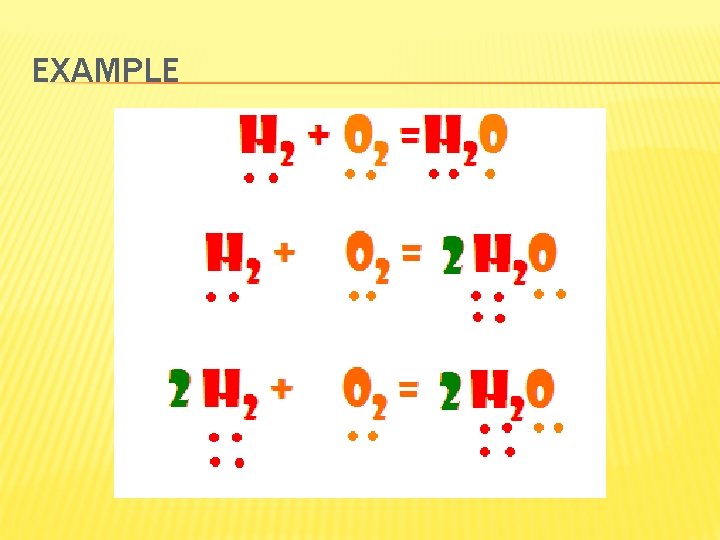
FORMULA EQUATIONS � Formula equation represents reactants and products of chemical reaction by their symbols or formulas CH 4(g) + O 2(g) CO 2(g) + H 2 O(g) � Is the law of conservation of mass satisfied here?

RULES TO BALANCING EQUATIONS l Rule 1 - the superscript or the two in CO 2 cannot be changed l Rule 2 - You can add a coefficient (the two) in 2 H 2 O l Rule 3 - the atoms on both sides must be equal

EXAMPLE

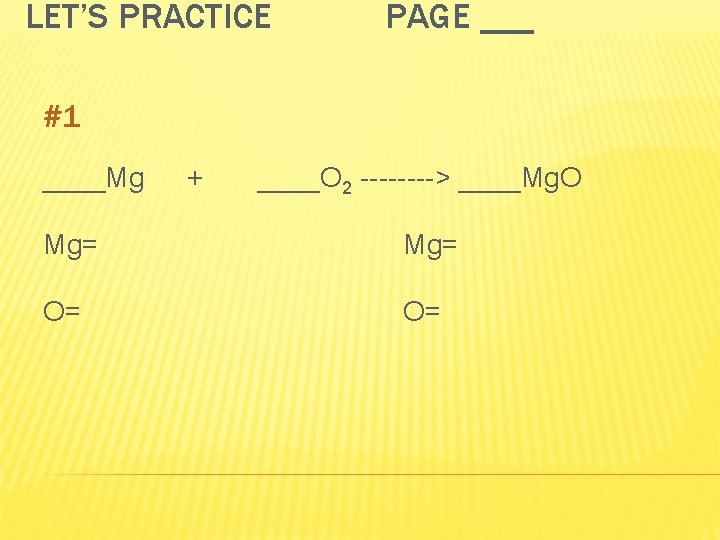
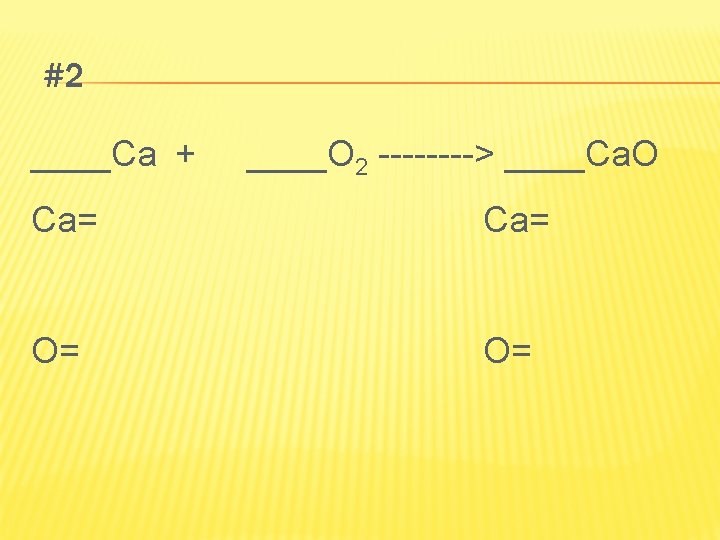
LET’S PRACTICE PAGE ___ #1 ____Mg + ____O 2 ----> ____Mg. O Mg= O= O=





GUIDELINES FOR BALANCING EQUATIONS � Balance the different types of atoms one at a time. � First balance the atoms of elements that are combined and that appear only once on each side of the equation. � Balance polyatomic ions that appear on both sides as single units. � Balance H and O atoms (or any other lone atoms) last.

DO NOT!. . . � DO NOT WRITE INCORRECT FORMULAS, THIS WILL MESS UP YOUR BALANCING � DO NOT CHANGE SUBSCRIPTS IN FORMULAS TO BALANCE THE EQUATION!

DO!!!!. . . � When you think you have balanced the equation, COUNT THE NUMBERS OF EACH TYPE OF ATOM ON EITHER SIDE OF THE EQUATION 2 H 2 O(l) 2 H 2(g) + O 2(g) H O Reactant side Product side 4 2

SAMPLE PROBLEM � The reaction of zinc with aqueous hydrochloric acid produces a solution of zinc chloride and hydrogen gas. Write a balanced equation for the reaction.

1. Write the word equation Zinc + hydrochloric acid zinc chloride + hydrogen 2. Write the formula equation Zn(s) + HCl(aq) Zn. Cl 2(aq) + H 2(g)

ADJUST THE COEFFICIENTS Zn(s) + HCl(aq) Zn. Cl 2(aq) + H 2(g) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) Count atoms: Zn – 1 on left, 1 on right H – 2 on left, 2 on right Cl – 2 on left, 2 on right

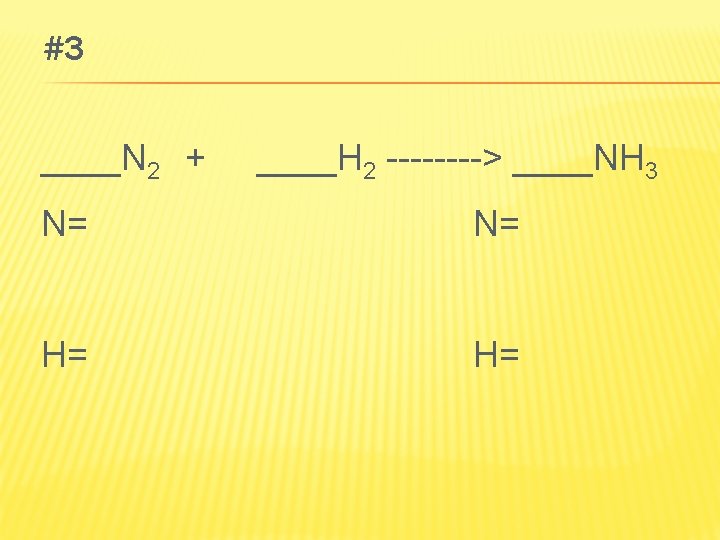
PRACTICE PROBLEM 1 A � Write word, formula, and balanced chemical equations for magnesium and hydrochloric acid react to produce magnesium chloride and hydrogen. � Magnesium + hydrochloric acid magnesium chloride + hydrogen � Mg + HCl Mg. Cl 2 + H 2 � Mg + 2 HCl Mg. Cl 2 + H 2

PRACTICE PROBLEM 1 B � Write word, formula, and balanced chemical equations for silicon dioxide and hydrofluoric acid reacting to produce silicon tetrafluoride and water. � Silicon dioxide+ hydrofluoric acid silicon tetrafluoride + water � Si. O 2+ HF Si. F 4 + H 2 O � Si. O 2+ 4 HF Si. F 4 + 2 H 2 O

PRACTICE PROBLEM 2 � Write word, formula and balanced equations for aqueous nitric acid reacts with solid magnesium hydroxide to produce aqueous magnesium nitrate and water. � Nitric acid + magnesium hydroxide magnesium nitrate + water � HNO 3(aq) + Mg(OH)2(s) Mg(NO 3)2(aq) + H 2 O(l) � 2 HNO 3(aq) + Mg(OH)2(s) Mg(NO 3)2(aq) + 2 H 2 O(l)

PRACTICE PROBLEM 3 � Ammonium sulfate crystals are made by treating ammonia gas, often a byproduct from coke-ovens, with aqueous sulfuric acid: � 2 NH 3(g) + H 2 SO 4(aq) → (NH 4)2 SO 4(s)


PRACTICE PROBLEM 4 � Aluminum sulfate and calcium hydroxide are used in a water-purification process. When added to water, they dissolve and react to produce 2 insoluble products, aluminum hydroxide and calcium sulfate. Write a balanced equation for this reaction. Al 2(SO 4)3(aq) + 3 Ca(OH)2(aq) 2 Al(OH)3(s) + 3 Ca. SO 4(s)

PRACTICE PROBLEM 5 � Write balanced chemical equations for the following reaction: Solid sodium combines with chlorine gas to produce solid sodium chloride. � 2 Na(s) + Cl 2(g) → 2 Na. Cl(s)

PRACTICE PROBLEM 6 � When solid copper reacts with aqueous silver nitrate, the products are aqueous copper(II) nitrate and solid silver. Cu(s) + 2 Ag. NO 3(aq) → Cu(NO 3)2(aq) + 2 Ag(s)

PRACTICE PROBLEM 7 � In a blast furnace, the reaction between solid iron(III) oxide and carbon monoxide gas produces solid iron and carbon dioxide gas. � Fe 2 O 3(s) + 3 CO(g) → 2 Fe(s) + 3 CO 2(g)

Types of Chemical Reactions SECTION 2

1. DECOMPOSITION REACTIONS � Decomposition reaction a single compound has a reaction that makes two or more simpler substances � General � Most equation AX A + X happen when energy (light/heat) is added

2. SYNTHESIS REACTIONS � Synthesis (composition) reaction two or more substances combine to form a new compound � General equation A + X AX

3. SINGLE-REPLACEMENT REACTIONS � Single-replacement reaction one element replaces a similar element in a compound � General equation A + BX AX + B Y + BX BY + X

4. DOUBLE REPLACEMENT REACTIONS � Double-replacement reaction ions of two compounds exchange places in an aqueous solution to form two new compounds � General equation AX + BY AY + BX

5. COMBUSTION REACTIONS � Combustion reaction substance combines with oxygen, releasing large amount of energy as heat and light � 2 H 2(g) + O 2(g) → 2 H 2 O(g)

REACTIONS OF ELEMENTS WITH OXYGEN AND SULFUR � One simple type of synthesis reaction is combination of element with oxygen to form an oxide of the element � Almost all metals react with oxygen to form oxides � Ex. Magnesium burned magnesium oxide

2 Mg(s) + O 2(g) 2 Mg. O(s)

GROUP 2 ELEMENTS � Group 2 elements react to form oxides with general formula MO � M represents metal � Group 1 metals form oxides with general formula M 2 O � Li + O 2 Li 2 O

REACTIONS WITH SULFUR � Groups 1 and 2 react with sulfur to make sulfides of the element � Group 1 M 2 S � Group 2 MS � 16 Rb(s) + S 8(s) → 8 Rb 2 S(s) � 8 Ba(s) + S 8(s) → 8 Ba. S(s)
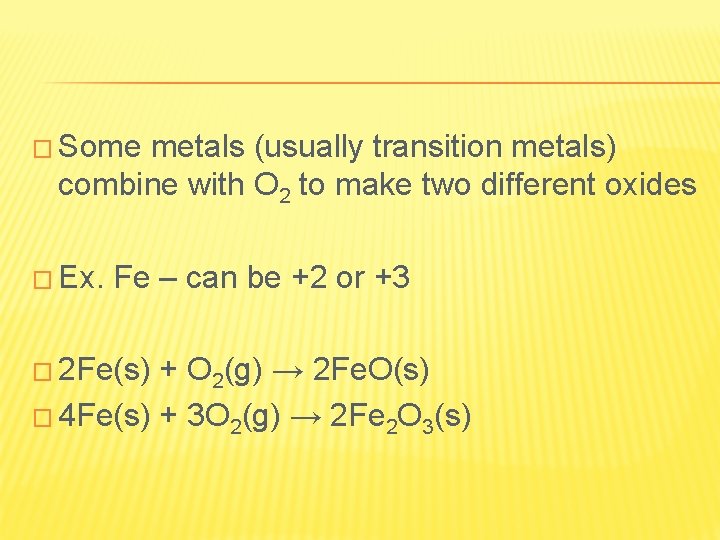
� Some metals (usually transition metals) combine with O 2 to make two different oxides � Ex. Fe – can be +2 or +3 � 2 Fe(s) + O 2(g) → 2 Fe. O(s) � 4 Fe(s) + 3 O 2(g) → 2 Fe 2 O 3(s)

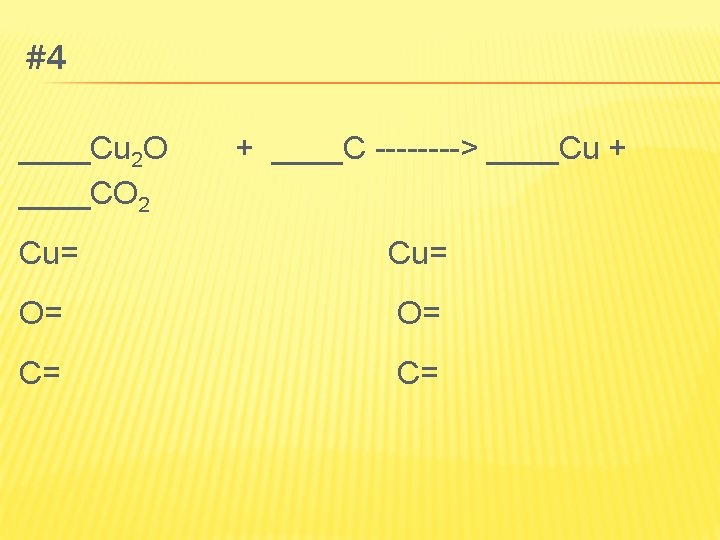
NONMETALS � Nonmetals also react with oxygen to make oxides � Sulfur reacts with oxygen to make sulfur dioxide � When carbon is burned, it makes carbon dioxide � S 8(s) + 8 O 2(g) → 8 SO 2(g) � C(s) + O 2(g) → CO 2(g)

� Hydrogen reacts with oxygen to make dihydrogen monoxide � 2 H 2(g) + O 2(g) → 2 H 2 O(g)

� Most metals react with halogens to make either ionic or covalent compounds � Ex. Group 1 reacts with halogens to form ionic compounds with formula MX � M = metal, X = halogen � 2 Na(s) + Cl 2(g) → 2 Na. Cl(s) � 2 K(s) + I 2(g) → 2 KI(s) REACTIONS OF METALS & HALOGENS
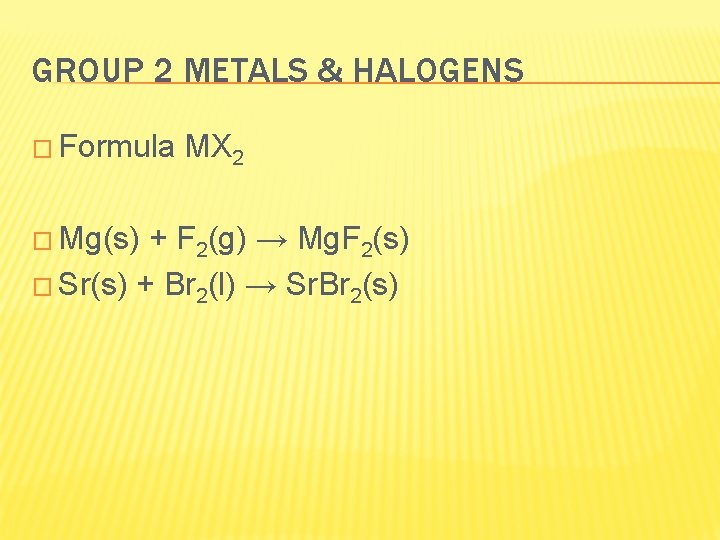
GROUP 2 METALS & HALOGENS � Formula � Mg(s) MX 2 + F 2(g) → Mg. F 2(s) � Sr(s) + Br 2(l) → Sr. Br 2(s)

� Active metals highly reactive metals � Oxides of active metals react with water to make metal hydroxides � Ca. O(s) � Ca. O + H 2 O(l) → Ca(OH)2(s) = lime � Ca(OH)2 is important in setting cement SYNTHESIS REACTIONS WITH OXIDES

OXYACIDS � Many oxides of nonmetals react with water to make oxyacids � SO 2(g) + H 2 O(l) → H 2 SO 3(aq) � In air polluted with SO 2, reacts with oxygen in air to form sulfuric acid (acid rain) � 2 H 2 SO 3(aq) + O 2(g) → 2 H 2 SO 4(aq)

PRACTICE PROBLEMS � Identify the products in each of the following reactions: � a. hydrogen burned in oxygen � H 2 O � b. H 2(g) + N 2(g) � NH 3 � c. Ca. O(s) + H 2 O(l) � Ca(OH)2(aq)

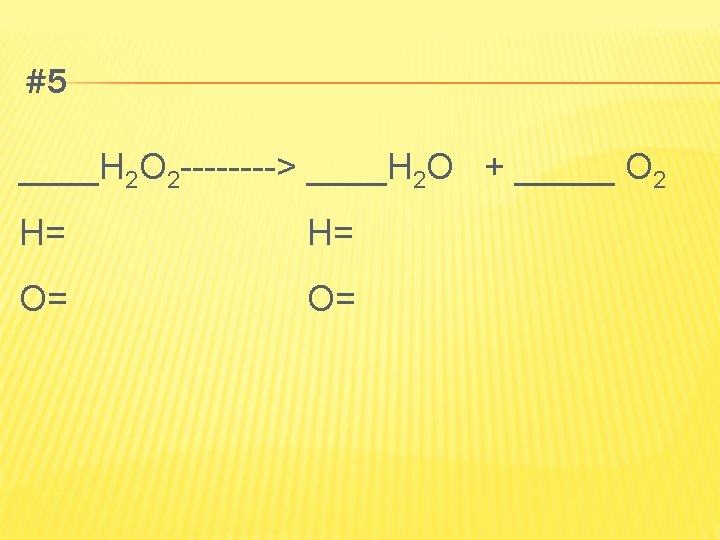
DECOMPOSITION OF BINARY COMPOUNDS � Simplest kind of decomposition reaction is binary compound into its elements � Ex. Passing electricity through water � 2 H 2 O(l) electricity 2 H 2(g) + O 2(g)

� Electrolysis decomposition of a substance by electricity

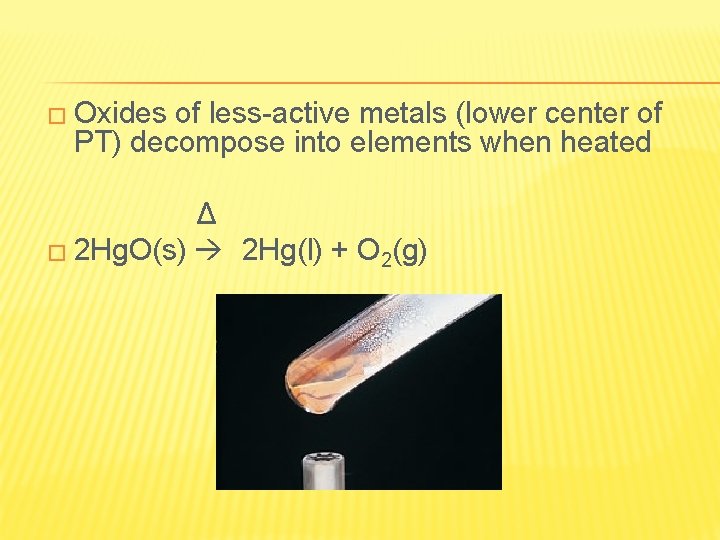
� Oxides of less-active metals (lower center of PT) decompose into elements when heated Δ � 2 Hg. O(s) 2 Hg(l) + O 2(g)

DECOMPOSITION OF METAL CARBONATES � When heated, metal carbonates break down to make a metal oxide and CO 2 Δ � Ca. CO 3(s) Ca. O(s) + CO 2(g)

DECOMPOSITION OF METAL HYDROXIDES � All except with Group 1 metals decompose when heated to make metal oxides and water Δ � Ca(OH)2(s) Ca. O(s) + H 2 O(g)

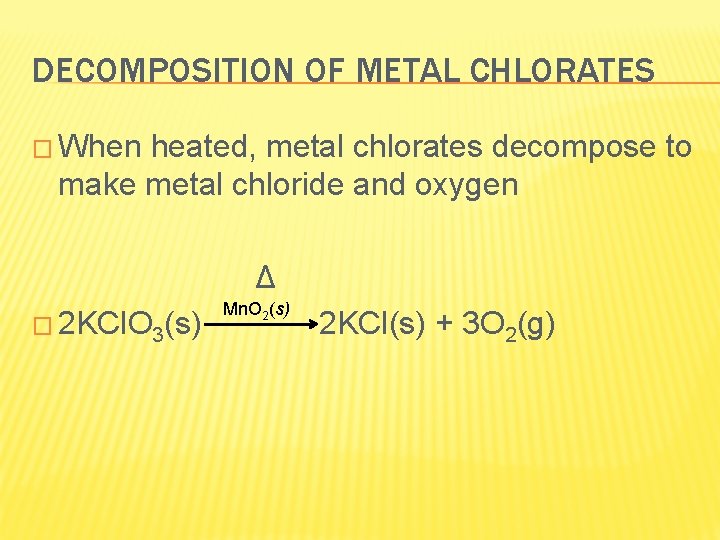
DECOMPOSITION OF METAL CHLORATES � When heated, metal chlorates decompose to make metal chloride and oxygen Δ � 2 KCl. O 3(s) Mn. O 2(s) 2 KCl(s) + 3 O 2(g)

DECOMPOSITION OF ACIDS � Certain acids decompose into nonmetal oxides and water � H 2 CO 3(aq) → CO 2(g) + H 2 O(l) Δ � H 2 SO 4(aq) SO 3(g) + H 2 O(l)

PRACTICE PROBLEMS � Predict the products for these decomposition reactions � a. sodium chlorate Sodium chloride + oxygen � b. calcium carbonate Calcium oxide + carbon dioxide � c. potassium bromide Potassium + bromine

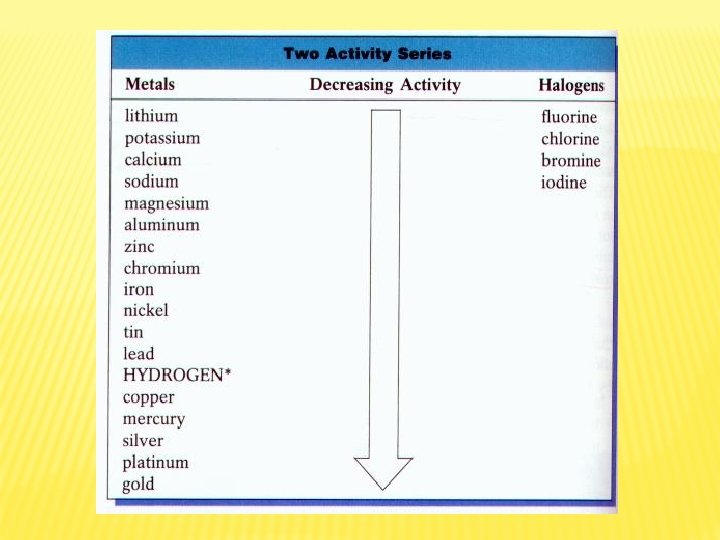
REPLACEMENT OF A METAL IN A COMPOUND BY ANOTHER METAL � Aluminum is more active than lead � When solid aluminum is placed in aqueous lead(II) nitrate, the aluminum replaces the lead 2 Al(s) + 3 Pb(NO 3)2(aq) → 3 Pb(s) + 2 Al(NO 3)3(aq) � Based on activity series of metals


REPLACEMENT OF HYDROGEN IN WATER BY METAL � Most-active metals (Group 1) react strongly with water to make metal hydroxides and hydrogen � 2 Na(s) + 2 H 2 O(l) → 2 Na. OH(aq) + H 2(g) � Less-active metals react with steam or other form of energy

REPLACEMENT OF HYDROGEN IN ACID BY METAL � More-active metals react with certain acidic solutions and replace hydrogen � Reaction products are metal compound (salt) and hydrogen gas � Mg(s) + 2 HCl(aq) → H 2(g) + Mg. Cl 2(aq)

REPLACEMENT OF HALOGENS � One halogen replaces another � Fluorine is most reactive � Can replace any other halogen � Cl 2(g) + 2 KBr(aq) → 2 KCl(aq) + Br 2(l) � F 2(g) + 2 Na. Cl(aq) → 2 Na. F(aq) + Cl 2(g) � Br 2(l) + KCl(aq) → no reaction

PRACTICE PROBLEMS � For the following equations, predict what the products will be: � a. Ag + KNO 3 → No reaction � b. Zn + Ag. NO 3 → Zn(NO 3)2 + Ag � c. Cl 2 + KI → I 2 + 2 KCl

� d. Cu + Fe. SO 4 → No reaction � e. Fe + Pb(NO 3)2 → Pb + Fe(NO 3)2 � f. Cu + Al 2(SO 4)3 → No reaction � g. Al + Pb(NO 3)2 → Pb + Al(NO 3)3 � h. Cl 2 + Na. I → I 2 + Na. Cl

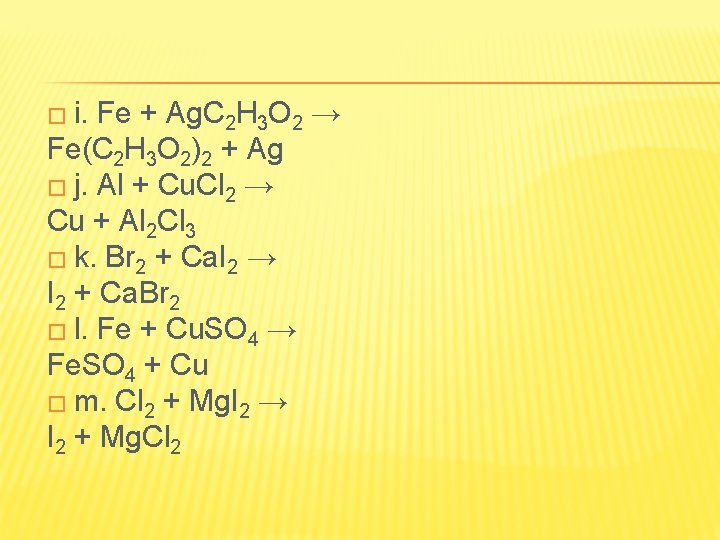
� i. Fe + Ag. C 2 H 3 O 2 → Fe(C 2 H 3 O 2)2 + Ag � j. Al + Cu. Cl 2 → Cu + Al 2 Cl 3 � k. Br 2 + Ca. I 2 → I 2 + Ca. Br 2 � l. Fe + Cu. SO 4 → Fe. SO 4 + Cu � m. Cl 2 + Mg. I 2 → I 2 + Mg. Cl 2

FORMATION OF A PRECIPITATE � Occurs when cations of one reactant combine with anions of another to form insoluble (or slightly soluble) compound � 2 KI(aq) + Pb(NO 3)2(aq) → Pb. I 2(s) + 2 KNO 3(aq)

FORMATION OF A GAS � In some D-R reactions, one product is insoluble gas that bubbles out of mixture � Fe. S(s) + 2 HCl(aq) → H 2 S(g) + Fe. Cl 2(aq)

FORMATION OF WATER � In some D-R reactions, water is one of the products � HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O(l)

PRACTICE PROBLEMS � Classify each of the following reactions as synthesis, decomposition, singlereplacement, double-replacement, or combustion: � a. N 2(g) + 3 H 2(g) → 2 NH 3(g) � synthesis � b. 2 Li(s) + 2 H 2 O(l ) → 2 Li. OH(aq) + H 2(g) � single-replacement � c. 2 Na. NO 3(s) → 2 Na. NO 2(s) + O 2(g) � decomposition

� d. 2 C 6 H 14(l ) + 19 O 2(g) → 12 CO 2(g) + 14 H 2 O(l ) � combustion � e. NH 4 Cl(s) → NH 3(g) + HCl(g) � decomposition � f. Ba. O(s) + H 2 O(l ) → Ba(OH)2(aq) � synthesis � g. Ag. NO 3(aq) + Na. Cl(aq) →Ag. Cl(s) + Na. NO 3(aq) � double-replacement

PRACTICE PROBLEM � For each of the following reactions, identify the missing substances, then balance the final equation. Each slot may be one OR MORE substances. � a. synthesis: _____ → Li 2 O � 4 Li + O 2 2 Li 2 O � b. decomposition: Mg(Cl. O 3)2 → _____ � Mg(Cl. O 3)2 Mg. Cl 2 + 3 O 2

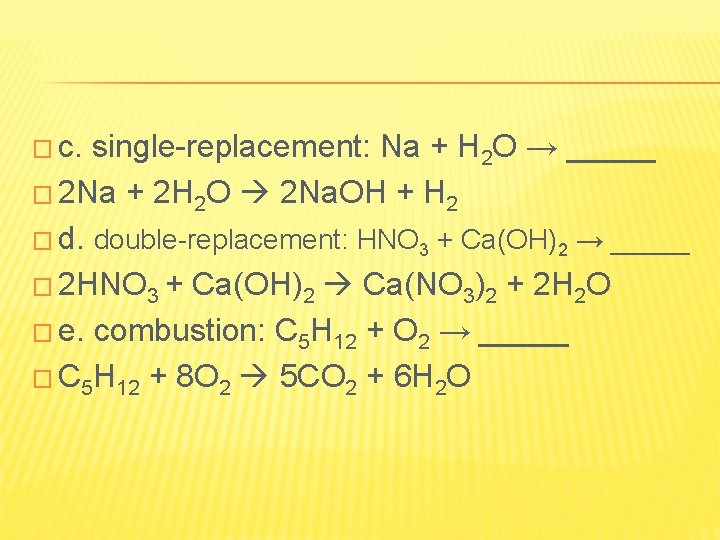
� c. single-replacement: Na + H 2 O → _____ � 2 Na + 2 H 2 O 2 Na. OH + H 2 � d. double-replacement: HNO 3 + Ca(OH)2 → _____ � 2 HNO 3 + Ca(OH)2 Ca(NO 3)2 + 2 H 2 O � e. combustion: C 5 H 12 + O 2 → _____ � C 5 H 12 + 8 O 2 5 CO 2 + 6 H 2 O