Molecular Geometry and Bonding Theories AP Chemistry Ch

Molecular Geometry and Bonding Theories AP Chemistry – Ch 9 Mr. Christopherson
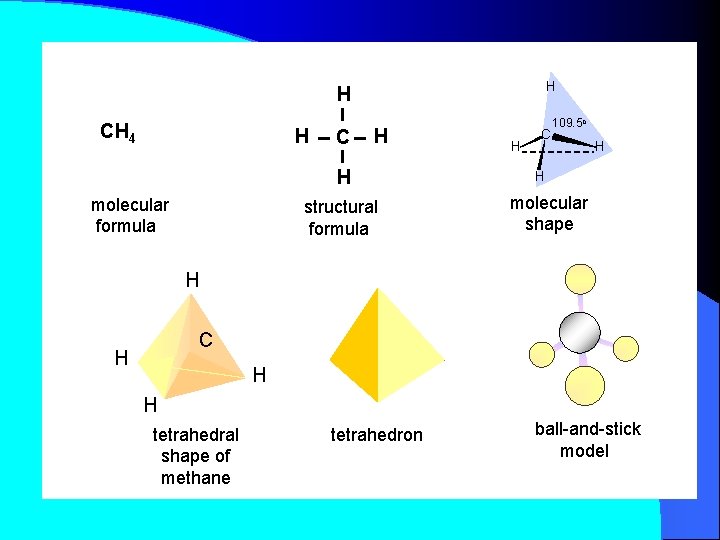
H H CH 4 H C H H molecular formula structural formula H C 109. 5 o H H molecular shape H C H H H tetrahedral shape of methane tetrahedron ball-and-stick model
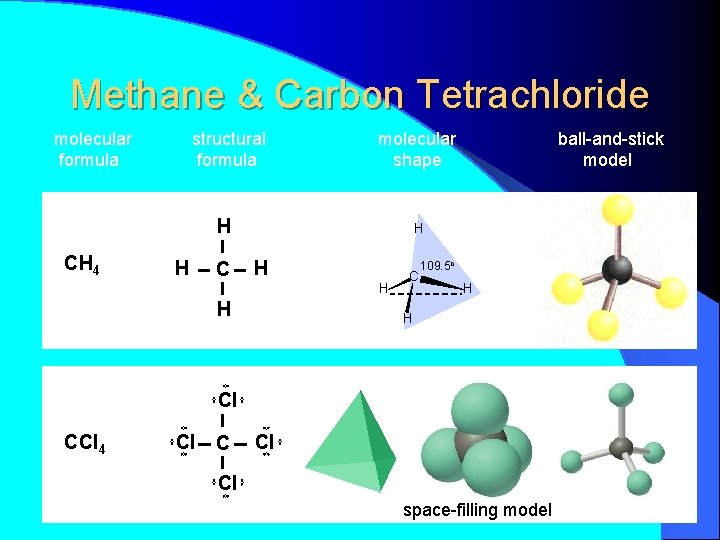
Methane & Carbon Tetrachloride molecular formula structural formula molecular shape H CH 4 H C ball-and-stick model H H C 109. 5 o H H Cl CCl 4 Cl Cl space-filling model
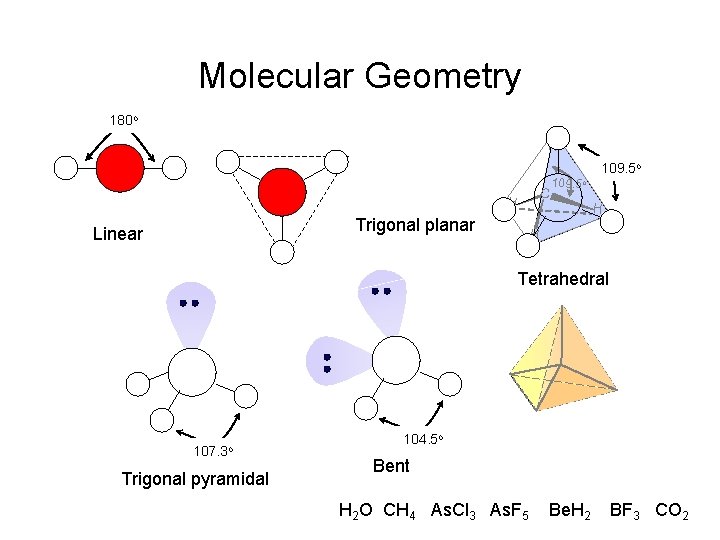
Molecular Geometry 180 o H H Trigonal planar Linear C 109. 5 o H H Tetrahedral 107. 3 o Trigonal pyramidal 104. 5 o Bent H 2 O CH 4 As. Cl 3 As. F 5 Be. H 2 BF 3 CO 2
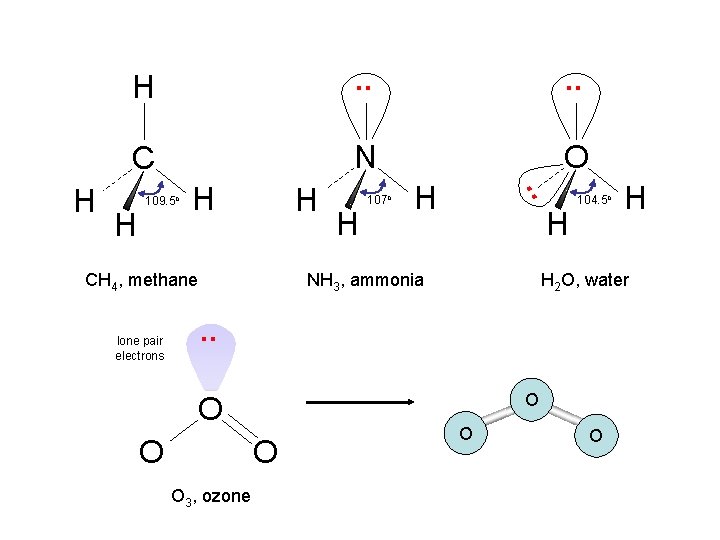
. . C N O H 109. 5 o H H CH 4, methane lone pair electrons H 107 o H . . H H H NH 3, ammonia 104. 5 o H 2 O, water . . O O O 3, ozone H O O
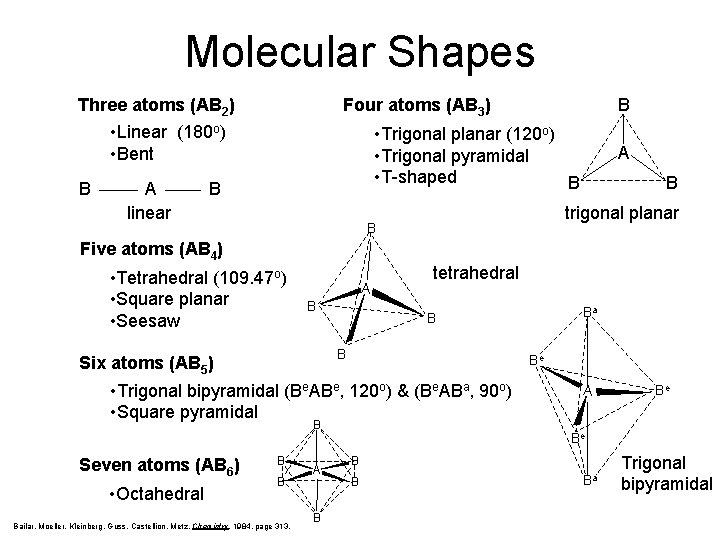
Molecular Shapes Three atoms (AB 2) Four atoms (AB 3) • Linear (180 o) • Bent B A linear B • Trigonal planar (120 o) • Trigonal pyramidal • T-shaped B B A B trigonal planar B Five atoms (AB 4) • Tetrahedral (109. 47 o) • Square planar • Seesaw A B tetrahedral B Six atoms (AB 5) Be • Trigonal bipyramidal (Be. ABe, 120 o) & (Be. ABa, 90 o) • Square pyramidal B Seven atoms (AB 6) • Octahedral B B Bailar, Moeller, Kleinberg, Guss, Castellion, Metz, Chemistry, 1984, page 313. A B Ba B A Be Be B B Ba Trigonal bipyramidal

Bonding and Shape of Molecules Number of Unshared Pairs Covalent Structure Shape Examples -Be- Linear Be. Cl 2 Trigonal planar BF 3 2 0 3 0 4 0 C Tetrahedral CH 4, Si. Cl 4 3 1 : Number of Bonds Pyramidal NH 3, PCl 3 2 2 Bent H 2 O, H 2 S, SCl 2 B : N O:
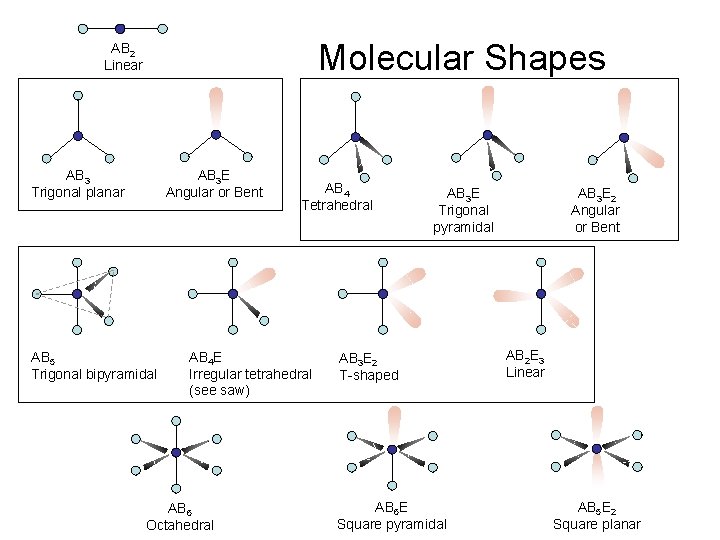
Molecular Shapes AB 2 Linear AB 3 Trigonal planar AB 3 E Angular or Bent AB 5 Trigonal bipyramidal AB 4 Tetrahedral AB 4 E Irregular tetrahedral (see saw) AB 6 Octahedral AB 3 E Trigonal pyramidal AB 3 E 2 T-shaped AB 6 E Square pyramidal AB 3 E 2 Angular or Bent AB 2 E 3 Linear AB 5 E 2 Square planar
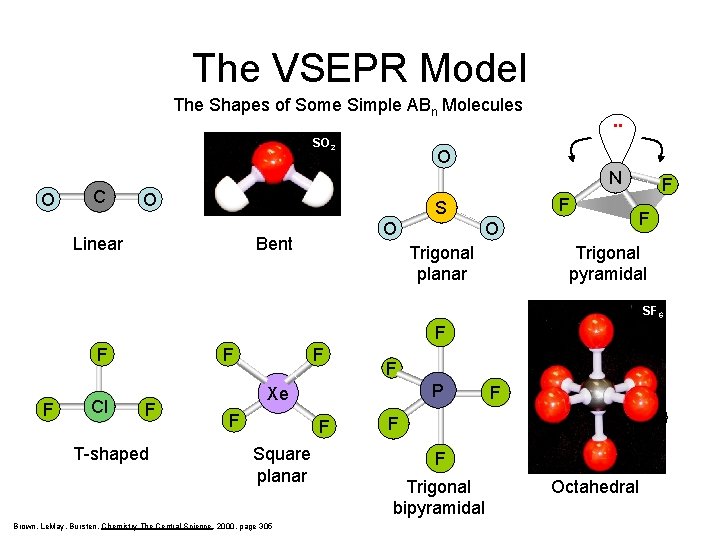
The VSEPR Model The Shapes of Some Simple ABn Molecules . . O C . . SO 2 . . O N S O O Linear O Bent F S O F F O Trigonal planar Trigonal pyramidal SF 6 F F F Cl F F T-shaped F F F Square planar Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 305 F F P Xe F F F S F F F Trigonal bipyramidal Octahedral
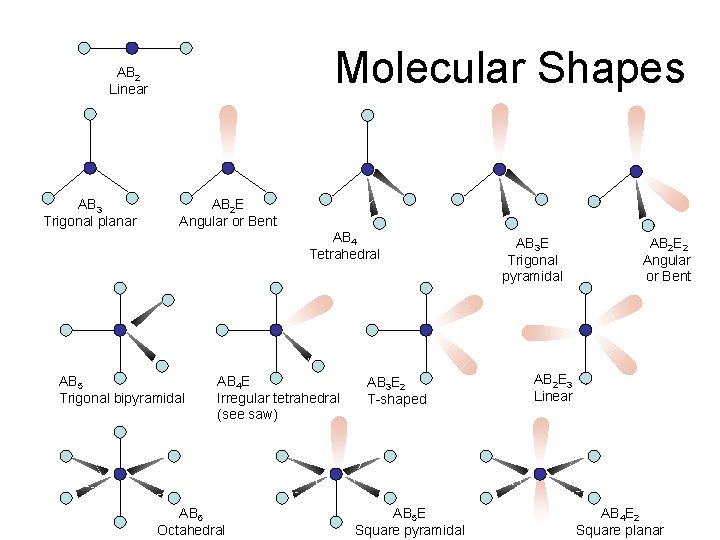
Molecular Shapes AB 2 Linear AB 3 Trigonal planar AB 2 E Angular or Bent AB 4 Tetrahedral AB 5 Trigonal bipyramidal AB 4 E Irregular tetrahedral (see saw) AB 6 Octahedral AB 3 E 2 T-shaped AB 5 E Square pyramidal AB 3 E Trigonal pyramidal AB 2 E 2 Angular or Bent AB 2 E 3 Linear AB 4 E 2 Square planar
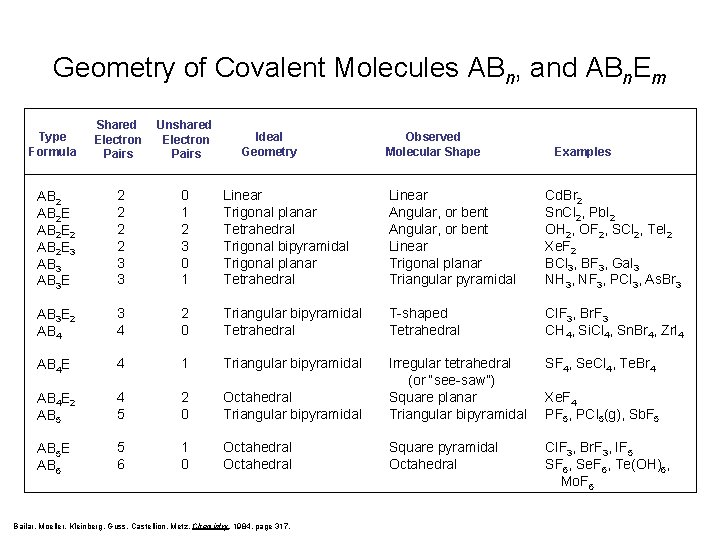
Geometry of Covalent Molecules ABn, and ABn. Em Type Formula Shared Electron Pairs Unshared Electron Pairs AB 2 E 2 AB 2 E 3 AB 3 E 2 2 3 3 0 1 2 3 0 1 Linear Trigonal planar Tetrahedral Trigonal bipyramidal Trigonal planar Tetrahedral Linear Angular, or bent Linear Trigonal planar Triangular pyramidal Cd. Br 2 Sn. Cl 2, Pb. I 2 OH 2, OF 2, SCl 2, Te. I 2 Xe. F 2 BCl 3, BF 3, Ga. I 3 NH 3, NF 3, PCl 3, As. Br 3 AB 3 E 2 AB 4 3 4 2 0 Triangular bipyramidal Tetrahedral T-shaped Tetrahedral Cl. F 3, Br. F 3 CH 4, Si. Cl 4, Sn. Br 4, Zr. I 4 AB 4 E 4 1 Triangular bipyramidal SF 4, Se. Cl 4, Te. Br 4 AB 4 E 2 AB 5 4 5 2 0 Octahedral Triangular bipyramidal Irregular tetrahedral (or “see-saw”) Square planar Triangular bipyramidal AB 5 E AB 6 5 6 1 0 Octahedral Square pyramidal Octahedral Cl. F 3, Br. F 3, IF 5 SF 6, Se. F 6, Te(OH)6, Mo. F 6 Ideal Geometry Bailar, Moeller, Kleinberg, Guss, Castellion, Metz, Chemistry, 1984, page 317. Observed Molecular Shape Examples Xe. F 4 PF 5, PCl 5(g), Sb. F 5
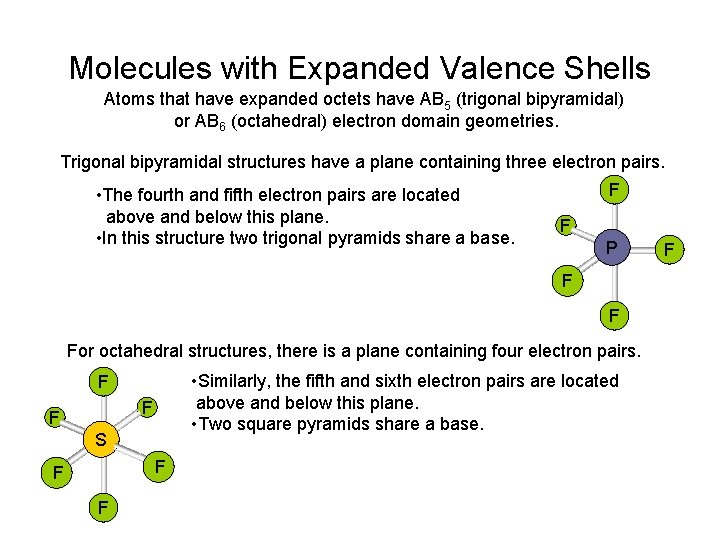
Molecules with Expanded Valence Shells Atoms that have expanded octets have AB 5 (trigonal bipyramidal) or AB 6 (octahedral) electron domain geometries. Trigonal bipyramidal structures have a plane containing three electron pairs. • The fourth and fifth electron pairs are located above and below this plane. • In this structure two trigonal pyramids share a base. F F P F F For octahedral structures, there is a plane containing four electron pairs. • Similarly, the fifth and sixth electron pairs are located above and below this plane. • Two square pyramids share a base. F F F S F F

Trigonal Bipyramid F F P F F • The three electron pairs in the plane are called equatorial. F • The two electron pairs above and below this plane are called axial. • The axial electron pairs are 180 o apart and 90 o from to the equatorial electrons. • The equatorial electron pairs are 120 o apart. • To minimize electron-electron repulsions, nonbonding pairs are always placed in equatorial positions, and bonding pairs in either axial or equatorial positions.

F Octahedron F F S • The four electron pairs in the plane are 90 o to each other. F F F • The remaining two electron pairs are 180 o apart and 90 o from the electrons in the plane. • Because of the symmetry of the system, each position is equivalent. • The equatorial electron pairs are 120 o apart. • If we have five bonding pairs and one nonbonding pair, it doesn’t matter where the nonbonding pair is placed. v The molecular geometry is square pyramidal. • If two nonbonding pairs are present, the repulsions are minimized by pointing them toward opposite sides of the octahedron. F F v. The molecular geometry is square planar. Xe F F
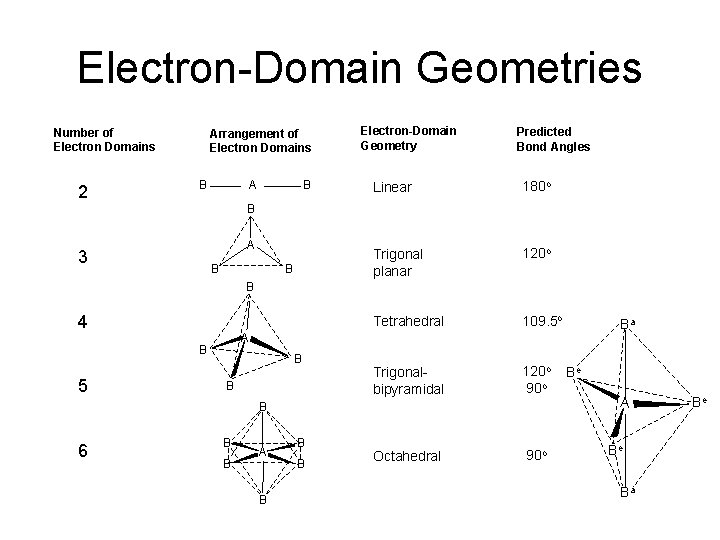
Electron-Domain Geometries Number of Electron Domains 2 Arrangement of Electron Domains B A B Electron-Domain Geometry Predicted Bond Angles Linear 180 o Trigonal planar 120 o Tetrahedral 109. 5 o Trigonalbipyramidal 120 o 90 o Octahedral 90 o B A 3 B B B 4 A B 5 B B B 6 Ba B B A B Be A Be Ba Be
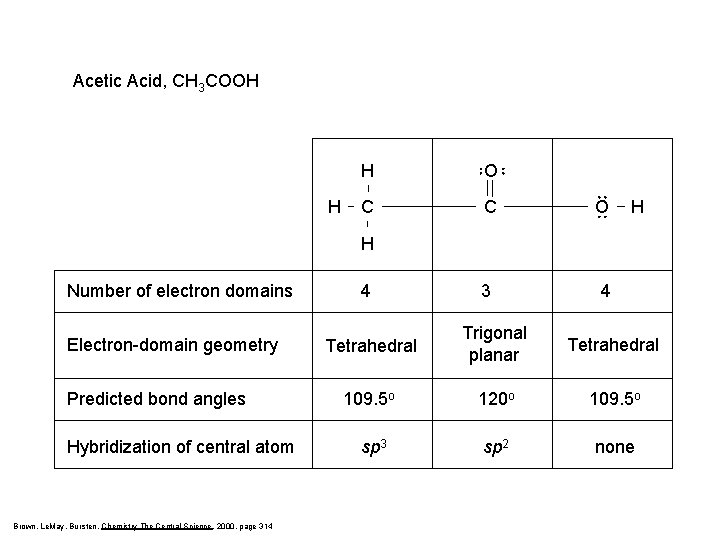
Acetic Acid, CH 3 COOH H H O C C O 3 4 H H Number of electron domains Electron-domain geometry Predicted bond angles Hybridization of central atom Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 314 4 Tetrahedral Trigonal planar Tetrahedral 109. 5 o 120 o 109. 5 o sp 3 sp 2 none
Molecular Polarity Molecular Structure Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
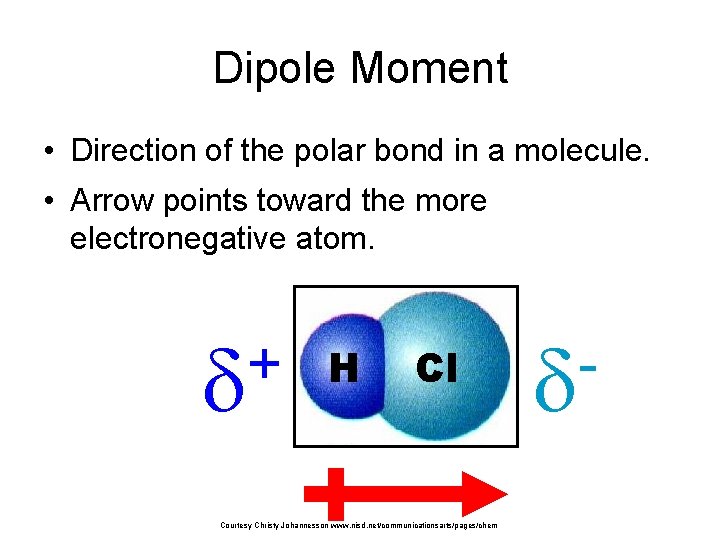
Dipole Moment • Direction of the polar bond in a molecule. • Arrow points toward the more electronegative atom. + H Cl Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Determining Molecular Polarity • Depends on: – dipole moments – molecular shape Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
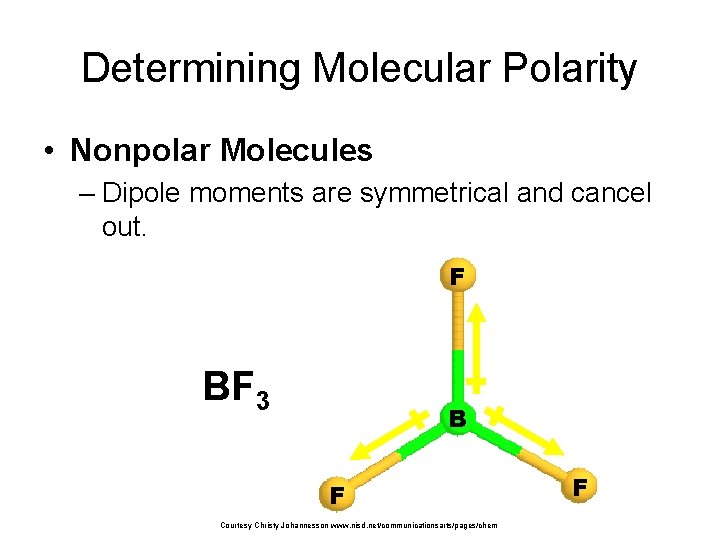
Determining Molecular Polarity • Nonpolar Molecules – Dipole moments are symmetrical and cancel out. F BF 3 B F Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem F
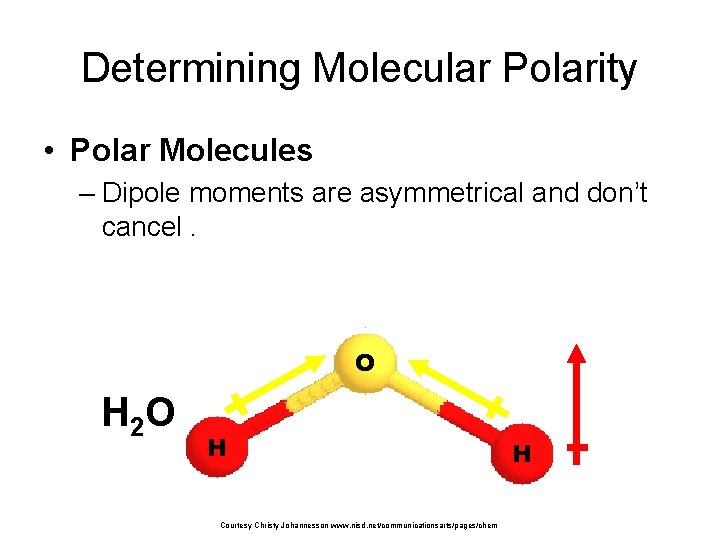
Determining Molecular Polarity • Polar Molecules – Dipole moments are asymmetrical and don’t cancel. O H 2 O H Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem H net dipole moment
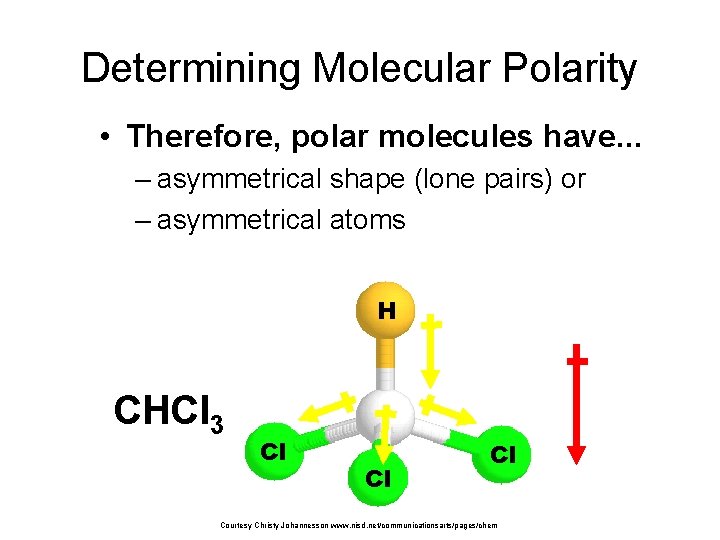
Determining Molecular Polarity • Therefore, polar molecules have. . . – asymmetrical shape (lone pairs) or – asymmetrical atoms H CHCl 3 Cl Cl Cl Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem net dipole moment
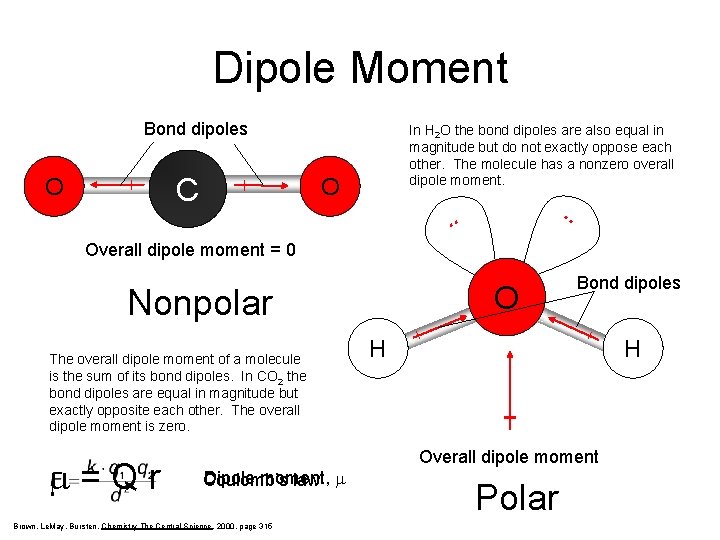
Dipole Moment Bond dipoles C O In H 2 O the bond dipoles are also equal in magnitude but do not exactly oppose each other. The molecule has a nonzero overall dipole moment. O . . Overall dipole moment = 0 O Nonpolar The overall dipole moment of a molecule is the sum of its bond dipoles. In CO 2 the bond dipoles are equal in magnitude but exactly opposite each other. The overall dipole moment is zero. m=Qr Dipole moment, Coulomb’s law m Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 315 Bond dipoles H H Overall dipole moment Polar
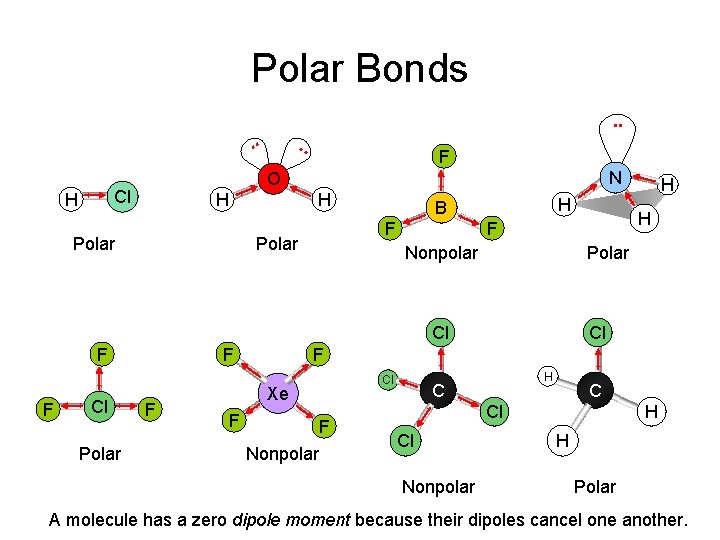
Polar Bonds. . F N O Cl H . . H H Polar F Polar H B Polar Cl F F Cl Polar F F Cl Xe F H F Nonpolar H C C Cl Cl Nonpolar H H Polar A molecule has a zero dipole moment because their dipoles cancel one another.
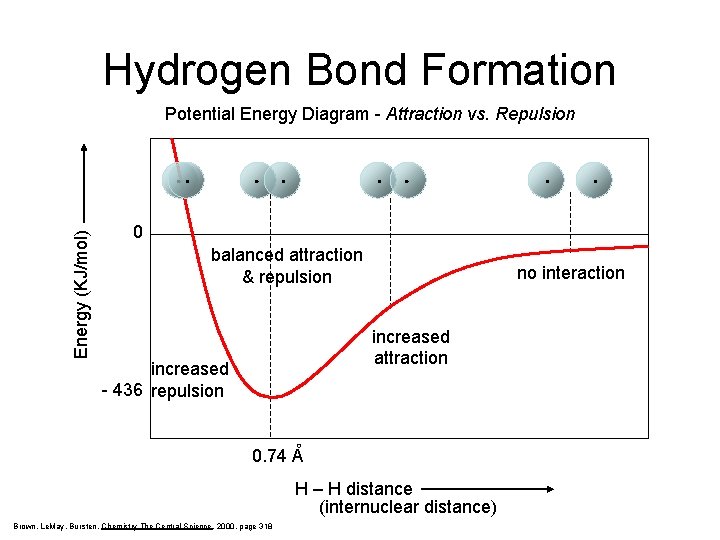
Hydrogen Bond Formation Energy (KJ/mol) Potential Energy Diagram - Attraction vs. Repulsion 0 balanced attraction & repulsion no interaction increased attraction increased - 436 repulsion 0. 74 A H – H distance (internuclear distance) Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 318
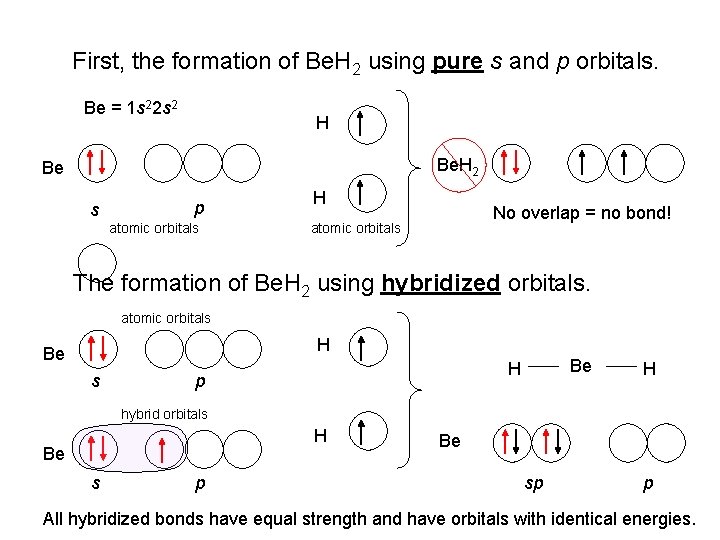
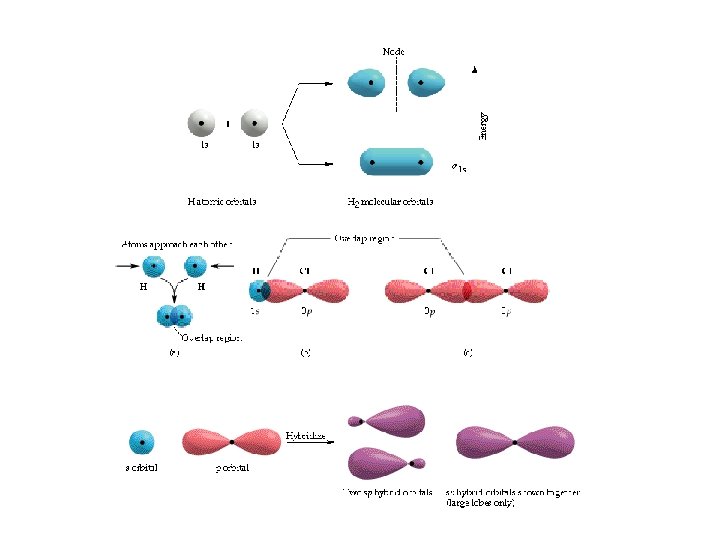
First, the formation of Be. H 2 using pure s and p orbitals. Be = 1 s 22 s 2 H Be. H 2 Be s p atomic orbitals H No overlap = no bond! atomic orbitals The formation of Be. H 2 using hybridized orbitals. atomic orbitals H Be s Be H p H hybrid orbitals H Be s p Be. H 2 Be sp p All hybridized bonds have equal strength and have orbitals with identical energies.
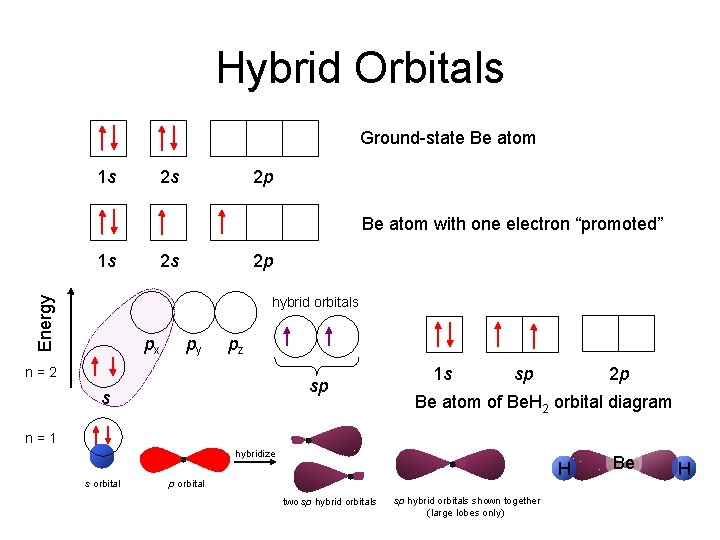
Hybrid Orbitals Ground-state Be atom 1 s 2 s 2 p Be atom with one electron “promoted” 1 s 2 s 2 p Energy hybrid orbitals px py pz n=2 sp s 1 s sp 2 p Be atom of Be. H 2 orbital diagram n=1 hybridize s orbital H p orbital two sp hybrid orbitals shown together (large lobes only) Be H
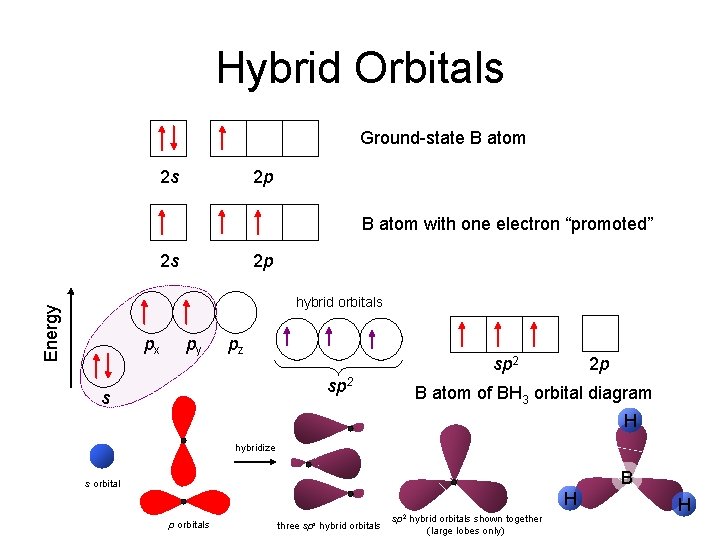
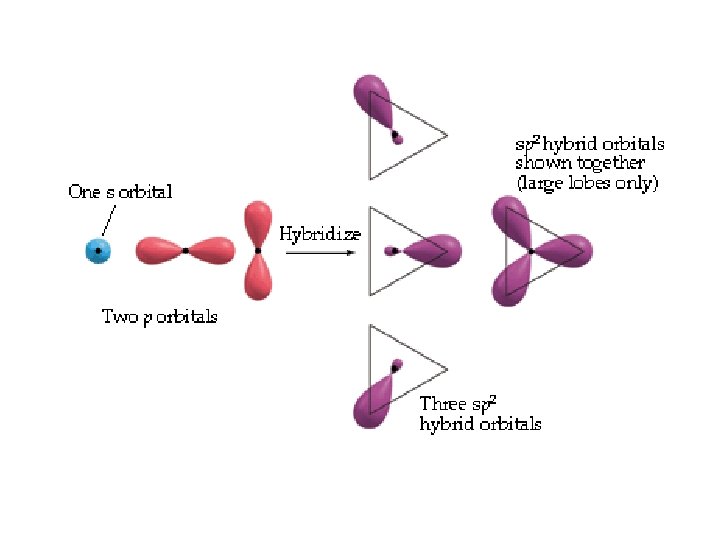
Hybrid Orbitals Ground-state B atom 2 s 2 p B atom with one electron “promoted” 2 s 2 p Energy hybrid orbitals px py pz sp 2 s 2 p B atom of BH 3 orbital diagram H hybridize B s orbital H p orbitals three sps hybrid orbitals sp 2 hybrid orbitals shown together (large lobes only) H
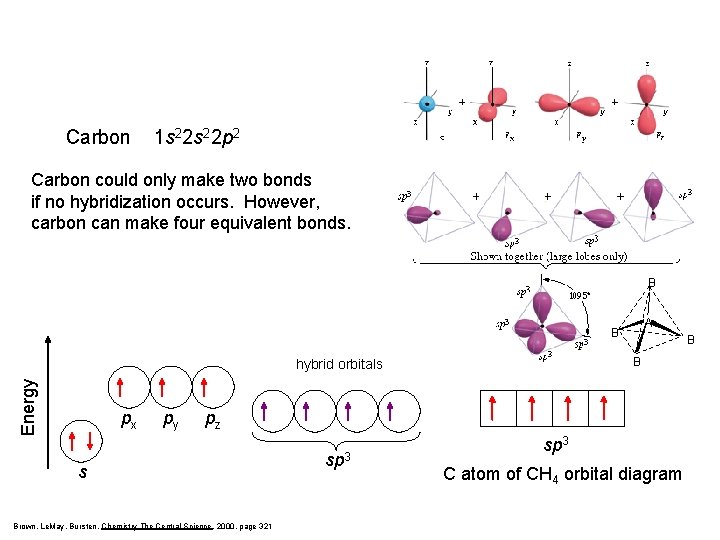
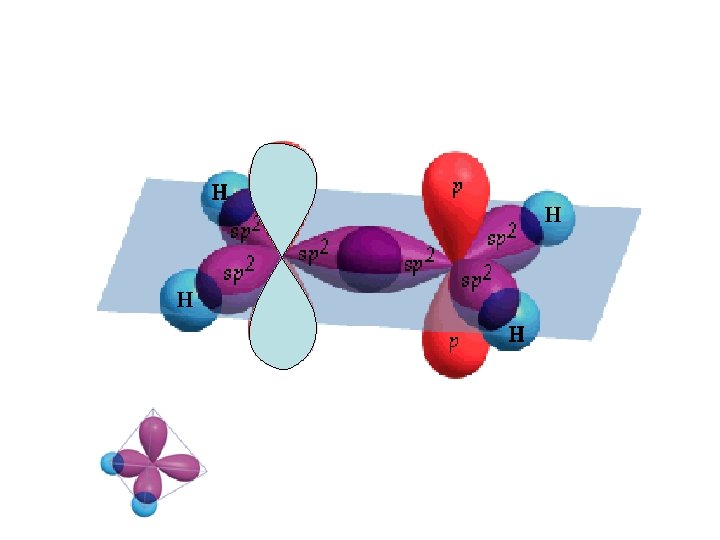
Carbon 1 s 22 p 2 Carbon could only make two bonds if no hybridization occurs. However, carbon can make four equivalent bonds. B A B B Energy hybrid orbitals px py B pz s Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 321 sp 3 C atom of CH 4 orbital diagram
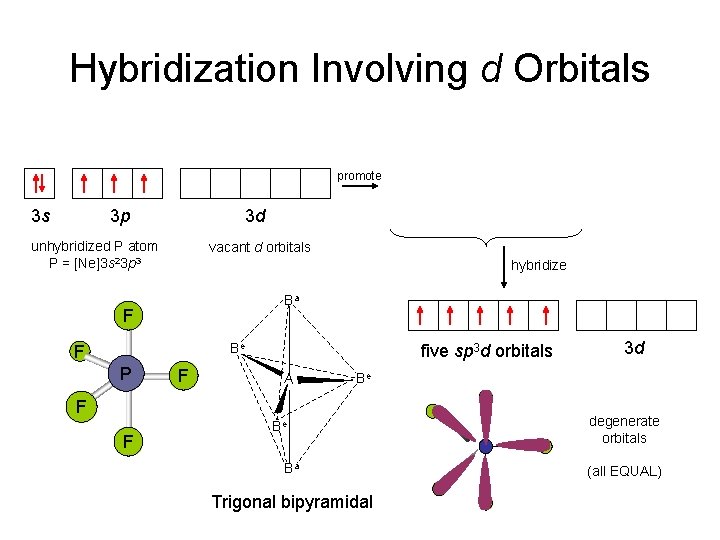
Hybridization Involving d Orbitals promote 3 s 3 p 3 d unhybridized P atom P = [Ne]3 s 23 p 3 3 s hybridize Ba Be F F five sp 3 d orbitals A 3 d Be F F 3 d vacant d orbitals F P 3 p Be Ba Trigonal bipyramidal degenerate orbitals (all EQUAL)
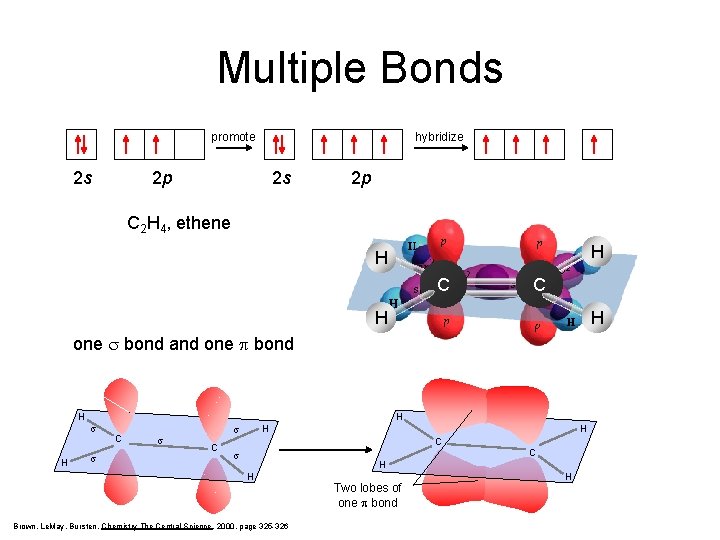
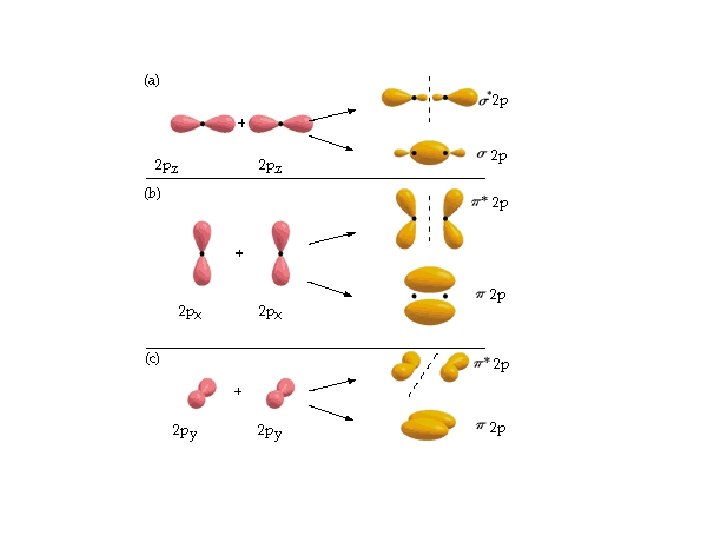
Multiple Bonds hybridize promote 2 s 2 p sp 2 2 p C 2 H 4, ethene H H C C H H one s bond and one p bond H H s C s H s C H C s C H H Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 325 -326 Two lobes of one p bond H
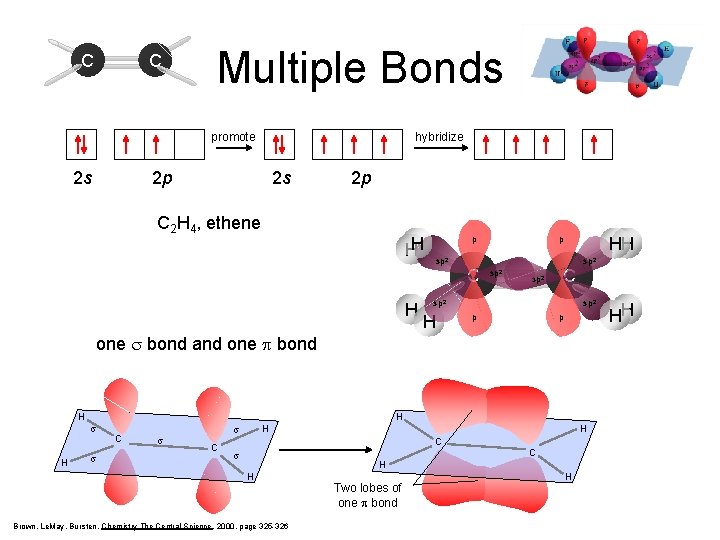
C C Multiple Bonds hybridize promote 2 s 2 p sp 2 C 2 H 4, ethene HH H p sp 2 C 2 p HH p sp 2 C sp 2 H sp 2 p p one s bond and one p bond H H s C s H s C H C s C H H Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 325 -326 Two lobes of one p bond H HH
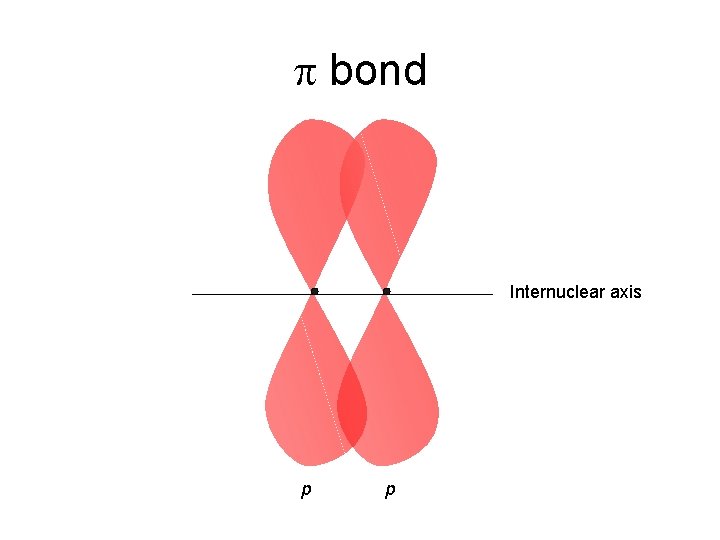
p bond Internuclear axis p p
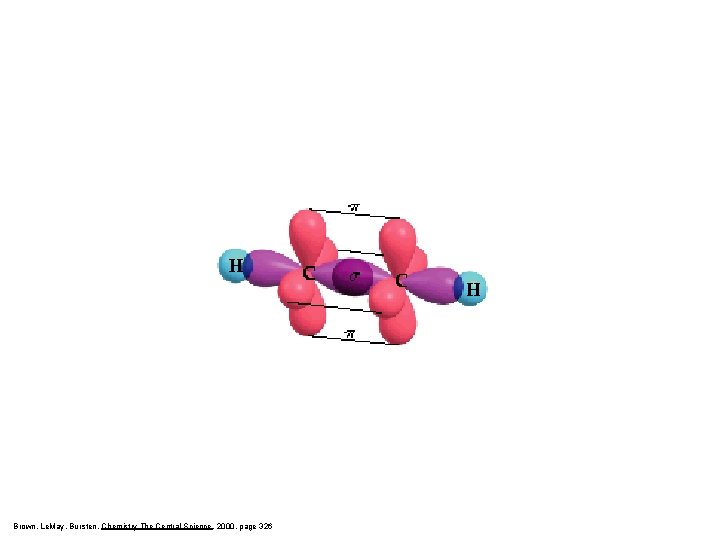
Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 326
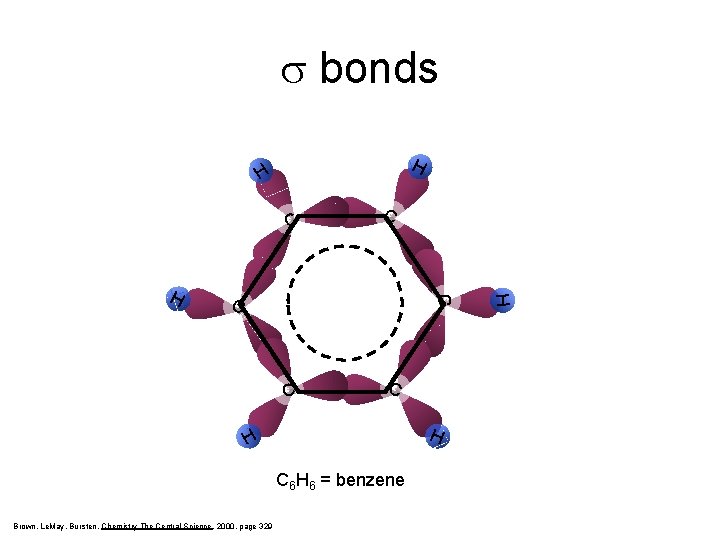
s bonds H H C C C H H C 6 H 6 = benzene Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 329 H C C H C
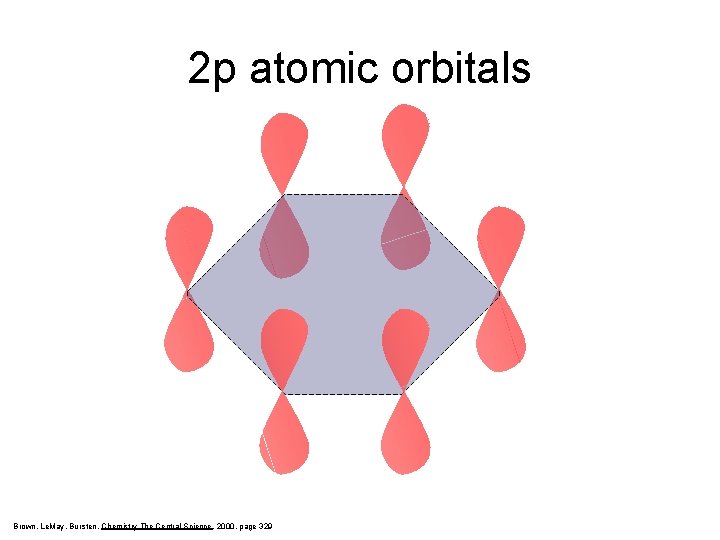
2 p atomic orbitals Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 329
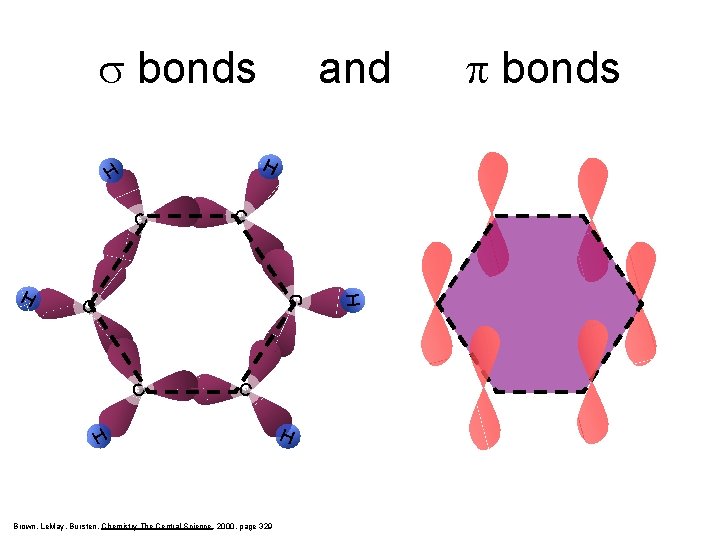
s bonds H H C C H Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 329 H H C C H and p bonds
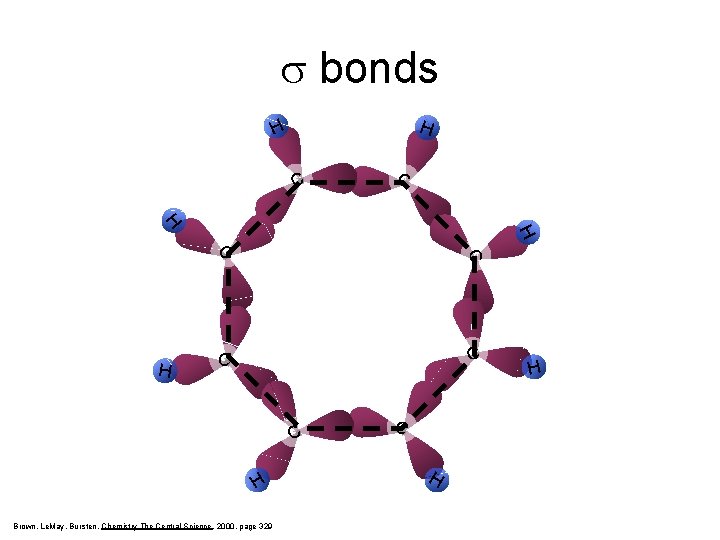
s bonds H H C C C H Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 329 C H H

s bonds H H C C C H Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 329 C H H
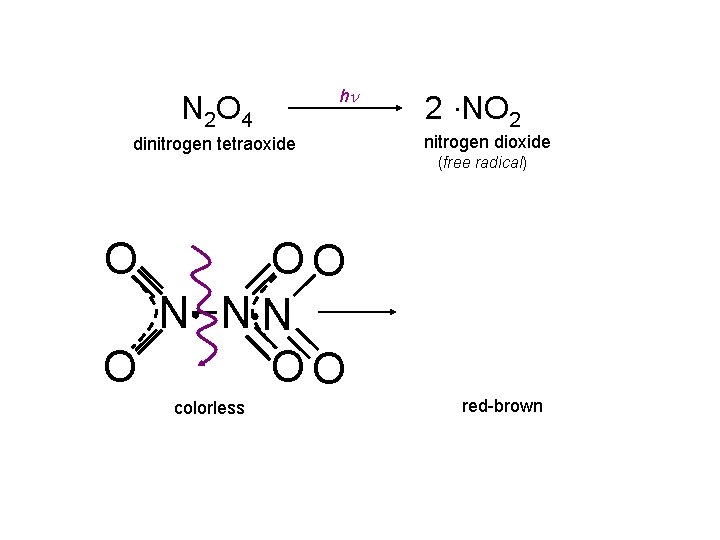
N 2 O 4 hn dinitrogen tetraoxide 2 NO 2 nitrogen dioxide (free radical) O OO N NN O OO colorless red-brown
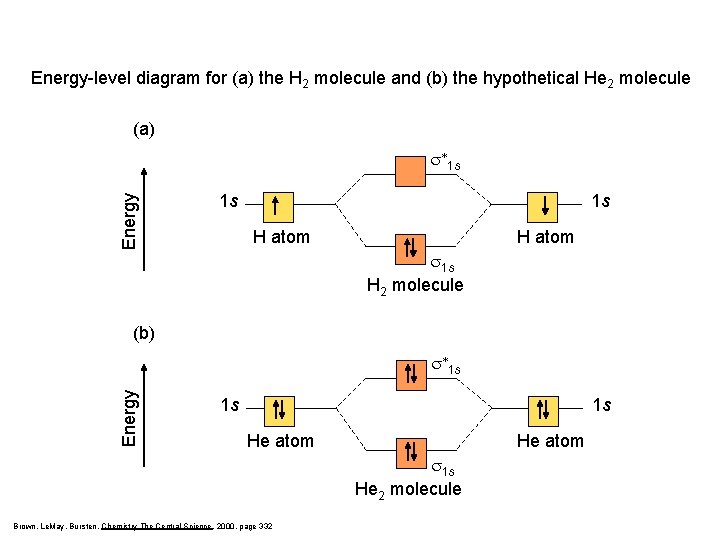
Energy-level diagram for (a) the H 2 molecule and (b) the hypothetical He 2 molecule (a) Energy s*1 s 1 s 1 s H atom s 1 s H 2 molecule (b) Energy s*1 s 1 s 1 s He atom s 1 s He 2 molecule Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 332

Bond Order Bond order = ½ (# or bonding electrons - # of antibonding electrons) • A bond order of 1 represents a single bond, • A bond order of 2 represents a double bond, • A bond order of 3 represents a triple bond. Because MO theory also treats molecules with an odd number of electrons, Bond orders of 1/2 , 3/2 , or 5/2 are possible. • A bond order of 0 means no bond exists.
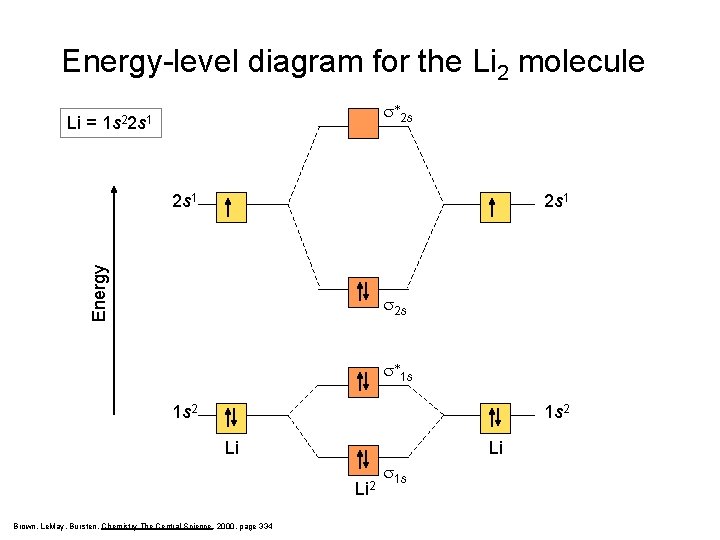
Energy-level diagram for the Li 2 molecule Li = s*2 s 1 s 22 s 1 Energy 2 s 1 s 2 s s*1 s 1 s 2 Li Li Li 2 Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 334 s 1 s
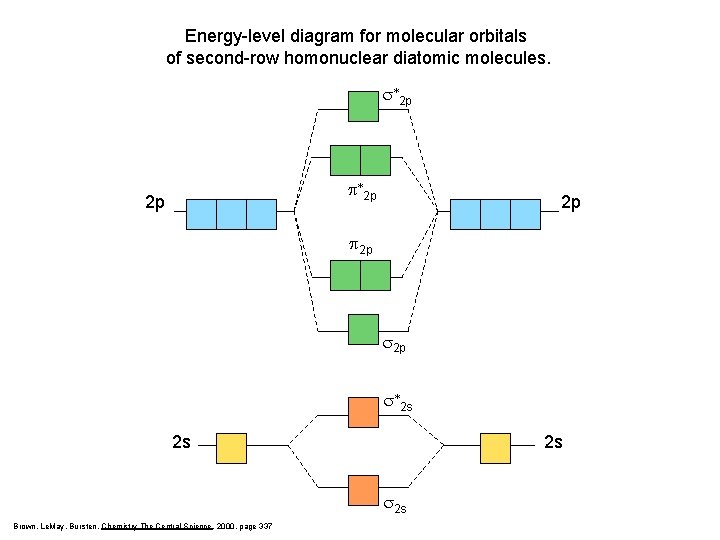
Energy-level diagram for molecular orbitals of second-row homonuclear diatomic molecules. s*2 p p*2 p 2 p 2 p s*2 s 2 s 2 s Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 337
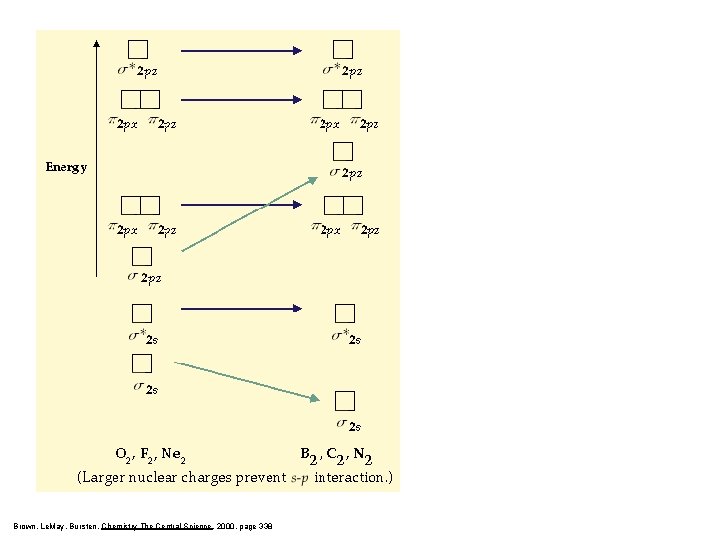
Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 338
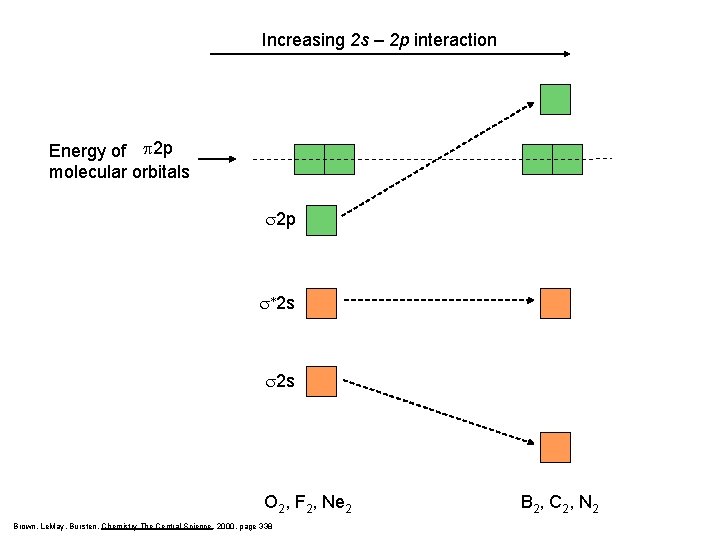
Increasing 2 s – 2 p interaction Energy of p 2 p molecular orbitals s 2 p s*2 s s 2 s O 2, F 2, Ne 2 Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 338 B 2, C 2, N 2
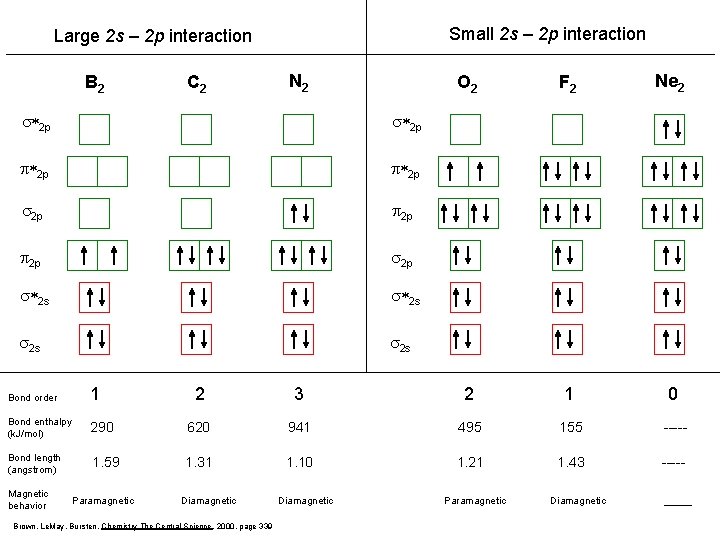
Small 2 s – 2 p interaction Large 2 s – 2 p interaction B 2 C 2 N 2 s*2 p p*2 p s 2 p p 2 p s*2 s s 2 s Bond order 1 Bond enthalpy (k. J/mol) Bond length (angstrom) Magnetic behavior O 2 F 2 Ne 2 2 3 2 1 0 290 620 941 495 155 ----- 1. 59 1. 31 1. 10 1. 21 1. 43 ----- Paramagnetic Diamagnetic Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 339 Diamagnetic Paramagnetic Diamagnetic _____

Magnetic Properties of a Sample PARAMAGNETISM – molecules with one or more unpaired electrons are attracted into a magnetic field. (appears to weigh MORE in a magnetic field) DIAMAGNETISM – substances with no unpaired electrons are weakly repelled from a magnetic field. (appears to weigh LESS in a magnetic field)
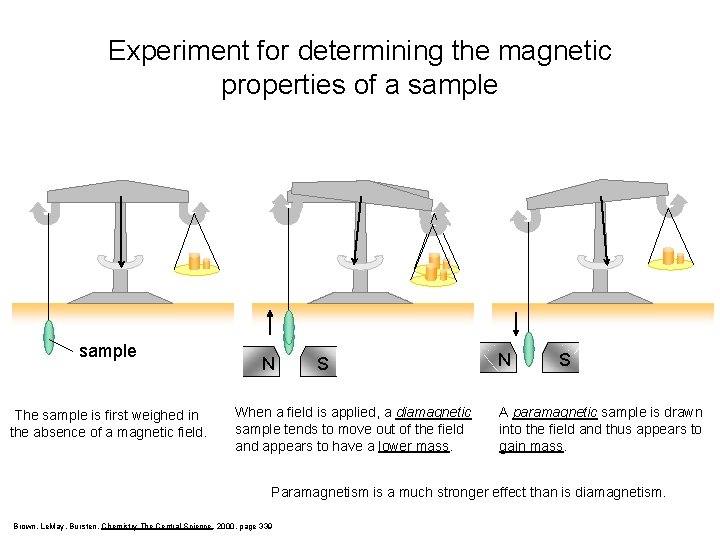
Experiment for determining the magnetic properties of a sample The sample is first weighed in the absence of a magnetic field. N S When a field is applied, a diamagnetic sample tends to move out of the field and appears to have a lower mass. N S A paramagnetic sample is drawn into the field and thus appears to gain mass. Paramagnetism is a much stronger effect than is diamagnetism. Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 339
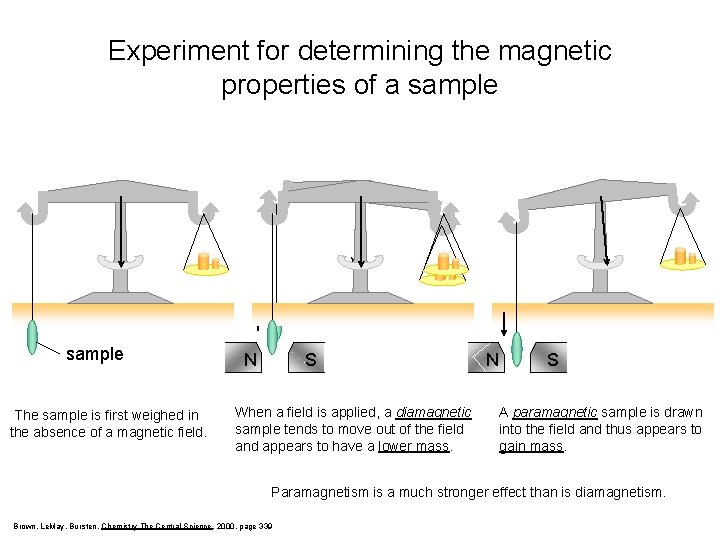
Experiment for determining the magnetic properties of a sample The sample is first weighed in the absence of a magnetic field. N S When a field is applied, a diamagnetic sample tends to move out of the field and appears to have a lower mass. N S A paramagnetic sample is drawn into the field and thus appears to gain mass. Paramagnetism is a much stronger effect than is diamagnetism. Brown, Le. May, Bursten, Chemistry The Central Science, 2000, page 339
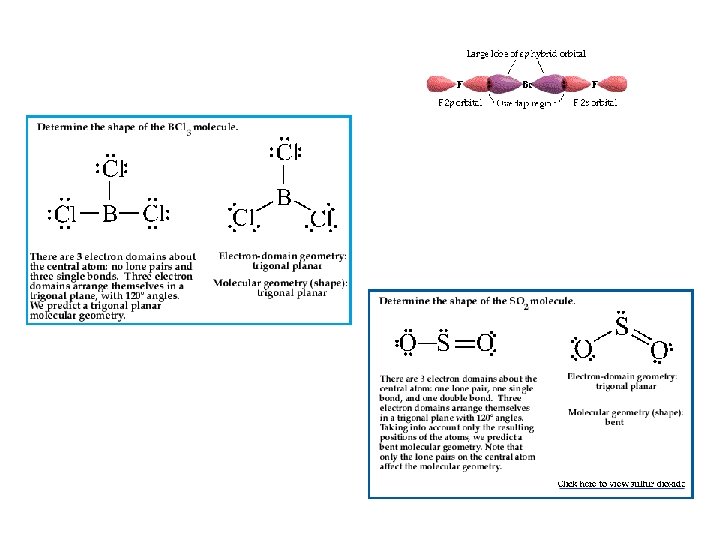
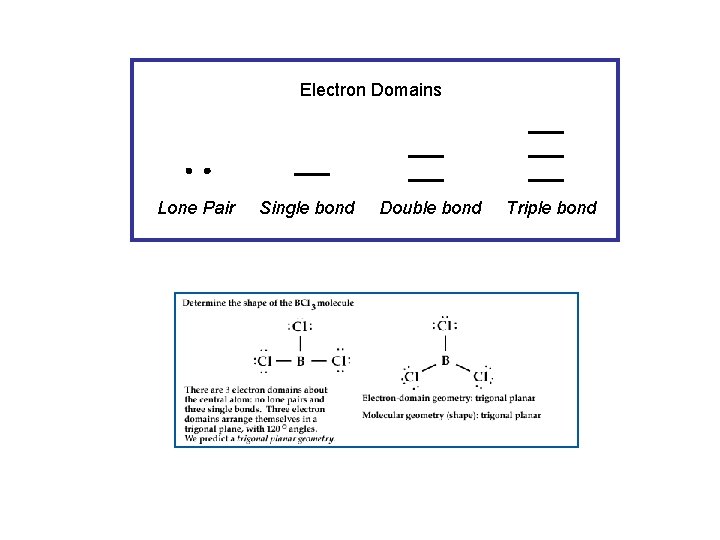
Electron Domains Lone Pair Single bond Double bond Triple bond
Molecular Geometry Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem I II III

VSEPR Theory l Valence Theory Shell Electron Pair Repulsion l Electron pairs orient themselves in order to minimize repulsive forces. Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
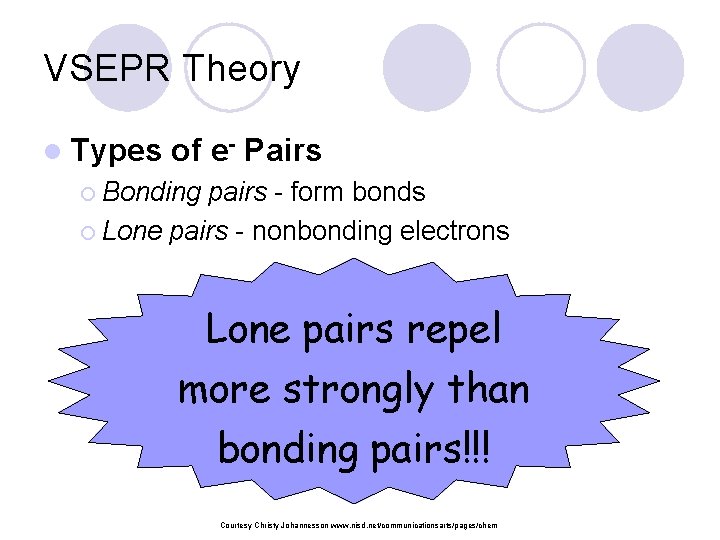
VSEPR Theory l Types of e- Pairs ¡ Bonding pairs - form bonds ¡ Lone pairs - nonbonding electrons Lone pairs repel more strongly than bonding pairs!!! Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
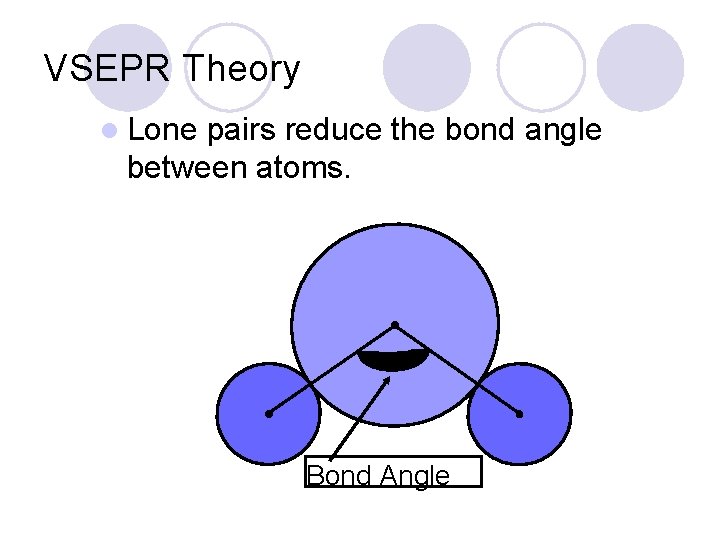
VSEPR Theory l Lone pairs reduce the bond angle between atoms. Bond Angle Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
Determining Molecular Shape l Draw the Lewis Diagram. l Tally up e- pairs on central atom. ¡ double/triple bonds = ONE pair l Shape is determined by the # of bonding pairs and lone pairs. Know the 8 common shapes & their bond angles! Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
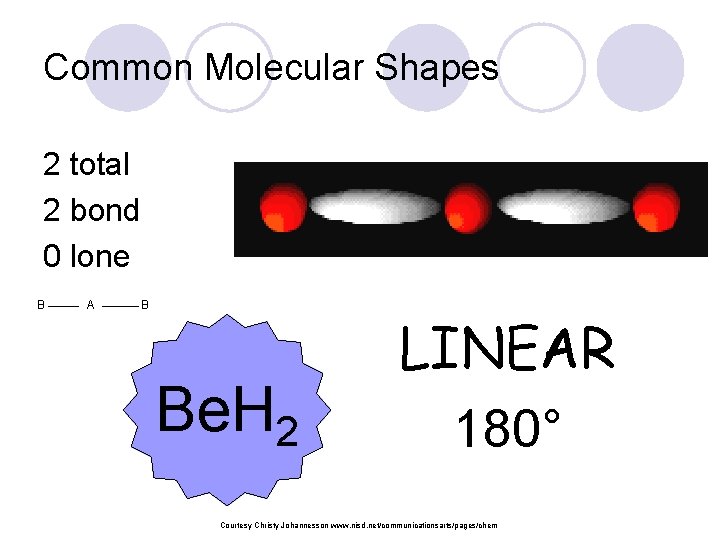
Common Molecular Shapes 2 total 2 bond 0 lone B A B Be. H 2 LINEAR 180° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
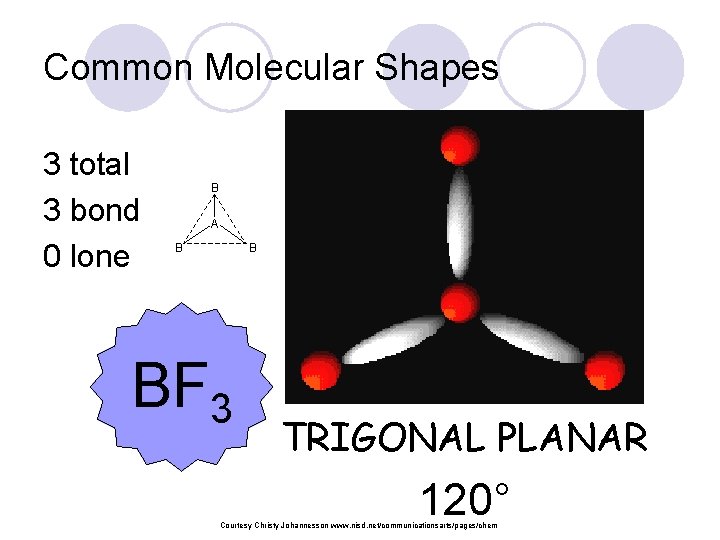
Common Molecular Shapes 3 total 3 bond 0 lone B A B B BF 3 TRIGONAL PLANAR 120° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
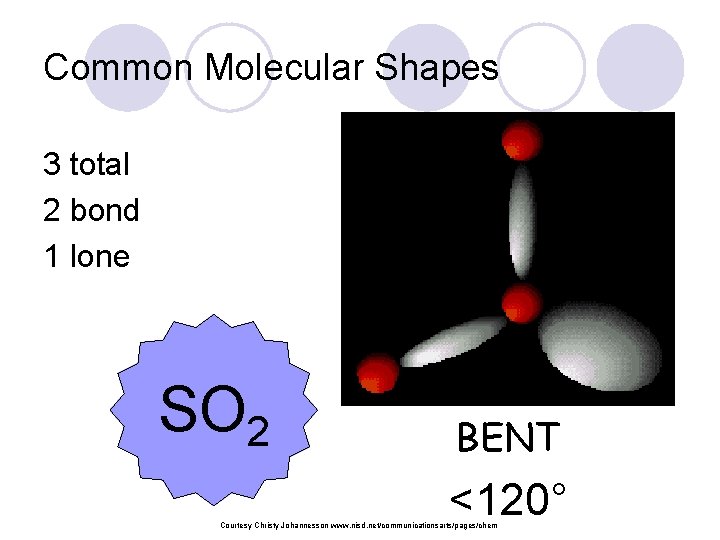
Common Molecular Shapes 3 total 2 bond 1 lone SO 2 BENT <120° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
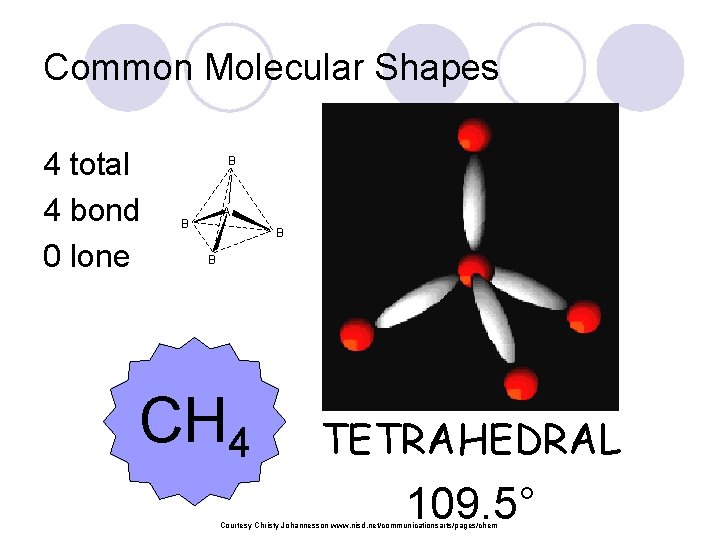
Common Molecular Shapes 4 total 4 bond 0 lone B A B B B CH 4 TETRAHEDRAL 109. 5° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
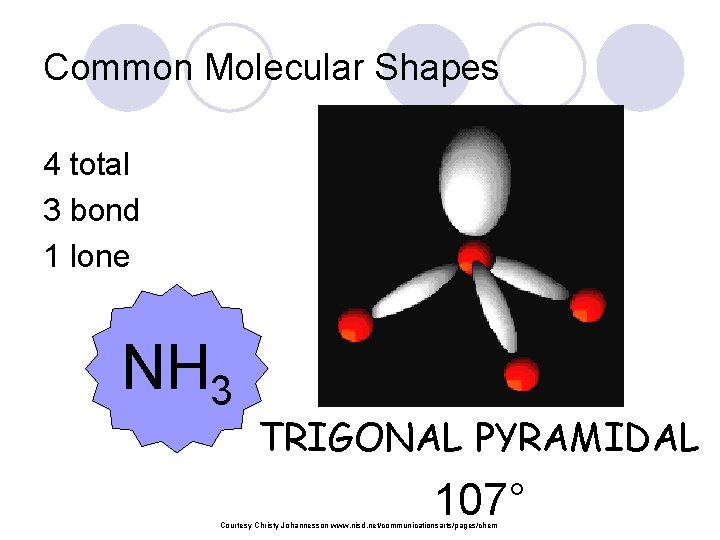
Common Molecular Shapes 4 total 3 bond 1 lone NH 3 TRIGONAL PYRAMIDAL 107° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
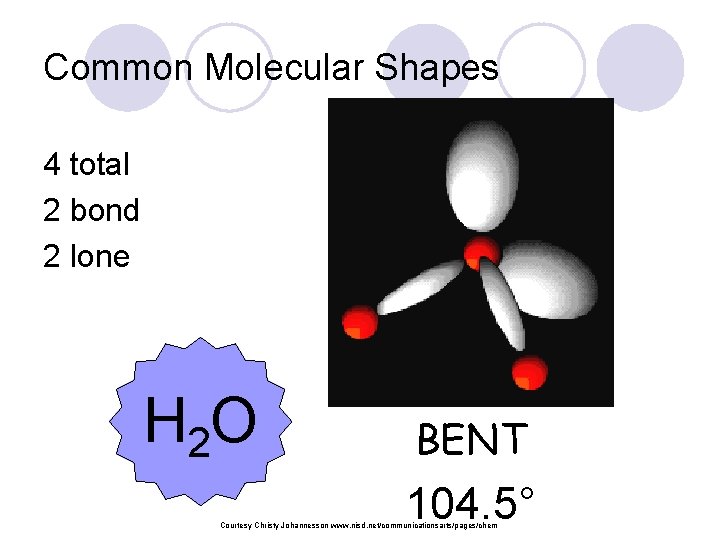
Common Molecular Shapes 4 total 2 bond 2 lone H 2 O BENT 104. 5° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
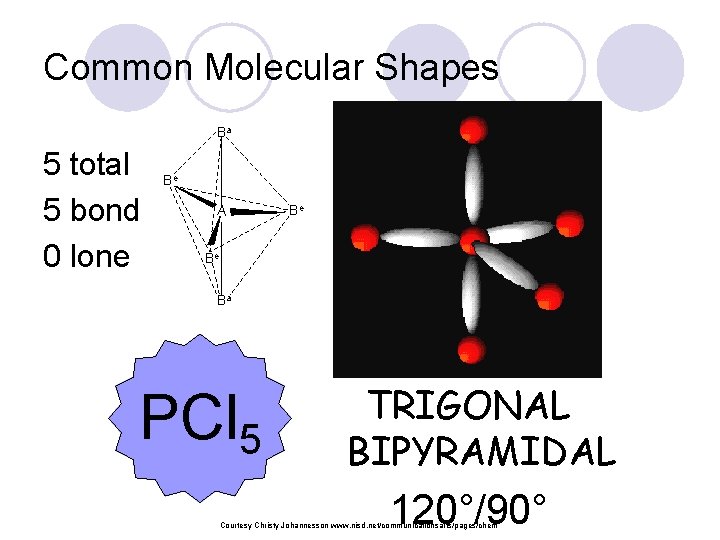
Common Molecular Shapes Ba 5 total 5 bond 0 lone Be A Be Be Ba PCl 5 TRIGONAL BIPYRAMIDAL 120°/90° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
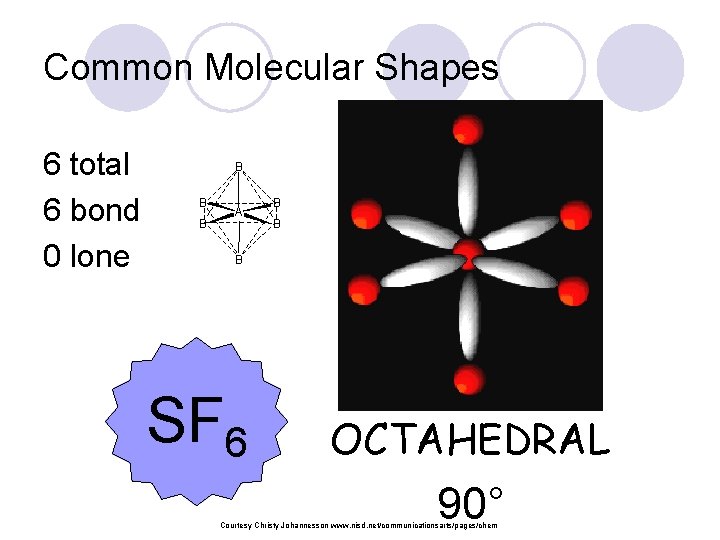
Common Molecular Shapes 6 total 6 bond 0 lone B B B A B B B SF 6 OCTAHEDRAL 90° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
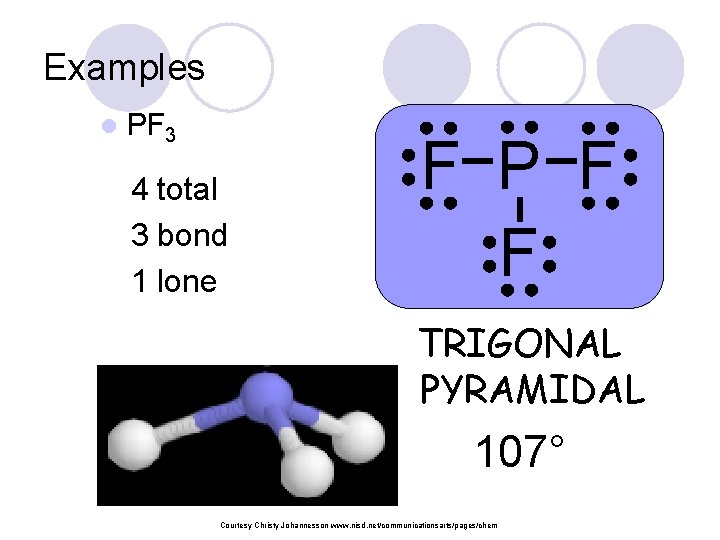
Examples l PF 3 4 total 3 bond 1 lone F P F F TRIGONAL PYRAMIDAL 107° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
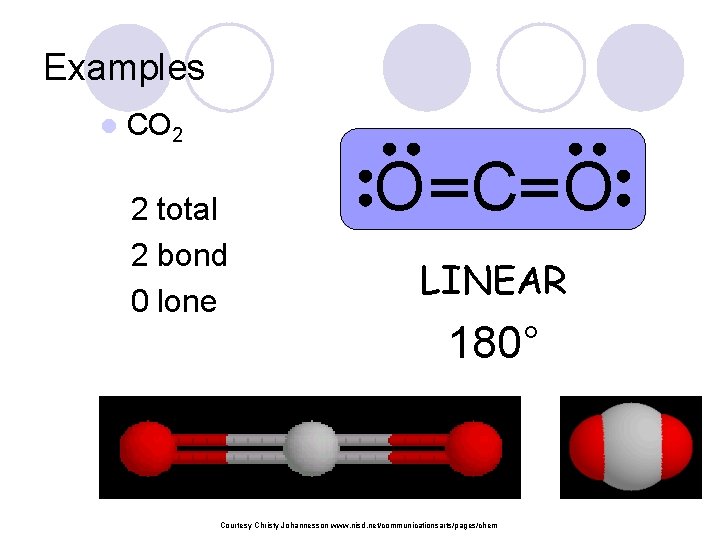
Examples l CO 2 2 total 2 bond 0 lone O C O LINEAR 180° Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
- Slides: 71