CHEMISTRY ATOMIC STRUCTURE TERMINOLOGY Chemistry study of composition
















- Slides: 27

CHEMISTRY ATOMIC STRUCTURE

TERMINOLOGY Chemistry study of composition of matter and processes that build up and break down substances. Biochemistry study of chemical processes that help to sustain living things

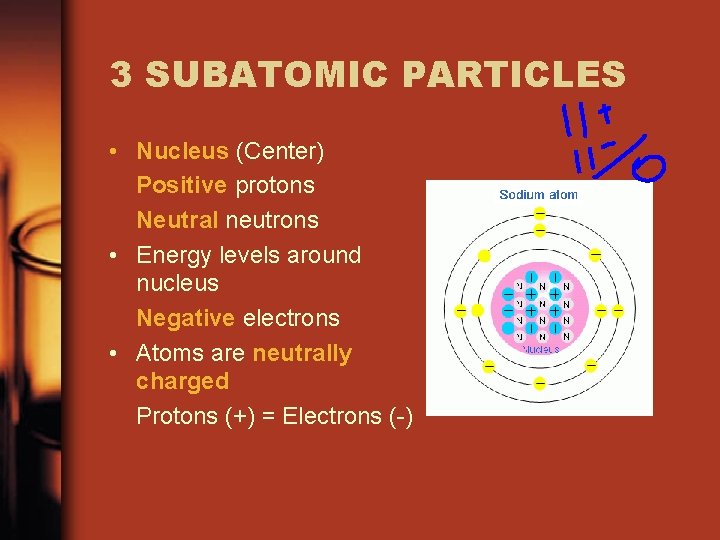
3 SUBATOMIC PARTICLES • Nucleus (Center) Positive protons Neutral neutrons • Energy levels around nucleus Negative electrons • Atoms are neutrally charged Protons (+) = Electrons (-)

PERIODIC TABLE

NUMBERS • Atomic number: number of protons Protons identify the element • Mass number: number of protons + neutrons • Neutrons = mass number – protons

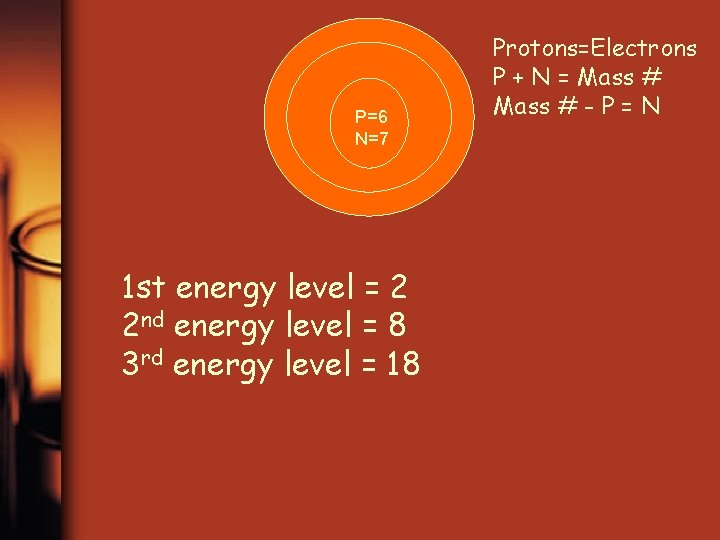
P=6 N=7 1 st energy level = 2 2 nd energy level = 8 3 rd energy level = 18 Protons=Electrons P + N = Mass # - P = N

CALCULATIONS At. # 8 __ 2 P N ___ 12 ___ 8 13 ___ E Mass # ____ ___ 5

ISOTOPES • Same number of protons but different number of neutrons and different masses • Atom: smallest unit of all matter • Element: made of only 1 kind of atom

ISOTOPES • Atoms have an atomic symbol, atomic number, and atomic mass.

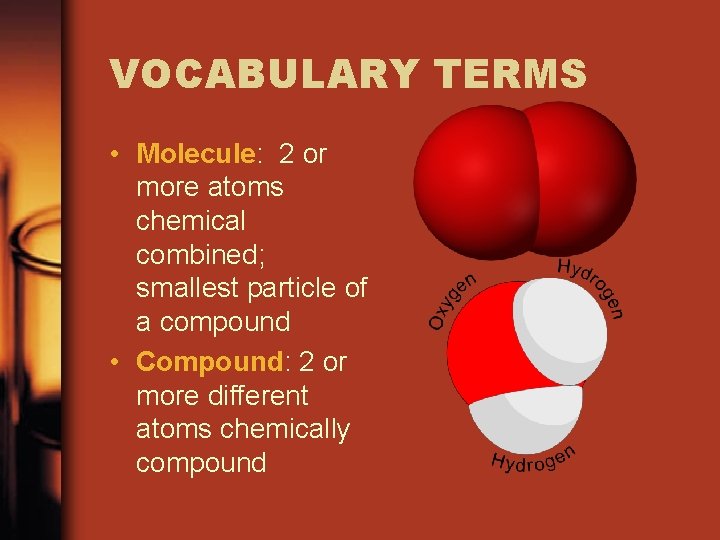
VOCABULARY TERMS • Molecule: 2 or more atoms chemical combined; smallest particle of a compound • Compound: 2 or more different atoms chemically compound

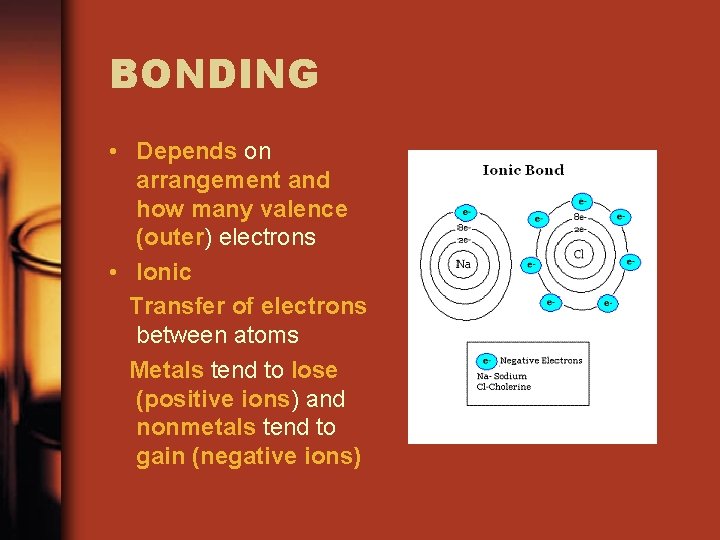
BONDING • Depends on arrangement and how many valence (outer) electrons • Ionic Transfer of electrons between atoms Metals tend to lose (positive ions) and nonmetals tend to gain (negative ions)

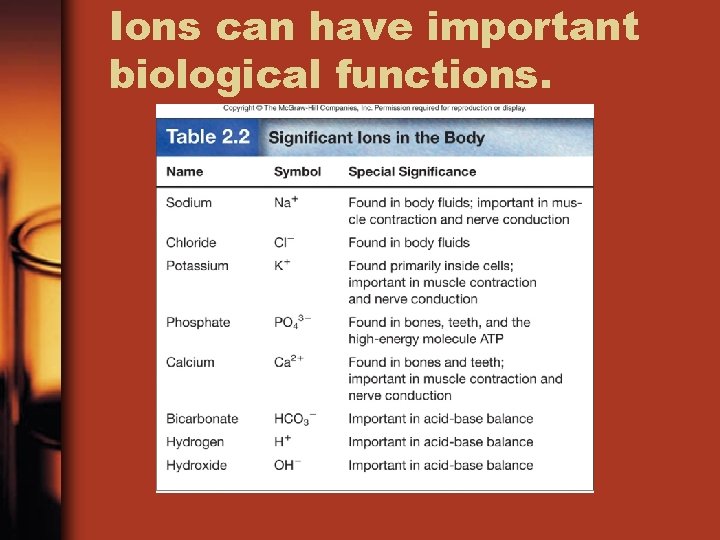
Ions can have important biological functions.

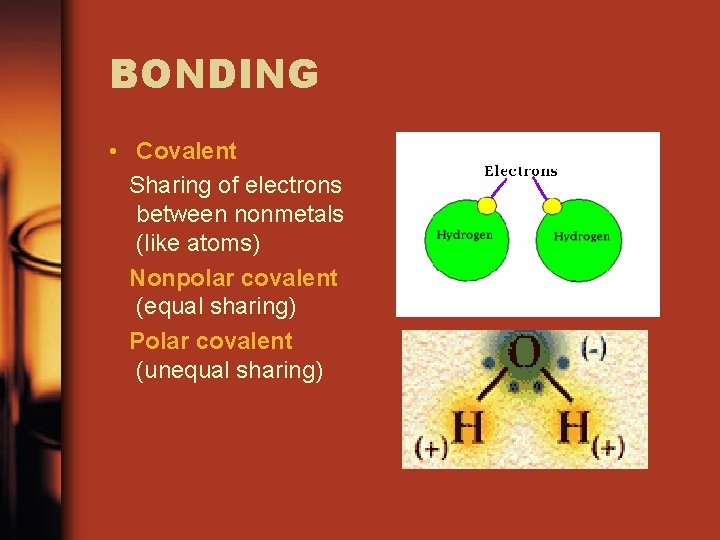
BONDING • Covalent Sharing of electrons between nonmetals (like atoms) Nonpolar covalent (equal sharing) Polar covalent (unequal sharing)

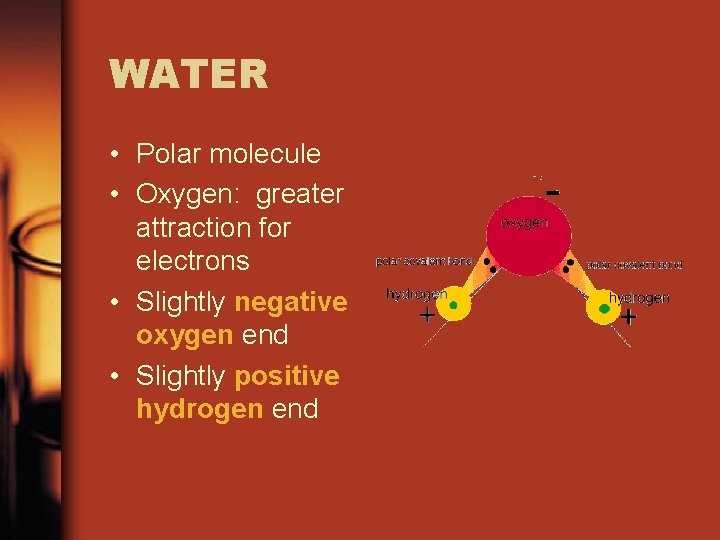
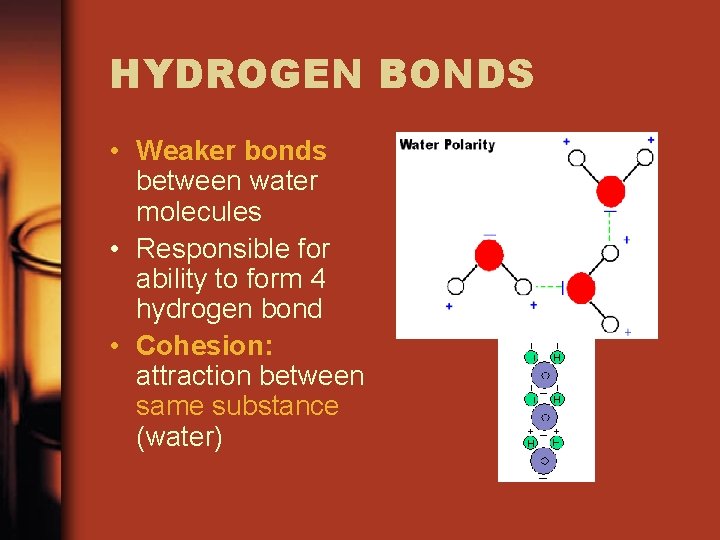
WATER • Polar molecule • Oxygen: greater attraction for electrons • Slightly negative oxygen end • Slightly positive hydrogen end

HYDROGEN BONDS • Weaker bonds between water molecules • Responsible for ability to form 4 hydrogen bond • Cohesion: attraction between same substance (water)

PROPERTIES OF WATER DUE TO COVALENT BOND • Surface tension: adhesion (different molecules) and cohesion (like molecules) • Capillary action: movement of water up roots • http: //www. youtube. com/watch? feature=pla yer_detailpage&v=VHn. FMPxte. Go

CHARACTERISTICS OF WATER • High specific heat: water warms and cools very slowly Biological importance: aquatic organisms are able to adjust slowly to the changing environment. • Freezing: bonds expand ice becomes less dense and will float Biological importance: top frozen layer helps insulate lake/pond

FREEZING • Ice is less dense as a solid than as a liquid (ice floats) • Frozen water forms a crystal-like lattice with molecules set at fixed distances.

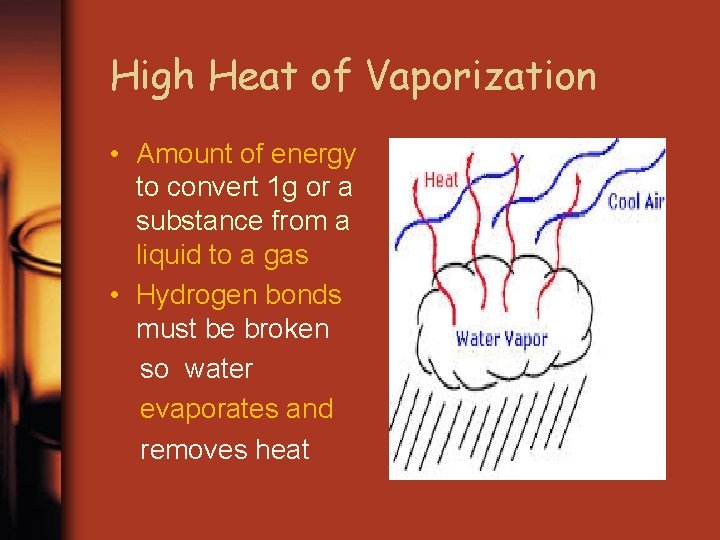
High Heat of Vaporization • Amount of energy to convert 1 g or a substance from a liquid to a gas • Hydrogen bonds must be broken so water evaporates and removes heat

Solutions & Suspensions • Many things dissolve in water--the Universal Solvent • Usually part of a mixture. • Two types of mixtures: Solutions Suspensions Defn: material composed of 2 or more substances physically combined

SOLUTIONS • Evenly distributed substances • Solute: material dissolved • Solvent: dissolving material • Solute: salt Solvent: water

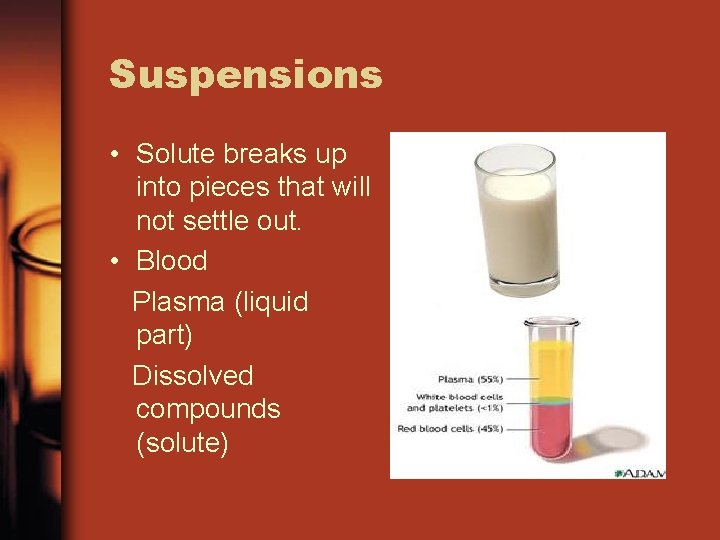
Suspensions • Solute breaks up into pieces that will not settle out. • Blood Plasma (liquid part) Dissolved compounds (solute)

ACIDS, BASES, p. H • p. H: measures concentration of H+ • 1 -14 scale • Below 7: acidic • 7: neutral • Above 7: basic

ACIDS/BASES • Acid: form H+ in solution • Bases: form OHin solution

REVIEW • Which bond is formed by the transfer of electrons between atoms? (Ionic) • Which bond is the weakest and forms between water molecules? (hydrogen) • Acids form ____ ions in solution. (H+ --hydrogen ions) • What is the solute? (material dissolved in solution)

WATER CHARACTERISTICS • http: //www. youtube. com/watch? feat ure=player_detailpage&v=0 e. NSnj 4 Z f. Z 8

REVIEW • What is adhesion? (attraction between molecules of different substances) • What is the importance of capillary action? (pulls water into roots) • Name the bond formed from unequal sharing of electrons. (polar covalent bond)