ATOMIC STRUCTURE AS Chemistry Basic Atomic Structure Hy

ATOMIC STRUCTURE
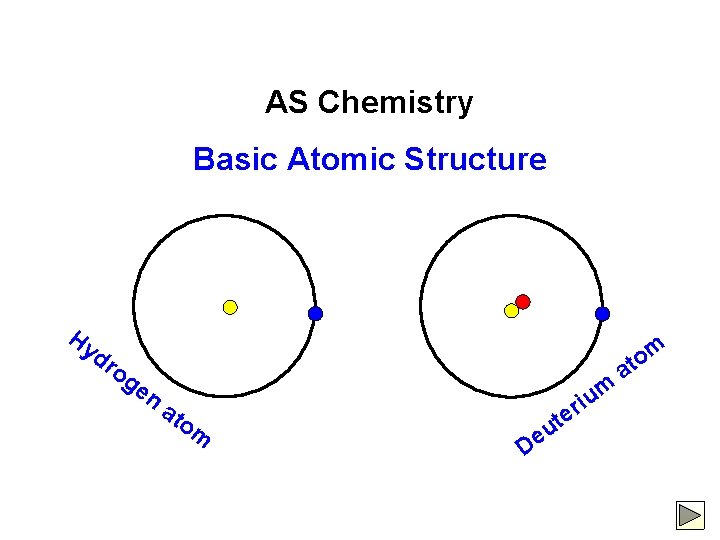
AS Chemistry Basic Atomic Structure Hy dr og en at om u e D iu r te m o at m

Structure of the atom A hundred years ago people thought that the atom looked like a “plum pudding” – a sphere of positive charge with negatively charged electrons spread through it… Ernest Rutherford, British scientist: I did an experiment that proved this idea was wrong. I called it the “Rutherford Scattering Experiment”
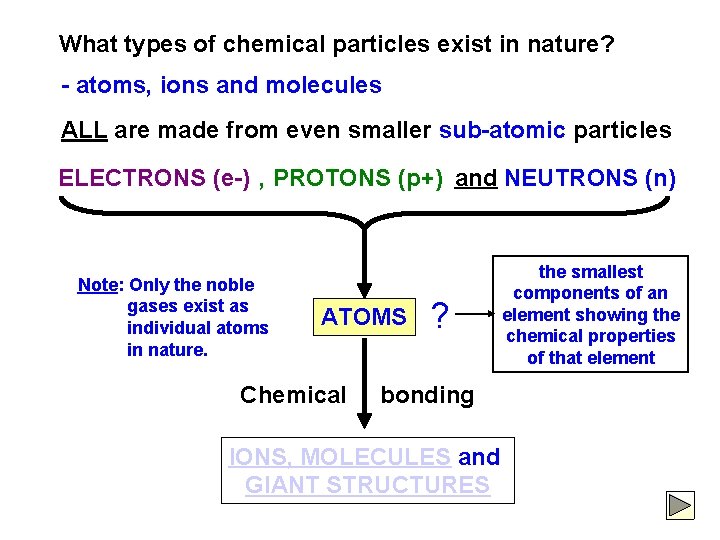
What types of chemical particles exist in nature? - atoms, ions and molecules ALL are made from even smaller sub-atomic particles ELECTRONS (e-) , PROTONS (p+) and NEUTRONS (n) Note: Only the noble gases exist as individual atoms in nature. ATOMS Chemical ? bonding IONS, MOLECULES and GIANT STRUCTURES the smallest components of an element showing the chemical properties of that element
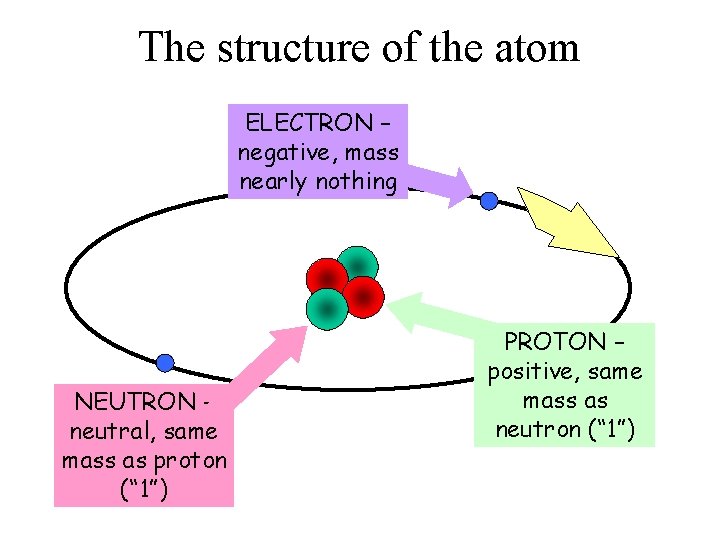
The structure of the atom ELECTRON – negative, mass nearly nothing NEUTRON – neutral, same mass as proton (“ 1”) PROTON – positive, same mass as neutron (“ 1”)

BASIC ATOMIC STRUCTURE Rutherford-Bohr model of the atom is used. Atoms are made up of electrons, protons & neutrons in overall spherical shape. Protons & neutrons in central, very small NUCLEUS and electrons ORBITING around nucleus. - electron orbits called ENERGY (or QUANTUM) LEVELS - levels numbered 1, 2, 3 etc away from nucleus 1 2 3

How do we know the existence and basic arrangements of protons, electrons and neutrons ? - by interpretation of experimental results! 1. Sir J J Thompson detected the electron and measured its mass : charge ratio. 2. Lord Rutherford detected the proton and measured its mass : charge ratio. 3. Millikan measured the charge on electron. 4. Chadwick detected and measured the properties of neutron. protons and neutrons have equal mass electrons have 1/1840 th of the mass of protons and electrons have equal but opposite charge neutrons have no charge
![5. Geiger-Marsden’s Experiment : Alpha particles, [ He 2+ ] were fired at a 5. Geiger-Marsden’s Experiment : Alpha particles, [ He 2+ ] were fired at a](http://slidetodoc.com/presentation_image_h2/6cec049d0953eda9eddb951b959a6739/image-8.jpg)
5. Geiger-Marsden’s Experiment : Alpha particles, [ He 2+ ] were fired at a thin gold foil Most pass through unaffected Few pass through but deflected VERY few reflected back towards source Suggests that: the mass (protons + neutrons) and positive charge (protons) of atom are concentrated in VERY small part of the atom ie the NUCLEUS and most of the atom’s volume is empty space containing the electrons. ie the ENERGY LEVELS

Effects of Electrical and Magnetic Fields on Protons, Electrons and Neutrons Electrical Magnetic p e S + n n N - p e = beam containing protons, electrons & neutrons NOT affected because no electrical charge p and e deflected in opposite directions because oppositely charged Protons deflected less than electron because heavier
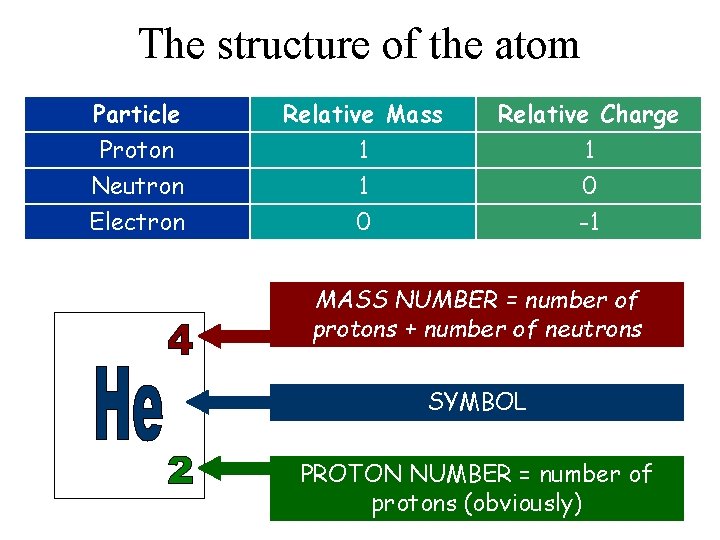
The structure of the atom Particle Proton Neutron Electron Relative Mass 1 1 0 Relative Charge 1 0 -1 MASS NUMBER = number of protons + number of neutrons SYMBOL PROTON NUMBER = number of protons (obviously)

SUMMARY : Protons, electrons and neutrons are the building blocks of ALL atoms, ions, molecules etc in nature. MASS NAME OF SUB RELATIVE -ATOMIC TO A PARTICLE PROTON Electron Proton Neutron ELECTRICAL CHARGE RELATIVE TO PROTON POSITION WITHIN THE ATOM 1 / 1840 1 - In quantum levels around nucleus 1 1+ In central, very small nucleus 0 In central, very small nucleus 1

RELATED DEFINITIONS LEARN!! 1. ATOMIC (PROTON) NUMBER, Z of an element = the number of protons in an atom (also equal to the number of electrons and the position in the Periodic Table) 2. MASS NUMBER, A of an atom = number of protons (Z) + number of neutrons (N) in the nucleus of the atom. A = Z + N (No units!) 3. ISOTOPES = atoms of the SAME ELEMENT (same Z) with DIFFERENT MASS NUMBERS (A) Isotopes have different numbers of NEUTRONS. NB Isotopes are chemically IDENTICAL!

4. NUCLIDE = a symbol of an atom showing its atomic number (Z) and a mass number (A). represents a PARTICULAR ISOTOPE eg A 20 Ne Z 10 Do not confuse this with the symbol used in the Periodic Table ! 20. 1 Ne 10 “ 20. 1” is the “relative atomic mass, Ar” (see later) which averages the mass numbers of the isotopes present in any element
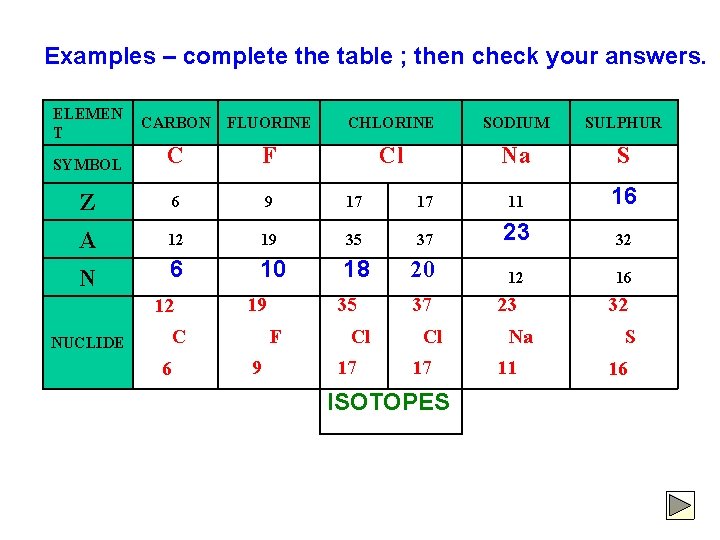
Examples – complete the table ; then check your answers. ELEMEN T CARBON FLUORINE CHLORINE SODIUM SULPHUR SYMBOL C F Cl Na S Z 6 9 17 17 11 16 A 12 19 35 37 23 32 6 10 18 20 12 16 N 12 NUCLIDE 19 F C 6 35 9 Cl 17 37 Cl 17 ISOTOPES 23 Na 11 32 S 16
- Slides: 14