Today Webquest Over the history of the atom

Today: Webquest • Over the history of the atom, atomic models, and atomic structure You can access links, powerpoint, and typable worksheet on: Teacherarrowsmith. com/chemistry Click on Atoms and Atomic Scientists Webquest link Choices: You can type and email to me: Victoria. Arrowsmith@ops. org Or turn in hard copy

Part 1: Atomic Scientists to Know http: //www. timetoast. com/timelines/atomic-scientist - * Know what they are important for regarding atomic theory. • Dmitri Mendeleev • John Dalton • Niels Bohr • JJ Thomson • Ernest Rutherford • Heisenberg

QUICK NOTES Part 2: John Dalton - What was his theory ? His 5 main parts to the atomic theory? • Proposed the atomic theory 1. *all matter is made up of atoms 2. Each element is composed of a single unique type of atom 3. Elements can combine to form chemical _________? (find all 5 main points of atomic theory on webquest Part II)

Part 3: JJ Thomson What did he discover? What was his atomic model called? • JJ Thomson used a cathode ray tube to test the properties of the particles • Shot cathode ray at opposite charged plates: ray repelled from negative charged plate, attracted to positive charged plate • cathode ray made up of negative charge particles: electrons • negative particles exist as part of atoms
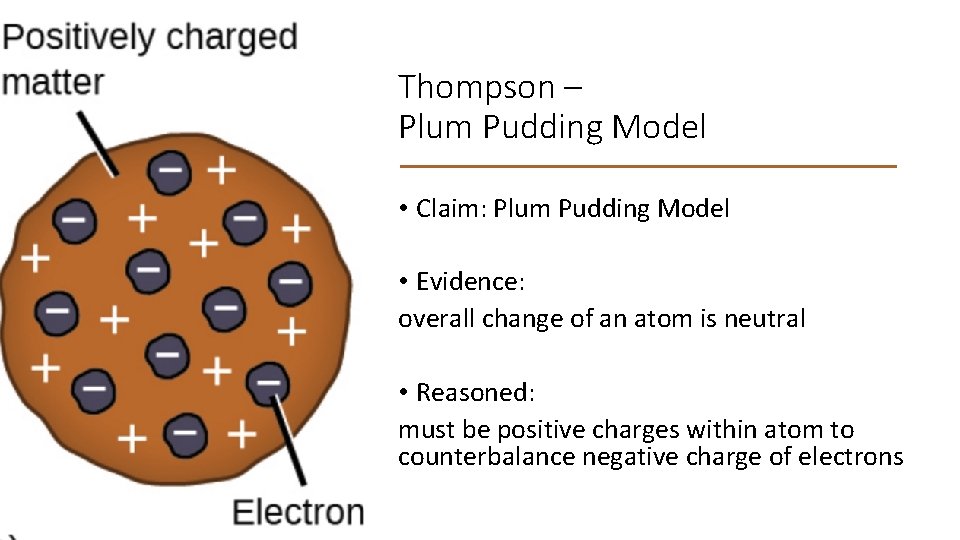
Thompson – Plum Pudding Model • Claim: Plum Pudding Model • Evidence: overall change of an atom is neutral • Reasoned: must be positive charges within atom to counterbalance negative charge of electrons
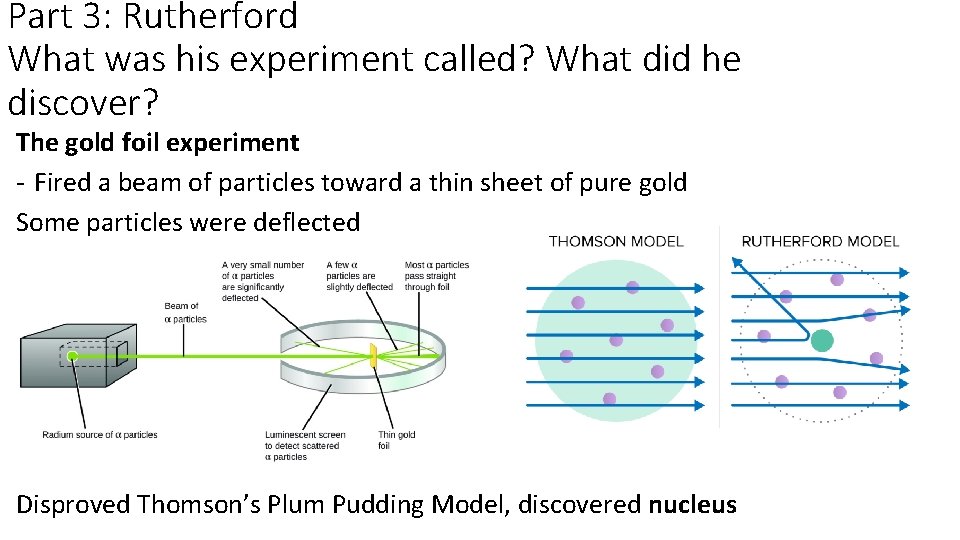
Part 3: Rutherford What was his experiment called? What did he discover? The gold foil experiment - Fired a beam of particles toward a thin sheet of pure gold Some particles were deflected Disproved Thomson’s Plum Pudding Model, discovered nucleus

Today: Webquest • Over the history of the atom, atomic models, and atomic structure You can access links, powerpoint, and typable worksheet on: Teacherarrowsmith. com/chemistry Click on Atoms and Atomic Scientists Webquest link Choices: You can type and email to me: Victoria. Arrowsmith@ops. org Or turn in hard copy **due at end of class

Study Guide • Dalton’s 5 main parts to his Atomic Theory • What are atoms made up of? • What is formed when elements are combined? • What subatomic particle did JJ Thompson discover? • How did Thompson discover this particle? What tool did he use? What was his conclusion? • What was Thompson’s atomic model called? • How did Rutherford disprove Thompson? • What was Rutherford’s experiment called? and what did he discover?
- Slides: 8