The Classical Second Law of Thermodynamics April 17


















- Slides: 42

The Classical Second Law of Thermodynamics April 17, 2012 Prof. Sanghee Kim 1 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

First Law : energy conservation in any ordinary process (no restriction on the direction of heat and work flow) • ex) A hot cup of coffee cools by virtue of heat transfer to the surroundings, • but heat will not flow from the cooler surroundings to the hotter cup of coffee. processes proceed in a certain direction Second Law : existence of such a restriction (difference in quality bwn heat & work ) 2 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

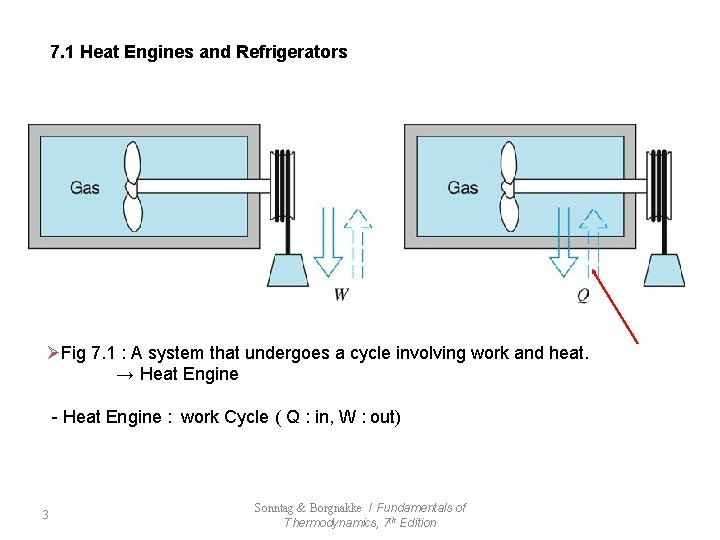
7. 1 Heat Engines and Refrigerators ØFig 7. 1 : A system that undergoes a cycle involving work and heat. → Heat Engine - Heat Engine : work Cycle ( Q : in, W : out) 3 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

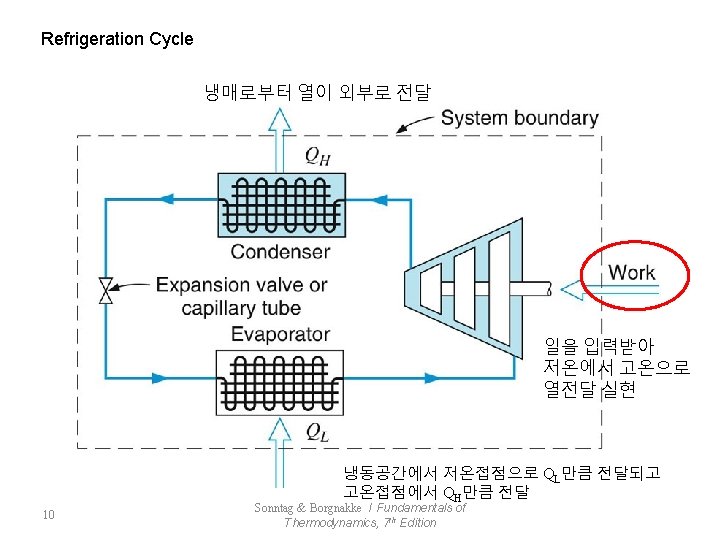
열전달만으로 두과정이 하나의 사이클을 완성할 수 없음 Fig 7. 2 : An example showing the impossibility of completing a cycle by transferring heat from a low-temperature body to a high-temperature body → Refrigerator (Heat Pump) QL - Refrigerator : work Cycle (TL → TH, W : in) (Heat Pump) 4 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

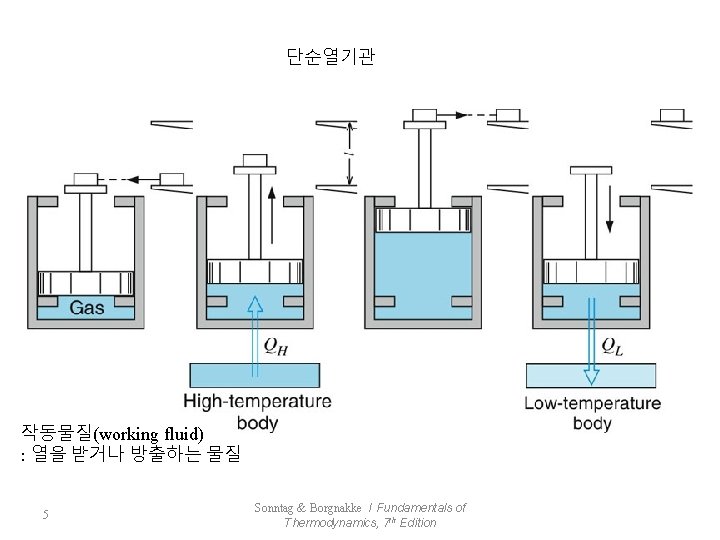
단순열기관 작동물질(working fluid) : 열을 받거나 방출하는 물질 5 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

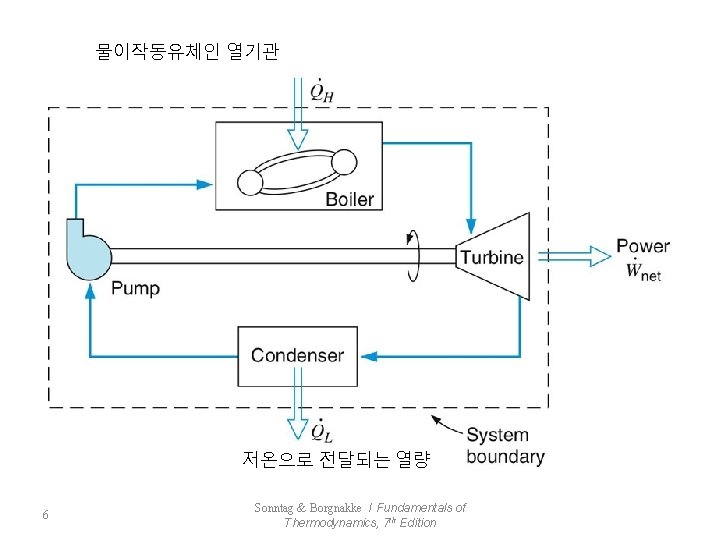
물이작동유체인 열기관 저온으로 전달되는 열량 6 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

Thermal Efficiency of a Heat Engine (7. 1) 7 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

8 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

9 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition


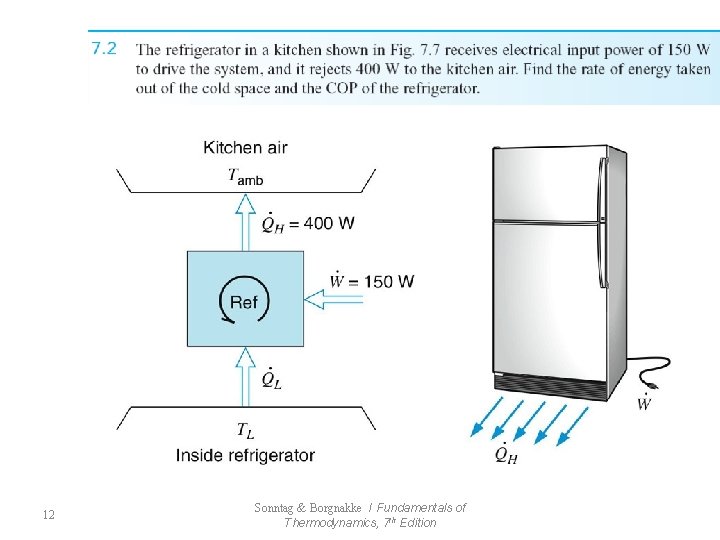
• Refrigerator (7. 2) Thermal Reservoir : heat can be transferred indefinitely without change in the temperature of the reservoir (열저장조: 저장조의 온도변화없이 무한정 으로 열교환/열저장조의 온도는 항상 일정) - from Source(발열원) or Sink (흡열원) ex) atmosphere , ocean 11 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

12 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

13 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

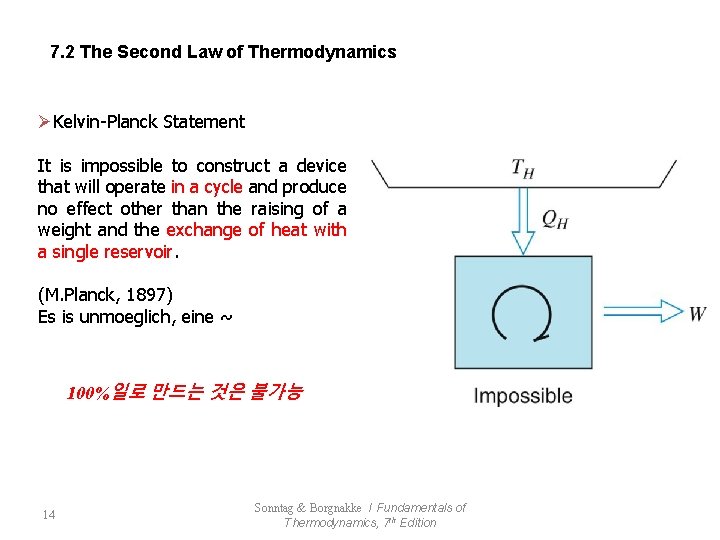
7. 2 The Second Law of Thermodynamics ØKelvin-Planck Statement It is impossible to construct a device that will operate in a cycle and produce no effect other than the raising of a weight and the exchange of heat with a single reservoir. (M. Planck, 1897) Es is unmoeglich, eine ~ 100%일로 만드는 것은 불가능 14 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

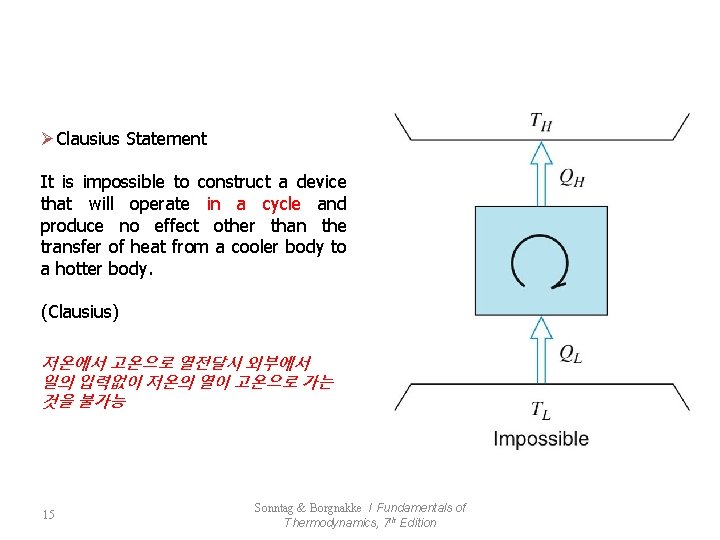
ØClausius Statement It is impossible to construct a device that will operate in a cycle and produce no effect other than the transfer of heat from a cooler body to a hotter body. (Clausius) 저온에서 고온으로 열전달시 외부에서 일의 입력없이 저온의 열이 고온으로 가는 것을 불가능 15 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

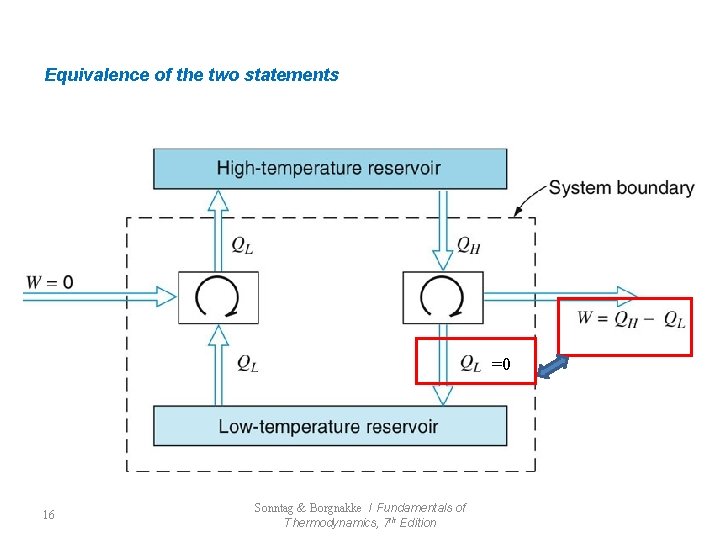
Equivalence of the two statements =0 16 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

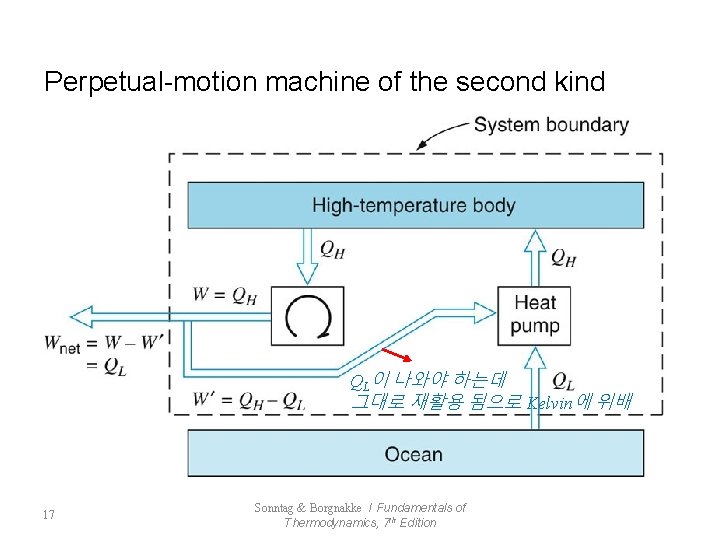
Perpetual-motion machine of the second kind QL이 나와야 하는데 그대로 재활용 됨으로 Kelvin에 위배 17 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

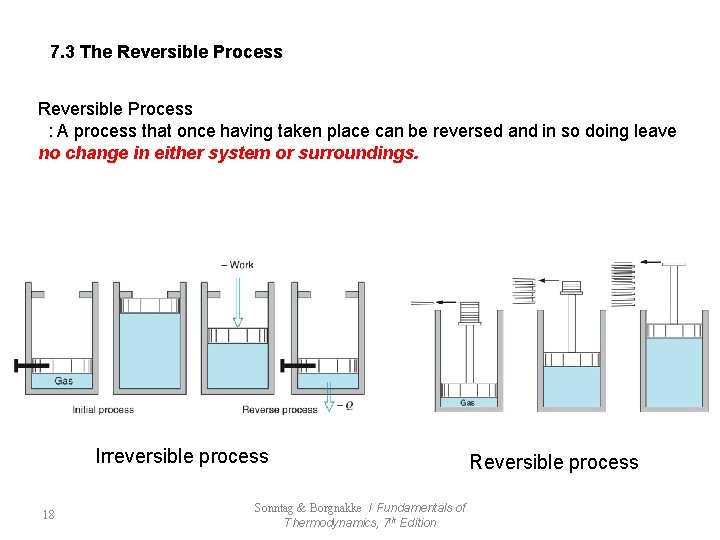
7. 3 The Reversible Process : A process that once having taken place can be reversed and in so doing leave no change in either system or surroundings. Irreversible process 18 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition Reversible process

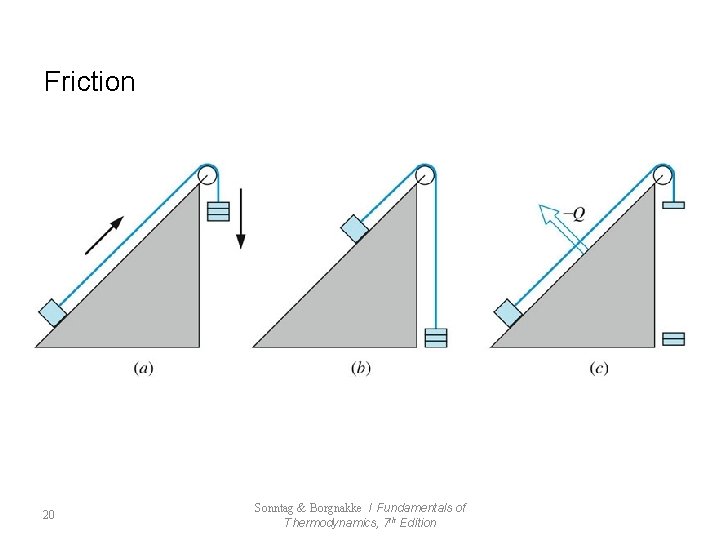
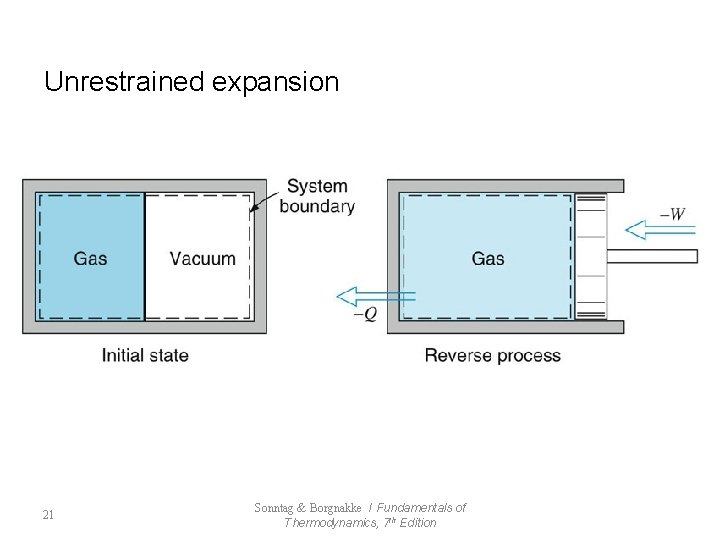
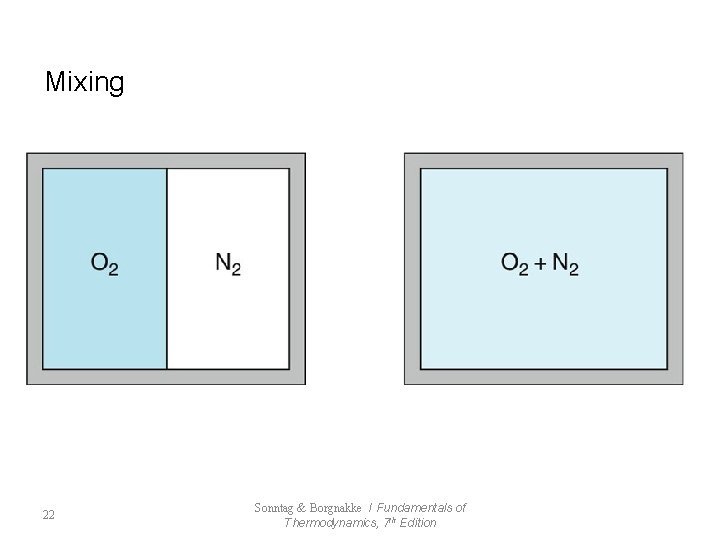
7. 4 Factors that Render Processes Irreversible l Reversible Process - frictionless mechanical process - frictionless adiabatic state change l Irreversible Process : impossible to return to original state (All Natural Process) - Friction - Unrestrained Expansion - Heat transfer Through a Finite Temperature Difference - Mixing of Two Different Substances - Other Factors 19 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

Friction 20 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

Unrestrained expansion 21 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

Mixing 22 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

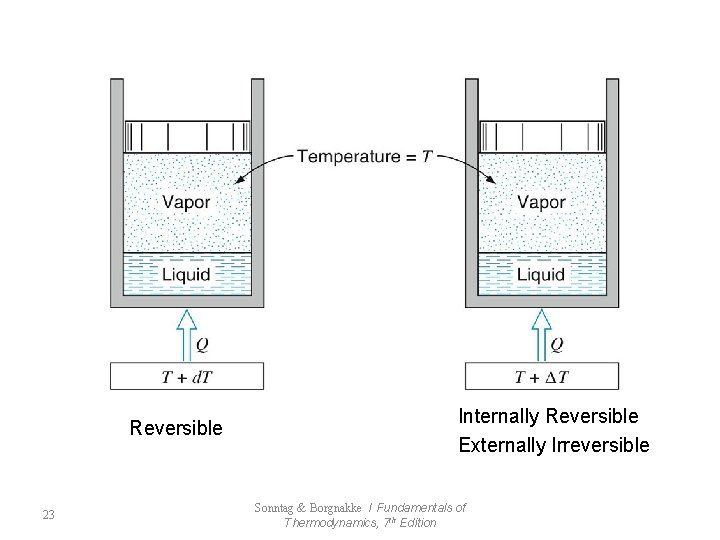
Reversible 23 Internally Reversible Externally Irreversible Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

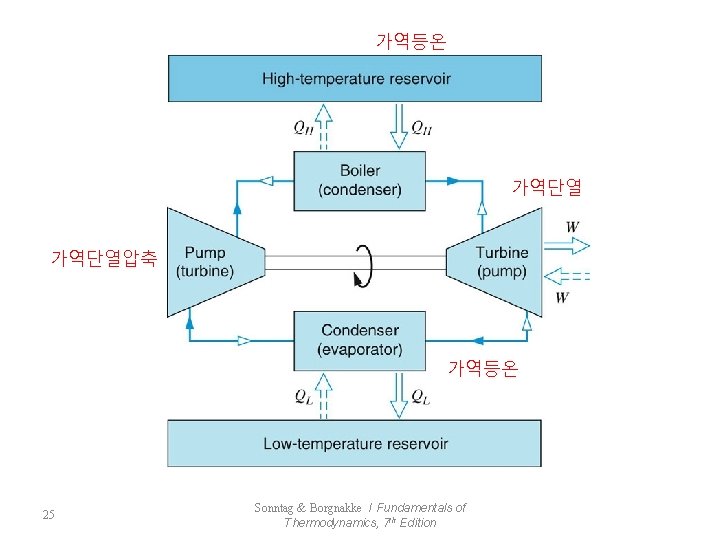
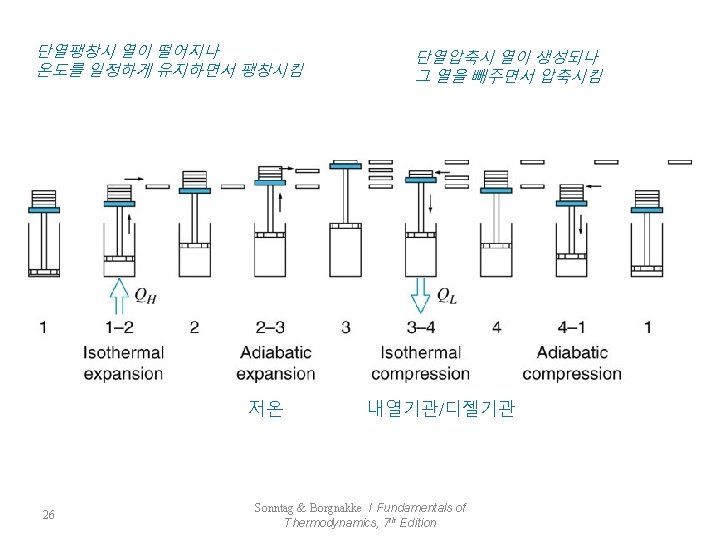
7. 5 The Carnot Cycle - Reversible : the most efficient cycle that can operate between two constant-temperature reservoirs 가역과정에서 가장 효율이 높은 사이클 1 : rev. isothermal process, QH is transferred to the system 2 : rev. adiabatic process, Working Fluid : TH→TL 3 : rev. isothermal process, QL is rejected from the system 4 : rev. adiabatic process, Working Fluid : TL→TH 24 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

가역등온 가역단열압축 가역등온 25 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition


7. 6 Two Propositions Regarding the Efficiency of Carnot Cycle Ø First Proposition Ø Second Proposition 27 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

7. 7 Thermodynamic Temperature Scale Carnot 사이클은 온도만의 함수 Carnot 사이클의 효율과 절대온도의 관계로 표시가능 28 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

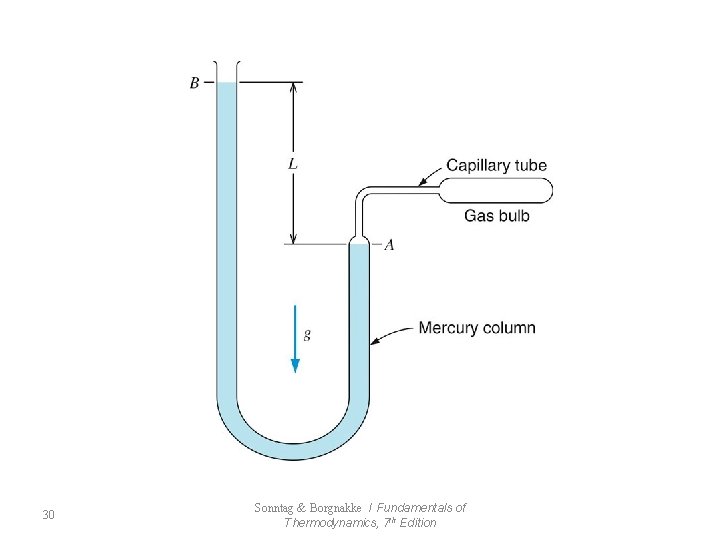
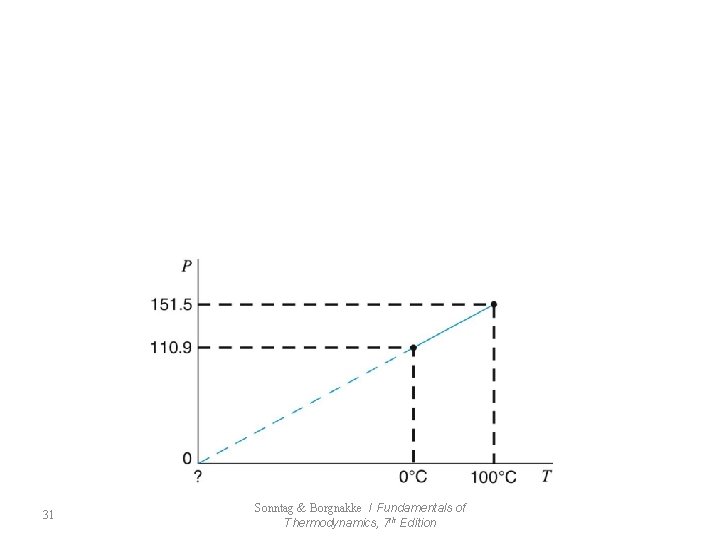
7. 8 The Ideal-Gas Temperature Scale 29 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

30 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

31 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

32 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

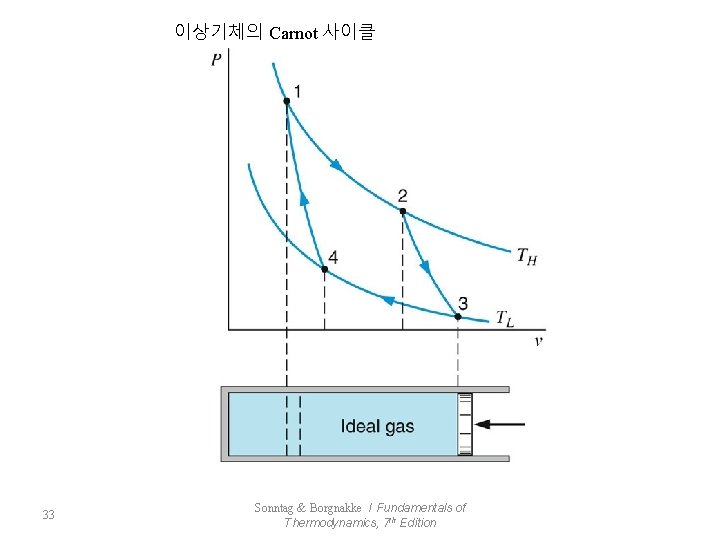
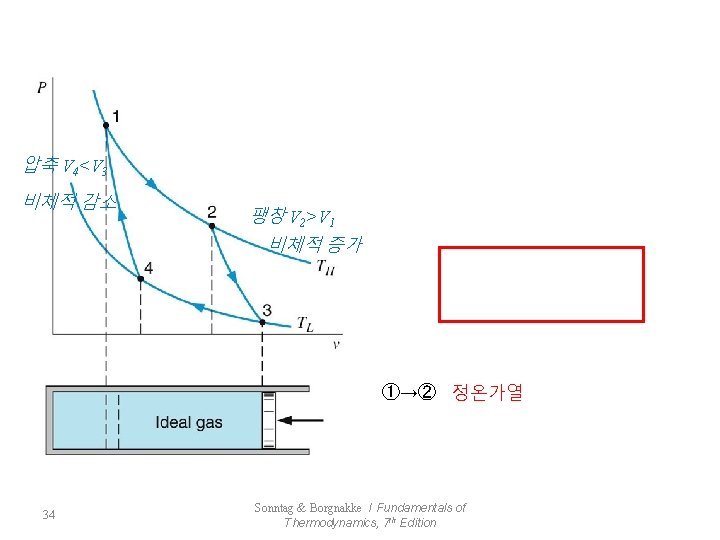
이상기체의 Carnot 사이클 33 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

압축 V 4<V 3 비체적 감소 팽창 V 2>V 1 비체적 증가 ①→② 정온가열 34 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

단열팽창 ②→③ * 정온방열 ③→④ 단열압축 ④→① 35 * Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

7. 9 Ideal vs. real machines 가역(Carnot)사이클 실제열기관/냉동기/열펌프 효율이 낮음 36 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

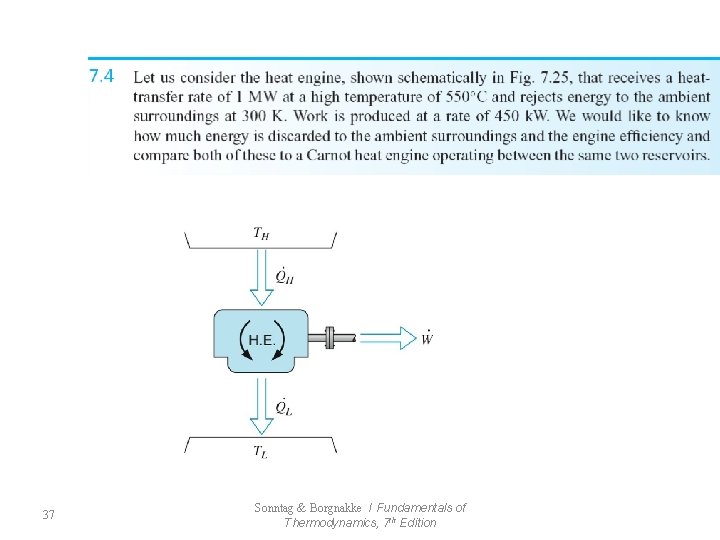
37 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

38 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

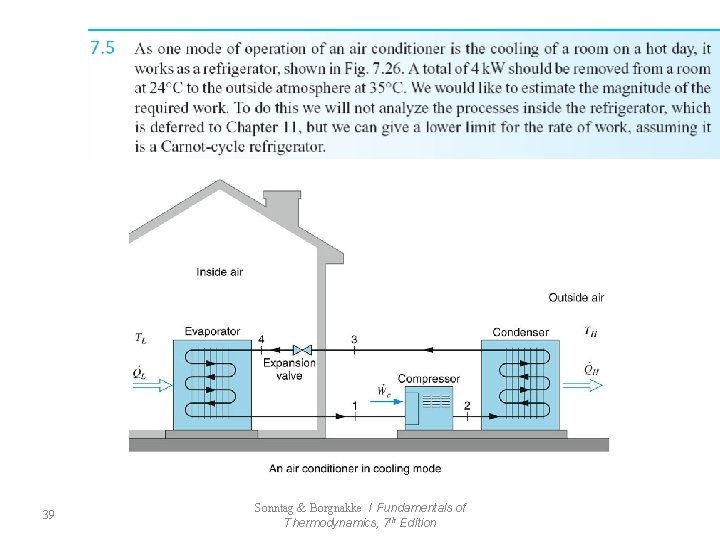
39 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

40 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

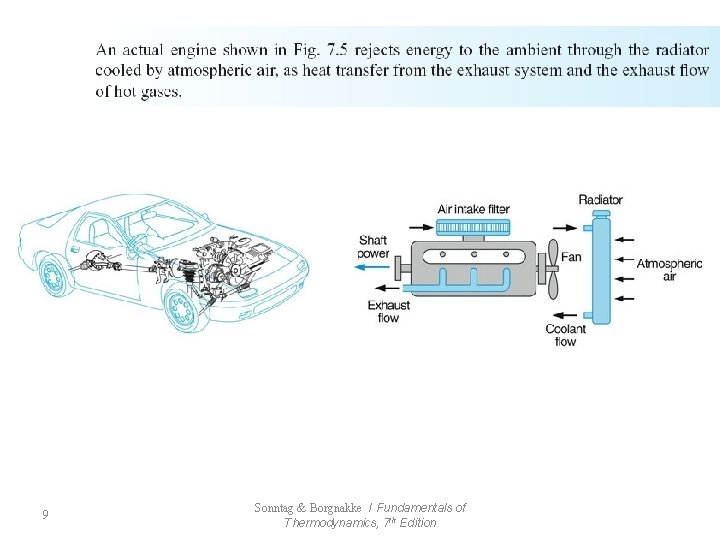
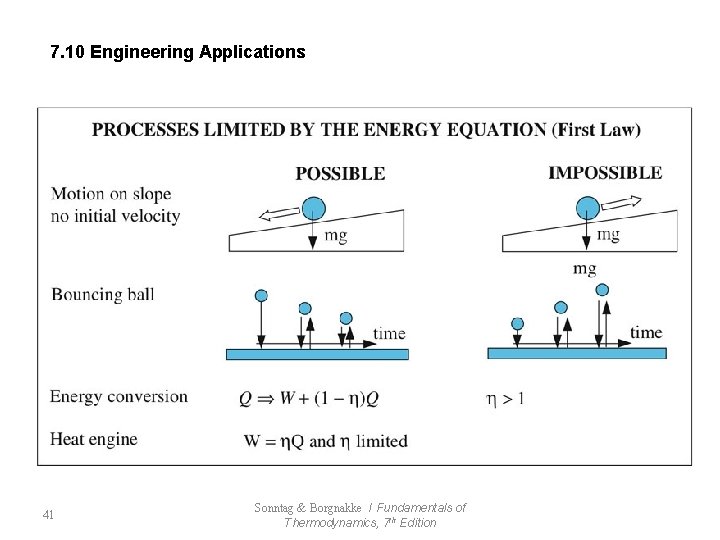
7. 10 Engineering Applications 41 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition

42 Sonntag & Borgnakke / Fundamentals of Thermodynamics, 7 th Edition