STATES OF MATTER REVIEW States of Matter Mass
























- Slides: 28

STATES OF MATTER REVIEW

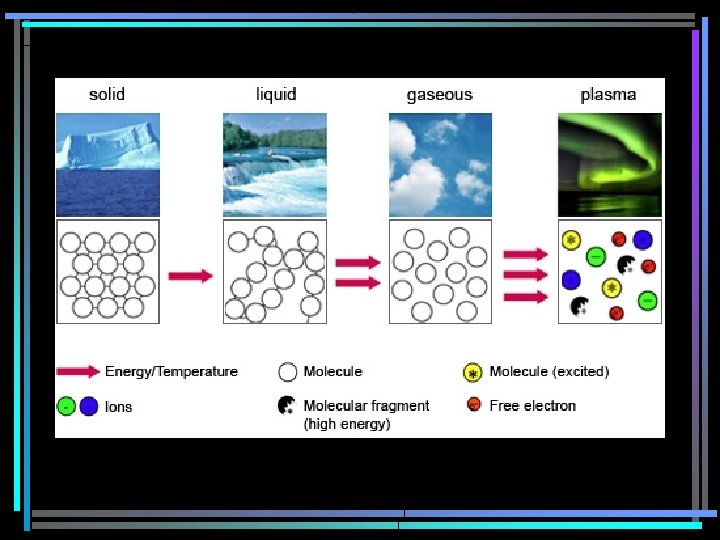
States of Matter • Mass & takes up space • No 2 pieces occupy the same space at the same time • 4 forms - depends on temperature – Solids – Liquids – Gases – Plasma

Solids • Definite volume & shape • Not enough energy to move – Crystalline – repeating geometric patterns – Noncrystalline – amorphous no true form, thick liquids

Liquids • Definite volume • No definite shape – takes shape of container • Particles have enough energy to move – Viscosity – property of how easily liquid flows

Gases • No definite volume or shape • Takes volume & shape of container • Particles have enough energy to overcome attractive forces holding them together & fill the container

Plasma • Like a gas • Lots of energy • Electrically charged, fast moving particles – the sun, space shuttle, lightening


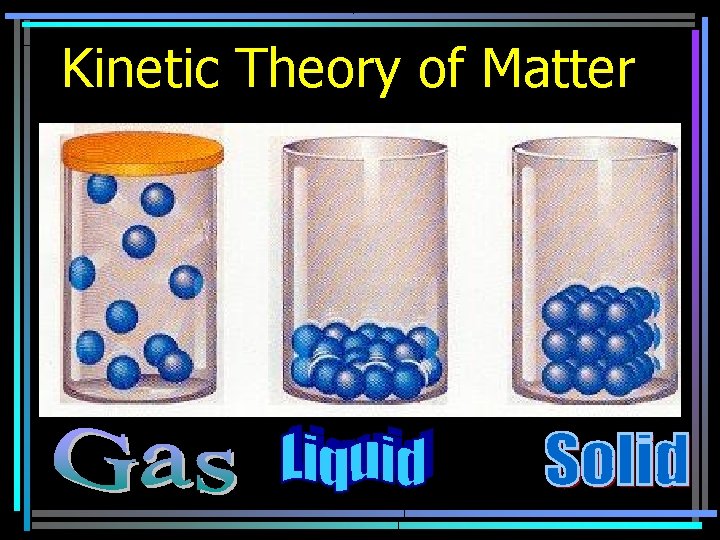
Kinetic Theory of Matter • Matter is made of tiny particles in constant motion • temperature, motion • KE = energy of motion • PE = stored energy

Kinetic Theory of Matter

Thermal Expansion • Increase temp, increase energy: particles move faster & further apart increasing the volume – Concrete slabs on bridge

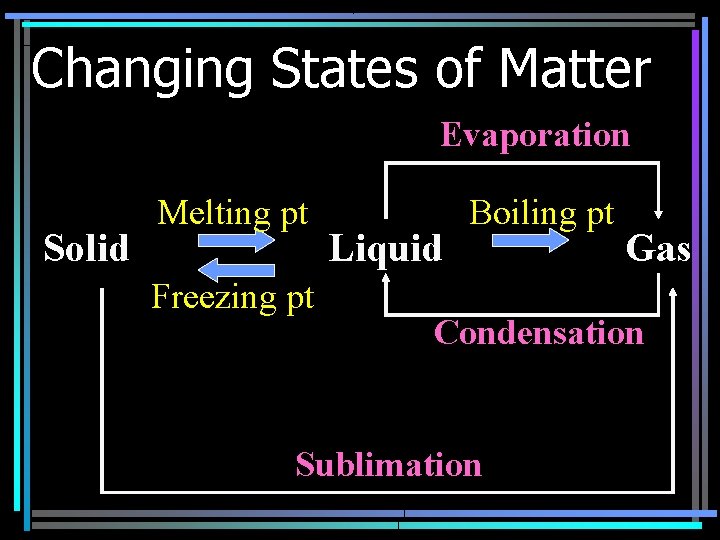
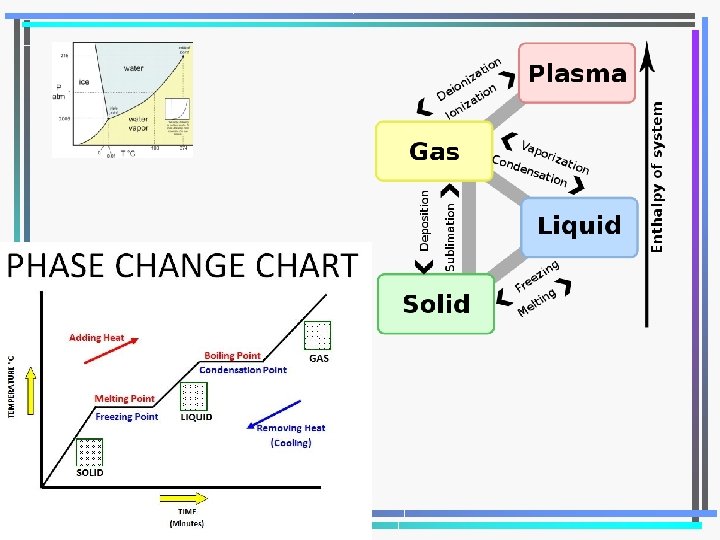
Changing States of Matter Evaporation Solid Melting pt Freezing pt Liquid Boiling pt Gas Condensation Sublimation


Heat of Fusion • Energy required for a substance to change from a solid to a liquid • Different for every substance • No temp Δ until complete – Ice to water 0°C until done – 334 k. J/kg for water

Heat of Vaporization • Energy required for a substance to change from a liquid to a gas • Different for every substance • No temp Δ until done – Water to steam 100°C – 2260 k. J/kg for water

Freezing Point Depression • Decrease the freezing point of water by dissolving particles in it – Salting icy roads & sidewalks • Ice melts as freezing pt is lowered – Ice cream makers • Ice Cream -3°C, Ice 0°C • Salt lowers freezing pt of ice so that ice cream can freeze

Boiling Point Elevation • Increase in the boiling pt of water by dissolving particles in it • Solute particles reduce how easily molecules get to surface to evaporate increasing bpt since water needs more energy

Part 2 Behavior of Gases & Fluids

• Pressure – amount of force per unit area (pascal, Pa) P = F/A • Atm. pressure @ sea level 101. 3 k. Pa – Pressure decreases w/ elevation: fewer gas particles • Gas Laws treat gases as ideal: no volume or attraction btwn molecules

Boyles Law • If you decrease the volume of a container of gas, the pressure of the gas will increase as long as temperature remains constant • Movie: Men of Honor

Boyles Law: constant temperature Pressure Volume

Charles Law • Volume of a gas increases as temp increases if pressure remains constant –Absolute Zero lowest possible temperature no movement of particles

Charles Law: constant pressure Temp. Volume

Fluids • Buoyancy – the ability of a fluid (liquid or gas) to exert an upward force on an object immersed in it – Force = weight… Floats – Force < weight… Sinks – Force > weight… Rises

Archimedes’ Principle • Bouyant force on an object in a fluid equals the weight of the fluid displaced by the object

Pascal’s Principle • Pressure applied to a fluid is transmitted unchanged throughout the fluid – Ex. pistons, toothpaste –P=F/A

Bernoulli’s Principle • As the velocity of a fluid increases, the pressure on it decreases – Ex. air plane wing

Venturi Effect • A fluid flows faster when forced through narrow spaces –Ex. windy cities
