ORGANIC CHEMISTRY Organic Chemistry The chemistry of carbon

ORGANIC CHEMISTRY Organic Chemistry: The chemistry of carbon and carbon-based compounds Organic Chemistry in everyday life: Smells & tastes: fruits, chocolate, fish, mint Medications: Aspirin, Tylenol, Decongestants, Sedatives Addictive substances: Caffeine, Nicotine, Alcohol, Narcotics Hormones/Neurotransmitters: Adrenaline, Epinephrine Food/Nutrients: Carbohydrates, Protein, Fat, Vitamins Genetics: DNA, RNA Consumer products: Plastics, Nylon, Rayon, Polyester
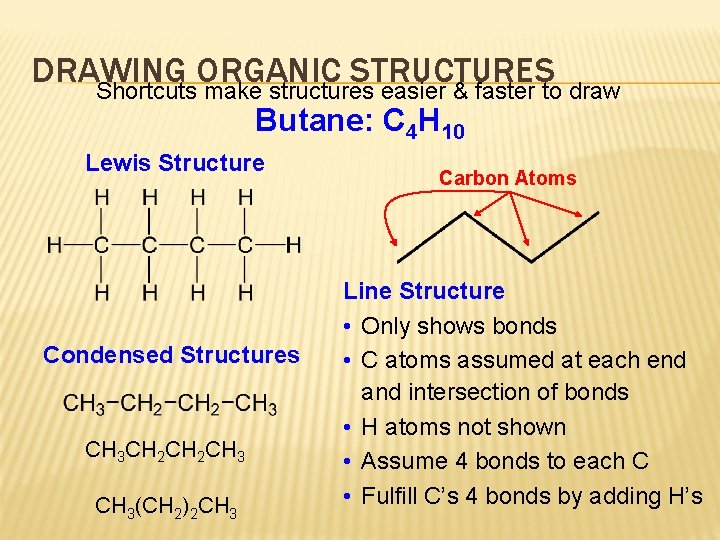
DRAWING ORGANIC STRUCTURES Shortcuts make structures easier & faster to draw Butane: C 4 H 10 Lewis Structure Condensed Structures CH 3 CH 2 CH 3(CH 2)2 CH 3 Carbon Atoms Line Structure • Only shows bonds • C atoms assumed at each end and intersection of bonds • H atoms not shown • Assume 4 bonds to each C • Fulfill C’s 4 bonds by adding H’s
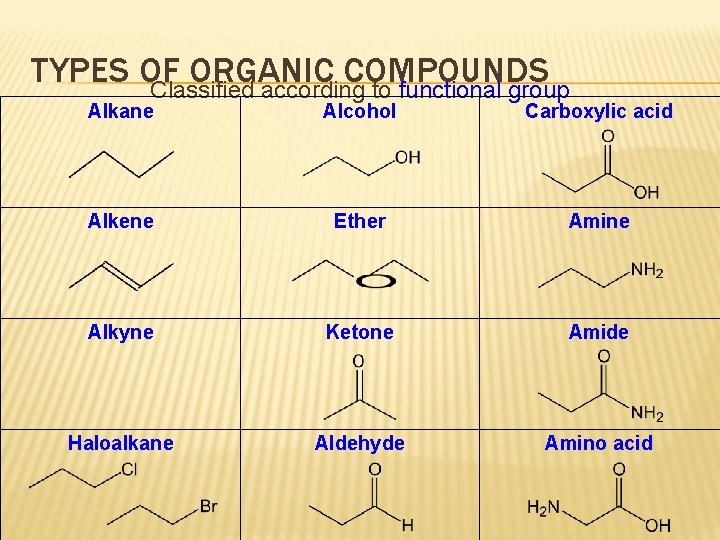
TYPES OF ORGANIC COMPOUNDS Classified according to functional group Alkane Alcohol Carboxylic acid Alkene Ether Amine Alkyne Ketone Amide Haloalkane Aldehyde Amino acid

BIG IDEA IN ORGANIC CHEMISTRY Structure controls Function Each functional group has predictable reactivity
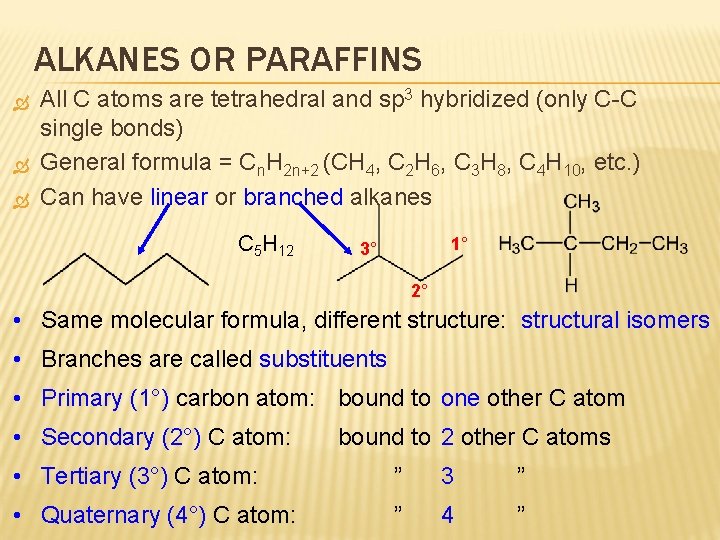
ALKANES OR PARAFFINS All C atoms are tetrahedral and sp 3 hybridized (only C-C single bonds) General formula = Cn. H 2 n+2 (CH 4, C 2 H 6, C 3 H 8, C 4 H 10, etc. ) Can have linear or branched alkanes C 5 H 12 1° 3° 2° • Same molecular formula, different structure: structural isomers • Branches are called substituents • Primary (1°) carbon atom: bound to one other C atom • Secondary (2°) C atom: bound to 2 other C atoms • Tertiary (3°) C atom: ” 3 ” • Quaternary (4°) C atom: ” 4 ”

ISOMERS The fat dog shook himself, and then rolled over on the wet rug. OR The dog shook the fat rug, then rolled over and wet on himself. These two statements use the same words. . . but have very different meanings! Likewise, isomers may have the same formula, but have very different structures…
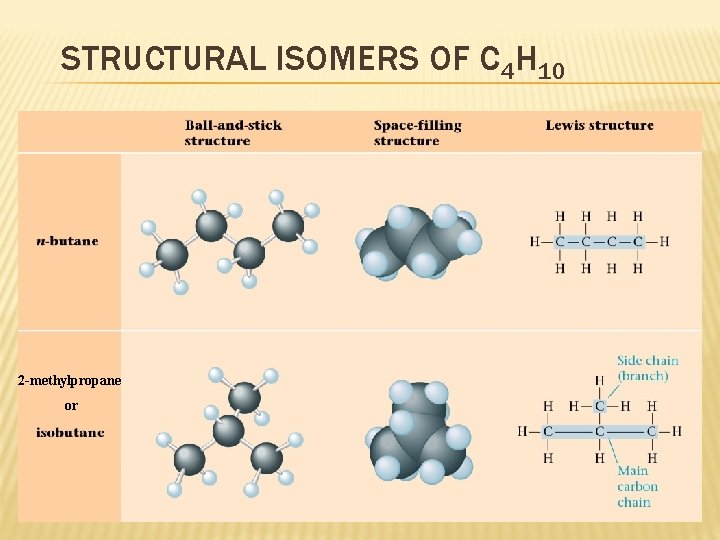
STRUCTURAL ISOMERS OF C 4 H 10 2 -methylpropane or

STRUCTURAL ISOMER PRACTICE On piece of your own paper, draw AND name ALL of the isomers for the following alkanes: Formulas Pentane C 5 H 12 Hexane C 6 H 14 Heptane C 7 H 16 # isomers 3 5 9 Some of your drawings may look different, but they are only different structures (isomers) if they also have different names If you complete that, try to draw and name all of the isomers for octane (C 8 H 18). There are 18 of them!
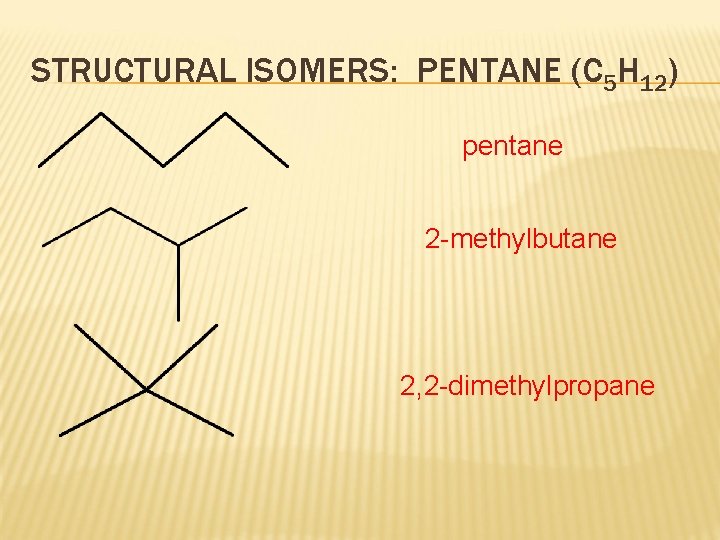
STRUCTURAL ISOMERS: PENTANE (C 5 H 12) pentane 2 -methylbutane 2, 2 -dimethylpropane
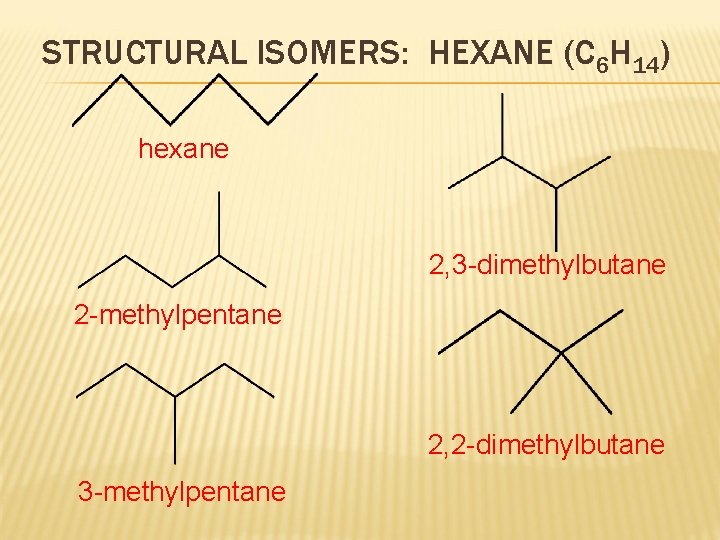
STRUCTURAL ISOMERS: HEXANE (C 6 H 14) hexane 2, 3 -dimethylbutane 2 -methylpentane 2, 2 -dimethylbutane 3 -methylpentane
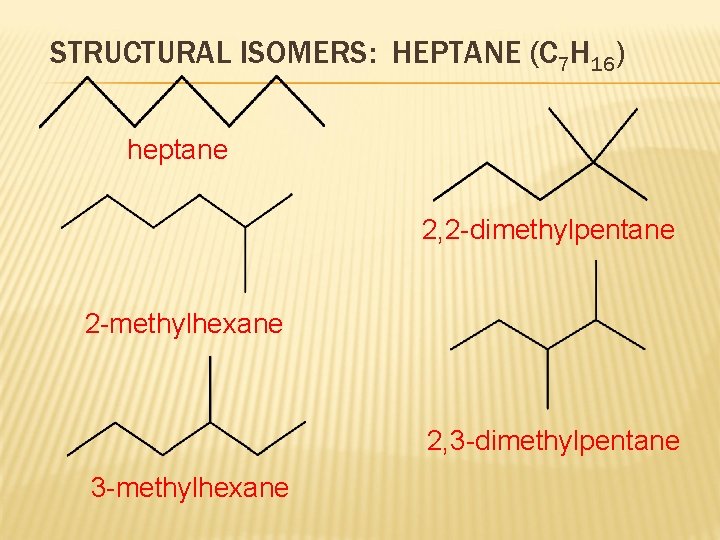
STRUCTURAL ISOMERS: HEPTANE (C 7 H 16) heptane 2, 2 -dimethylpentane 2 -methylhexane 2, 3 -dimethylpentane 3 -methylhexane
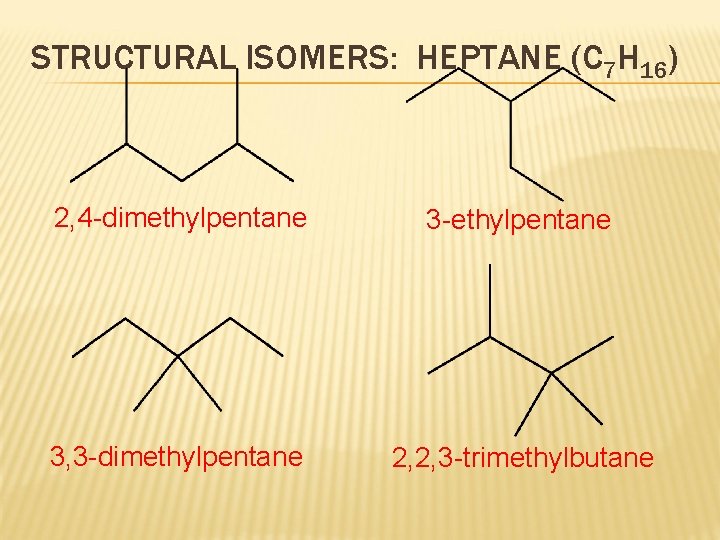
STRUCTURAL ISOMERS: HEPTANE (C 7 H 16) 2, 4 -dimethylpentane 3 -ethylpentane 3, 3 -dimethylpentane 2, 2, 3 -trimethylbutane
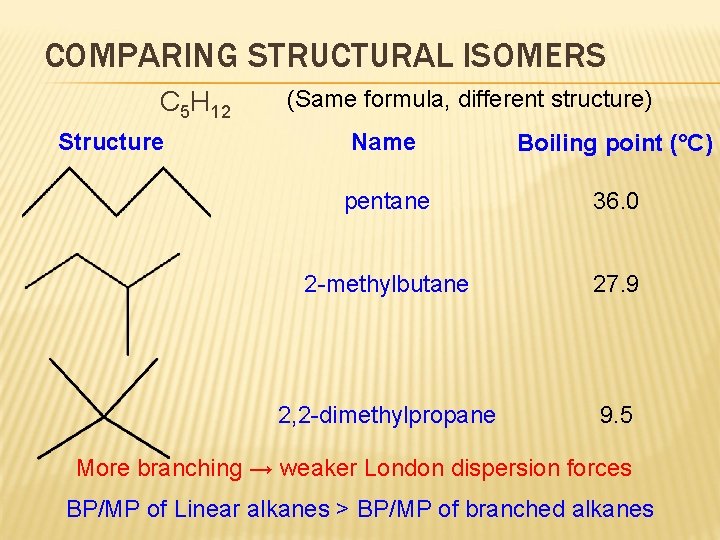
COMPARING STRUCTURAL ISOMERS C 5 H 12 Structure (Same formula, different structure) Name Boiling point (°C) pentane 36. 0 2 -methylbutane 27. 9 2, 2 -dimethylpropane 9. 5 More branching → weaker London dispersion forces BP/MP of Linear alkanes > BP/MP of branched alkanes
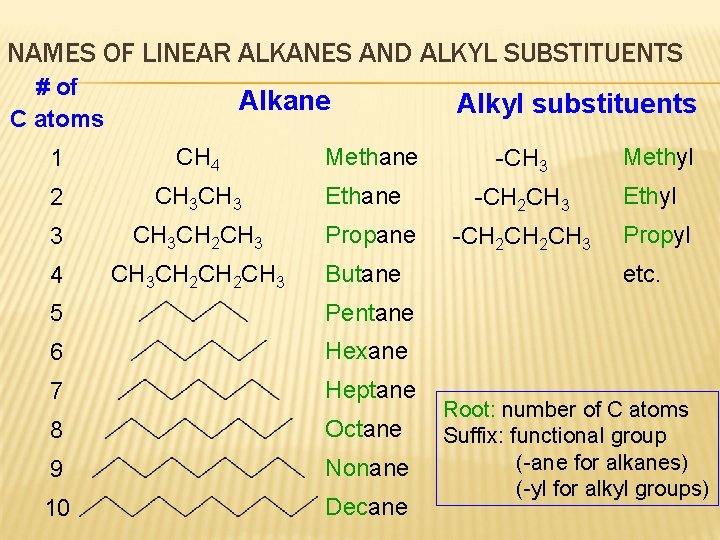
NAMES OF LINEAR ALKANES AND ALKYL SUBSTITUENTS # of C atoms Alkane 1 CH 4 2 CH 3 3 CH 3 CH 2 CH 3 4 CH 3 CH 2 CH 3 Methane Ethane Propane Butane 5 Pentane 6 Hexane 7 Heptane 8 Octane 9 Nonane 10 Decane Alkyl substituents -CH 3 -CH 2 CH 2 CH 3 Methyl Ethyl Propyl etc. Root: number of C atoms Suffix: functional group (-ane for alkanes) (-yl for alkyl groups)
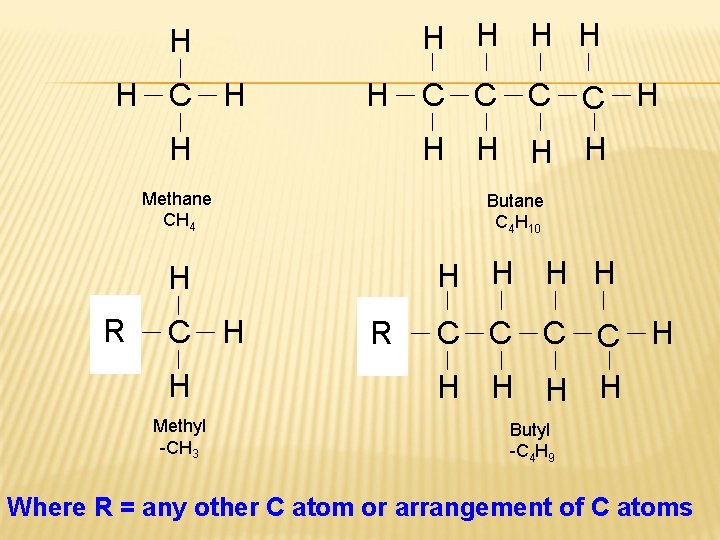
H H H C H H H H Methane CH 4 Butane C 4 H 10 H H H R? C C H C H R ? C C H H H Methyl -CH 3 Butyl -C 4 H 9 Where R = any other C atom or arrangement of C atoms
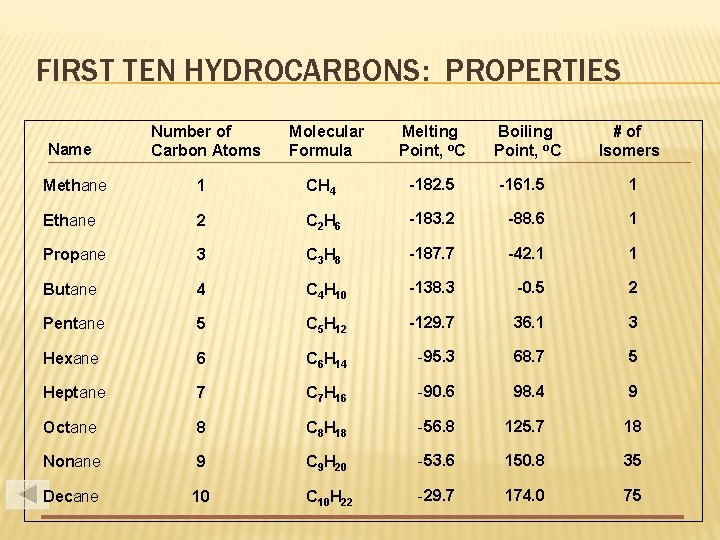
FIRST TEN HYDROCARBONS: PROPERTIES Name Number of Carbon Atoms Molecular Formula Melting Point, o. C Boiling Point, o. C # of Isomers Methane 1 CH 4 -182. 5 -161. 5 1 Ethane 2 C 2 H 6 -183. 2 -88. 6 1 Propane 3 C 3 H 8 -187. 7 -42. 1 1 Butane 4 C 4 H 10 -138. 3 -0. 5 2 Pentane 5 C 5 H 12 -129. 7 36. 1 3 Hexane 6 C 6 H 14 -95. 3 68. 7 5 Heptane 7 C 7 H 16 -90. 6 98. 4 9 Octane 8 C 8 H 18 -56. 8 125. 7 18 Nonane 9 C 9 H 20 -53. 6 150. 8 35 Decane 10 C 10 H 22 -29. 7 174. 0 75
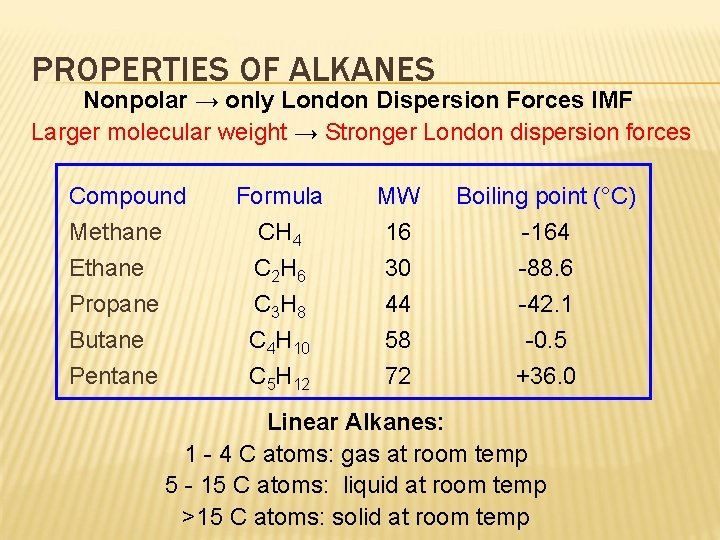
PROPERTIES OF ALKANES Nonpolar → only London Dispersion Forces IMF Larger molecular weight → Stronger London dispersion forces Compound Methane Ethane Propane Butane Pentane Formula CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 MW 16 30 44 58 72 Boiling point (°C) -164 -88. 6 -42. 1 -0. 5 +36. 0 Linear Alkanes: 1 - 4 C atoms: gas at room temp 5 - 15 C atoms: liquid at room temp >15 C atoms: solid at room temp
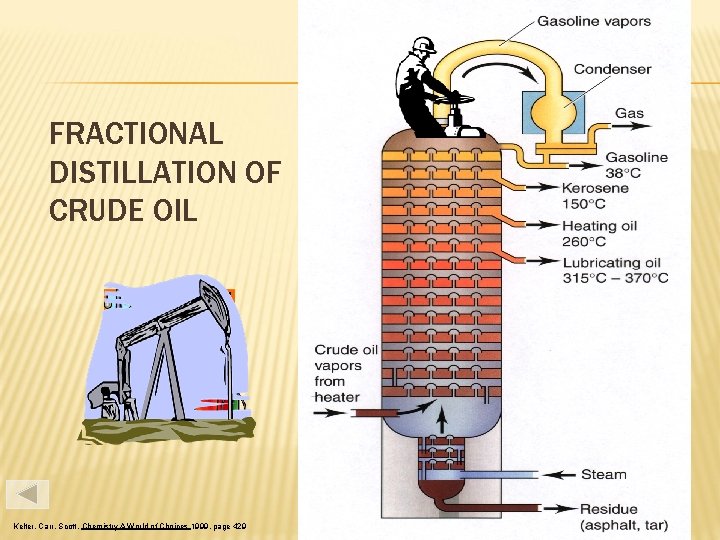
FRACTIONAL DISTILLATION OF CRUDE OIL Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 429
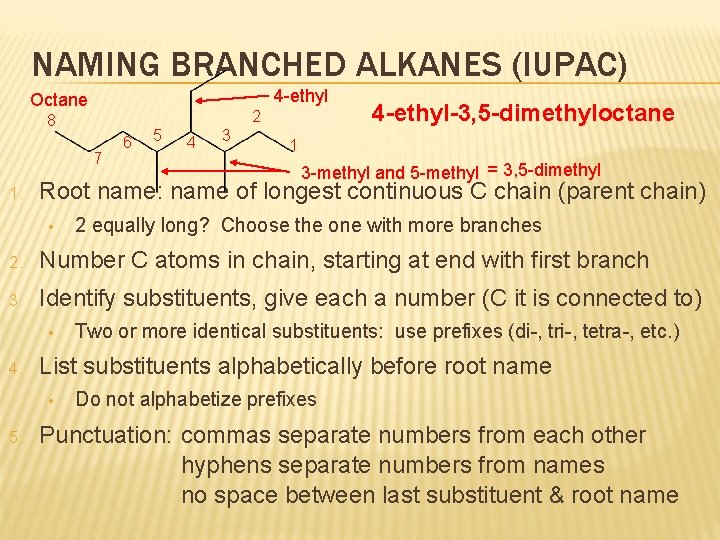
NAMING BRANCHED ALKANES (IUPAC) 4 -ethyl Octane 2 8 7 1. 6 5 4 3 4 -ethyl-3, 5 -dimethyloctane 1 3 -methyl and 5 -methyl = 3, 5 -dimethyl Root name: name of longest continuous C chain (parent chain) • 2 equally long? Choose the one with more branches 2. Number C atoms in chain, starting at end with first branch 3. Identify substituents, give each a number (C it is connected to) • 4. List substituents alphabetically before root name • 5. Two or more identical substituents: use prefixes (di-, tri-, tetra-, etc. ) Do not alphabetize prefixes Punctuation: commas separate numbers from each other hyphens separate numbers from names no space between last substituent & root name
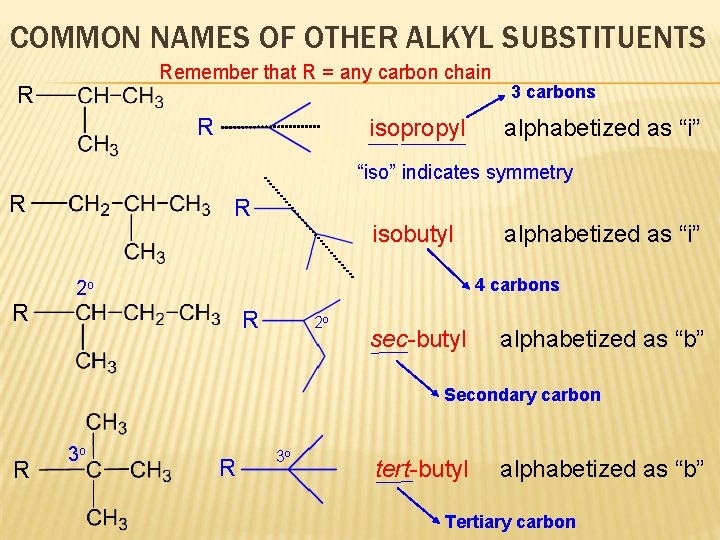
COMMON NAMES OF OTHER ALKYL SUBSTITUENTS Remember that R = any carbon chain R R isopropyl 3 carbons alphabetized as “i” “iso” indicates symmetry R R R isobutyl alphabetized as “i” 4 carbons 2 o R 2 o sec-butyl alphabetized as “b” Secondary carbon R 3 o tert-butyl alphabetized as “b” Tertiary carbon
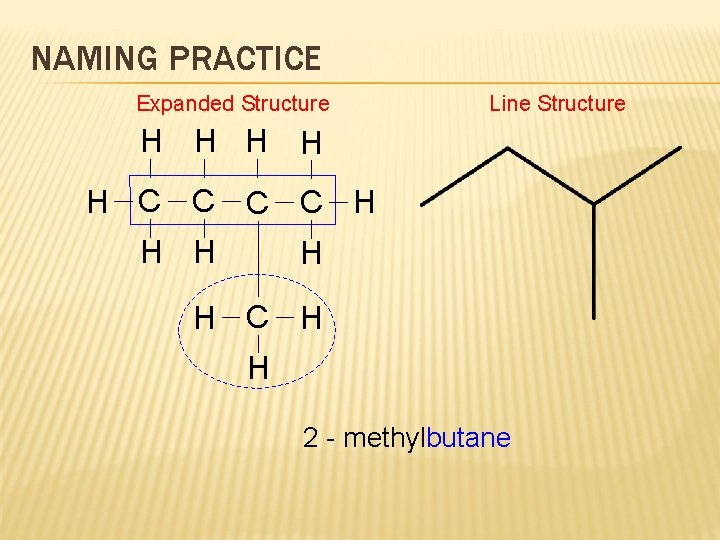
NAMING PRACTICE Expanded Structure H H H Line Structure H H C C H H H C H H butane 2 - methylbutane
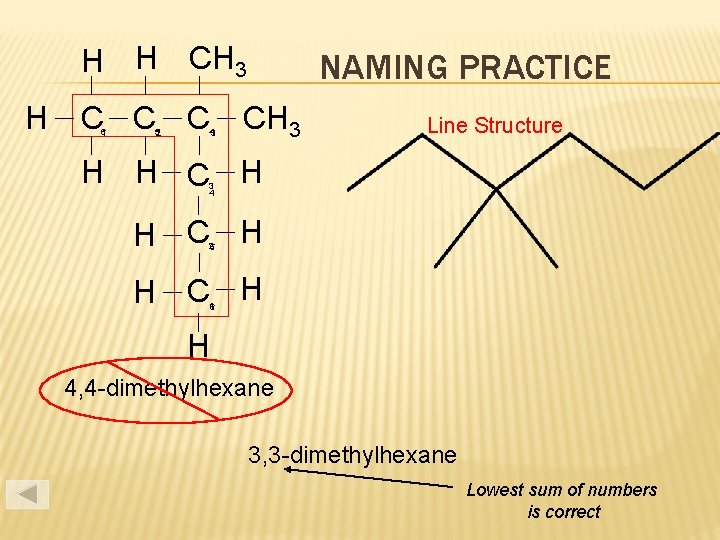
H H CH 3 H C C C 6 1 52 43 NAMING PRACTICE CH 3 Line Structure H H C H 3 4 H C H 25 H C H 16 H 4, 4 -dimethylhexane 3, 3 -dimethylhexane Lowest sum of numbers is correct
- Slides: 22