Chemistry of Carbon Chemistry of Carbon Carbon is

Chemistry of Carbon

Chemistry of Carbon � Carbon is the 4 th most abundant element in universe by mass after H, He, and O. � It is 15 th most abundant element in the earth’s crust. � It is the first element of group 14. � Atomic 2 P 2 No. 6, electronic configuration: 1 S 2 2 S 2

�It shows +4 oxidation state. �Its �It maximum covalency remains 4. has highest melting point (39300 C) �Boiling �Forms bond) point (4833 0 C) multiple bonds (double bond, triple

Isotopes of Carbon � C 12 : It is stable isotope and it is used as the standard for measurement of atomic masses of all other nuclides. ( 6 P, 6 N, 6 e-) � C 13 : It is also stable isotope and used in the metabolic process by mass spectroscopy. � It is used in identification of carbon in NMR spectroscopy. ( 6 P, 7 N, 6 e-)

� C 14 : It is radioactive isotope of carbon. 14 -β 14 C N 6 7 C 14 required for research is made by neutron bombardment on N 14 14 + n 1 14 + H 1 7 N 0 6 C 1 It is used as carbon dating. (bombardment of neutron on N- C 14 - reacts with O 2 - CO 2 plants-photosynthesis-glucose sugar- used by plants – starch, protein cellulose- animal-decreases after death-careful measuring the radioactive decay rate- possible to calculate how long plant/ died-dating plant/animal origin)

Allotropes of Carbon � Carbon is stabilized by forming different multiatomic structures having different molecular configuration called allotropes of carbon.

• Diamond • Graphite Crystalline Carbon Cluster • Fullerene • Carbon nanotubes • Graphene Amorphou • Carbon Black s/ partially • Activated Carbon crystalline • Carbon Fibers

Diamond: Occurrence v Diamonds occurs in the form of rounded pebbles in South Africa, Brazil, Australia & India. v Large v 80% Diamond cut as gemstones. of mined diamonds has less clarity, color, etc. & are used for industrial purpose for cutting, polishing etc.
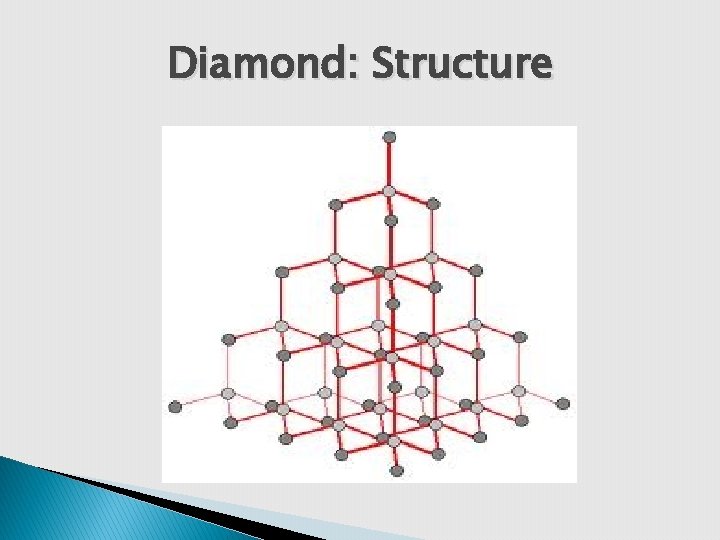
Diamond: Structure

Diamond: Structure 1. It is crystalline structure. three dimensional 2. Large numbers of carbon arranged in tetrahedral manner (sp 3 hybridization). 3. C-C bond length is 1. 54 A 0. 4. Due to high strength of covalent bonds, it is very hard & has high density.

Diamond: Synthesis 1. q q High pressure, High temperature method: Synthesized by using graphite & transition metal like Ni heated to 1500 – 2000 K & at 7 Gpa pressure Synthesized by using graphite without metal catalyst if the temperature & pressure are high enough.

Diamond: Synthesis 2. Chemical vapour deposition method The method involves feeding a mixture of gases (1: 99, methane & hydrogen) into a chamber & splitting them to chemically active radicals in plasma ignited by laser or hot filament & under a low pressure.

Diamond: Properties � Hardest solid � M. P. 39300 C � Density = 3. 51 gm/cc � High refractive index � Bad conductor of electricity � Good conductor of heat � Diamond is colorless but industrial diamonds are black (N- yellow, Al- blue) � Burns in air at 600 -8000 C

Diamond: Applications 1. Because of its durability, clarity & high refractive index diamond is used as highly priced gemstone, jwellery. 2. Diamond can be used for industrial purposed in cutting, drilling, grinding, polishing tools because of high hardness. 3. In optical instruments

Graphite: Occurrence § § § Occurs in China, India, Brazil, North Korea, Canada. Usually found as a mixture with mica, quartz & silicates. Graphite is evaporated from most of the impurities by floatation. It is purified by heating with HCl & HF in vacuum to remove trace of silicon compound of Si. F 4. It is occur in metamorphic rock.

Graphite: Structure § Crystalline allotropes of carbon. § Each carbon atom is sp 2 hybridized § It produced hexagonal rings (formed by joining of each sp 2 carbon to three other sp 2 carbons by σbonds) connected together to form two dimensional large plane sheet with network of single & double bond. § The sheet are held together by weak Van der Waal’s forces. § C – C bond length is 1. 42 A 0. The distance between two successive layers is 3. 36 A 0


Graphite q It is a good conductor of electricity – the 4 th valency of each carbon remains unsatisfied (remains unpaired or free). q This free electron can easily move from one carbon to another under the influence of potential. q Conductivity can occur in sheet but not from one sheet to another.

Graphite: Properties � Density= � Very 2. 22 gm/cc. soft (due to easy sliding of weakly bonded planes of carbon atoms). � Higher � Due chemical reactivity (C=C) than diamond. to unsatisfied valency of carbon, graphite has very good adsorption ability. � Due to π e- clouds on both sides of planes, it is good conductor of electricity. � Graphite sheet nanotubes. on folding, used as carbon

Graphite: Applications 1. As a lubricant. 2. As a electrode in all major batteries. 3. It is used as refractory material. 4. Graphite crucible are used to hold molten metal. 5. It is used in making lead pencils. 6. Making carbon fibres.

Comparison of Diamond and Graphite Diamond � Each carbon Graphite is sp 3 � Each carbon is sp 2 hybridized. � Each carbon is covalently hybridized. � Each carbon is covalently bonded with three carbons. bonded with four carbons. � � � The density is 2. 22 gm/cc. � Good conductor of electricity. The density is 3. 51 gm/cc. Bad conductor of electricity but good conductor of heat.

Comparison of Diamond and Graphite Diamond � 3 -D arranged Graphite carbon � atoms. � Hardest substance, useful Thermodynamically � Chemically non-reactive, transparent colorless. carbon Soft, used as lubricant and for electrodes. � lesser stable. � arranged atoms. cutting & drilling. � 2 -D Thermodynamically more stable. � Chemically more reactive and black colored.

Graphene

Fullerenes � Fullerenes are carbon clusters allotropes. � They consist of hollow spherical cluster of large number of carbon atom. � The most common fullerenes is C 60 molecules, known as Buckminister fullerene.

Fullerenes: Structure Ø zero dimensional allotrope of carbon. Ø carbon atoms arranged in a series of pentagon & hexagon rings to form structure of football. The diameter of ball is 0. 7 nm Ø C 60 consist of 20 hexagonal faces and 12 pentagonal faces Ø 60 C atoms are sp 2 hybridized. There are 30 double bonds in C 60.

Fullerenes: Structure ØC – C bond length is 1. 45 A 0 & C = C bond length is 1. 38 A 0. Ø The π electrons of double bonds are delocalized. Ø Show reaction such as electrophilic addition reaction, Diels-alder reaction, reaction with alkali metal etc.
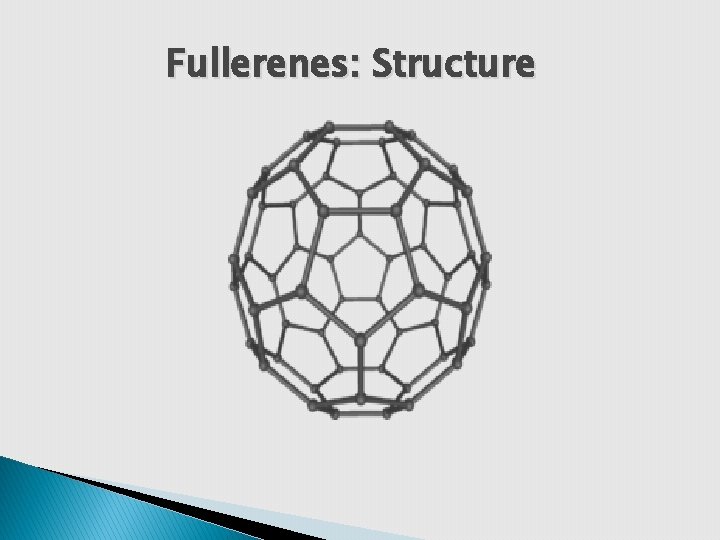
Fullerenes: Structure

Fullerenes: Properties v Semiconductor but alkali metal compound of formula M 3 C 60 are superconductors. v C 60 is mustered coloured and looks brown and black as thickness increases. v Due to cage structure, ions can be trapped or functional group can be attached. v Fullerene shows poor aromatic nature (as pentagonal rings have tendency to avoid double bonds)

Fullerenes: Applications 1) Electrical conductivity is low. It is increased by doping with alkali metals, then it is used as a superconductor. 2) It is used as a lubricant. 3) C 60 incorporated with alkali metal can act as catalyst for chemical reaction. 4) C 60 molecules can bind large number of hydrogen atoms. So it can be act as medium for hydrogen storage.

Carbon Nanotubes (CNT) q q 1) 2) CNTs can be imagined as cylinders formed by graphene sheet (single sheet of graphene) & capping by hemisphere structure similar to fullerene hemisphere (half of fullerene including pentagon & hexagon) Types of Nanotubes Single walled Nanotubes (SWNT) Multi-walled Nanotubes (MWNT)

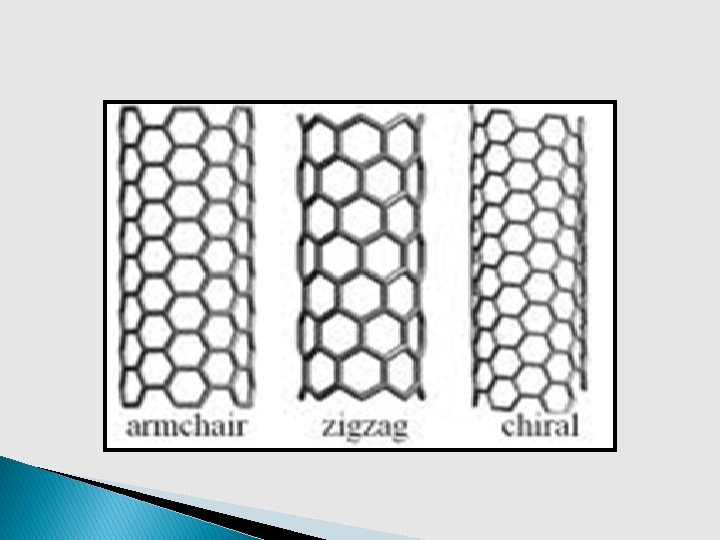
1) SWNT: Structure § SWNT can be wrapping a single atom thick layer of graphene in to a cylinder. § Three types of SWNT - zigzag, armchair & chiral can be imagined by three different ways of wrapping graphene sheet.

§ Bonding in carbon nanotubes is sp 2 § Armchair SWNT show metallic conductivity while other two are semiconductor. § CNTs are stiffer than steel. They have high tensile strength & high damage from physical force. resistance to
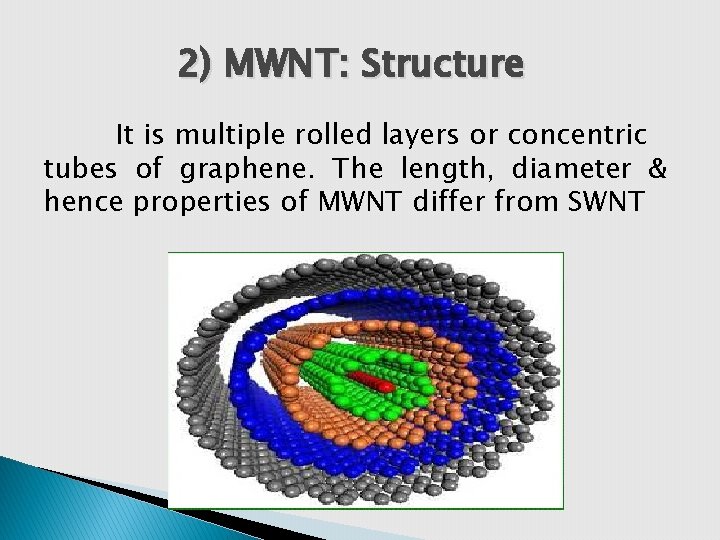
2) MWNT: Structure It is multiple rolled layers or concentric tubes of graphene. The length, diameter & hence properties of MWNT differ from SWNT

CNT – Preparation 1) Pyrolysis of hydrocarbons: • Pyrolysis of acetylene at 700 o. C in the presence of Fe-graphite under inert condition yield CNT. 2) Arc vaporization of Graphite • It is carried out by striking a direct current arc between the graphite electrodes separated by about 1 mm in inert atmosphere to give MWNT.

CNT – Preparation 3) Laser ablation or evaporation: • A pulsed laser vaporizes a graphite target containing small amount of Co & Ni in a high temperature (1200 degree C) quartz tube reactor in inert atmosphere to give SWNT. 4) Chemical vapor deposition (CVD): • Decomposition of hydrocarbon (methane, acetylene, ethylene) at 1100 o. C in the presence of metal catalyst (Ni, Co, Fe supported on Mg. O/Al 2 O 3). Carbon atoms formed then condense on cooler surface to form CNT.

Carbon Nanotubes: Applications 1. Hydrogen storage material – in fuel cell as CNT can hold 7. 7% hydrogen by weight by physical adsorption. 2. Batteries – used in lithium batteries. 3. Sensors – On exposure to environment, which contain NO 2, NH 3 or O 2, the electric resistance changes.

Carbon Nanotubes: Applications 4. Composite material – Because of high stiffness, CNT can be used as reinforcement in high strength, low weight composite. 5. drug delivery vessel – used for drug delivery within the body by placing the drug within the tube or by attaching the drug to the side of the tube.
- Slides: 38