Organic Chemistry The Chemistry of Carbon Compounds Carbon

Organic Chemistry The Chemistry of Carbon Compounds

Carbon
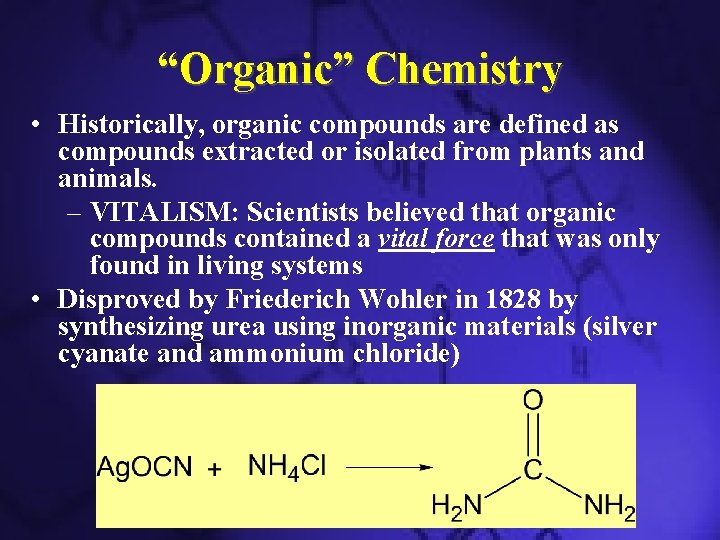
“Organic” Chemistry • Historically, organic compounds are defined as compounds extracted or isolated from plants and animals. – VITALISM: Scientists believed that organic compounds contained a vital force that was only found in living systems • Disproved by Friederich Wohler in 1828 by synthesizing urea using inorganic materials (silver cyanate and ammonium chloride)

Friederich Wohler

Organic Chemistry • Study of carbon compounds – Composed of carbon, hydrogen, oxygen, nitrogen, sulfur, phosphorus, and the halogens • More than 95% of known compounds are organic compounds (Carbon-containing) • Recall – Carbons can have 4 bonds – Nitrogen can have 3 bonds – Oxygen can have 2 bonds – Hydrogen can have 1 bond
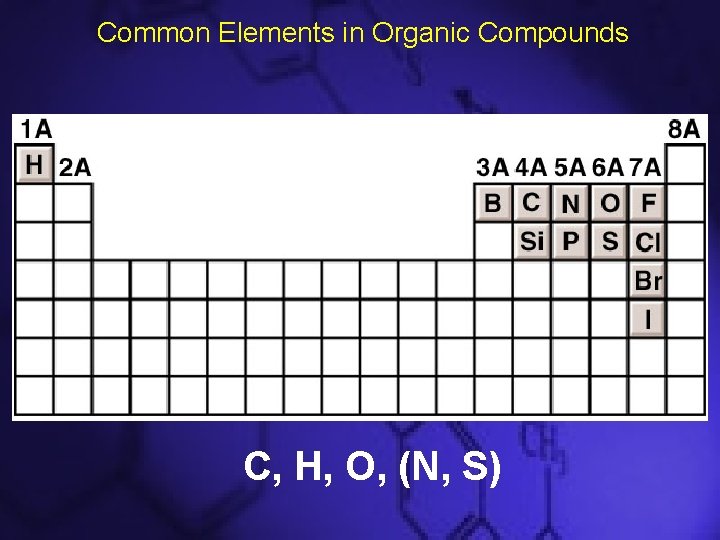
Common Elements in Organic Compounds C, H, O, (N, S)

The Secret to Organic Compounds: the unique carbon atom • CATENATION – Ability of C-atoms to bond to each other so strongly, they can form long chains. Eg. Thousands of C-atoms bonded. – Each carbon can have four bonds, maximum

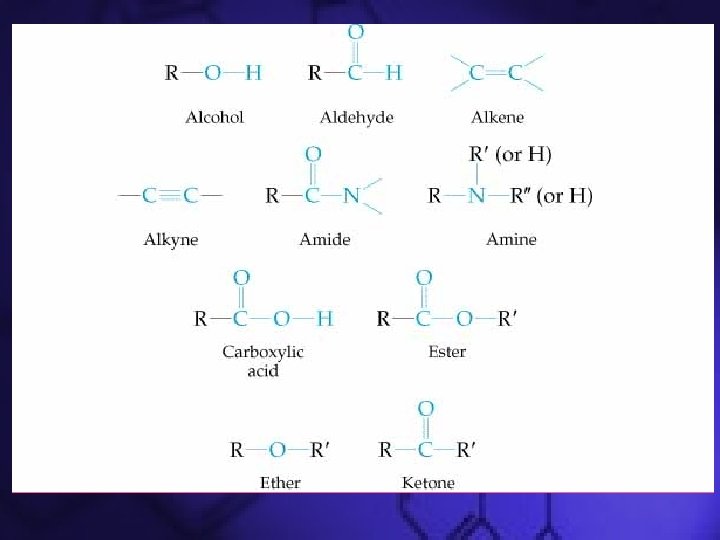
Common organic compound families • HYDROCARBONS: Only C and H – Alkanes, alkenes, alkynes, arenes, aromatic compounds • OXYGEN-CONTAINING – Alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, acid anhydrides • NITROGEN-CONTAINING – Amines, Amides
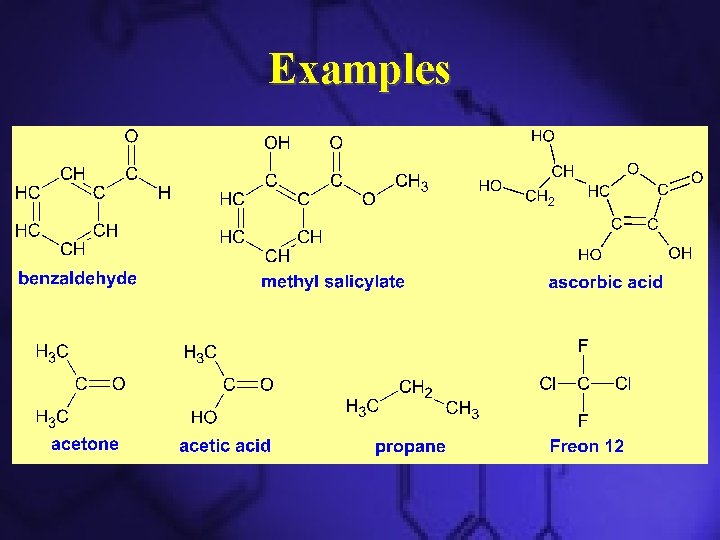
Examples
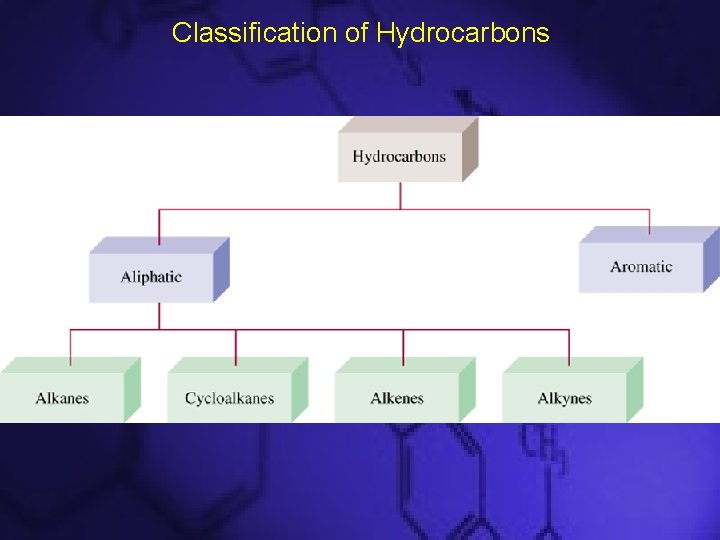
Classification of Hydrocarbons
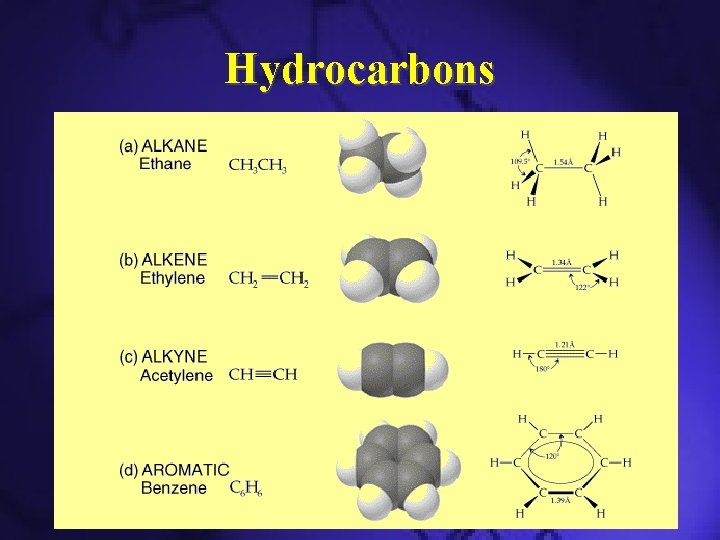
Hydrocarbons

Hydrocarbons: Alkanes • • Simplest hydrocarbons Contains only C–C and C–H bonds Usually used as fuels Three types – Straight chain alkanes – Branched alkanes – Cyclic alkanes

Examples of Alkanes
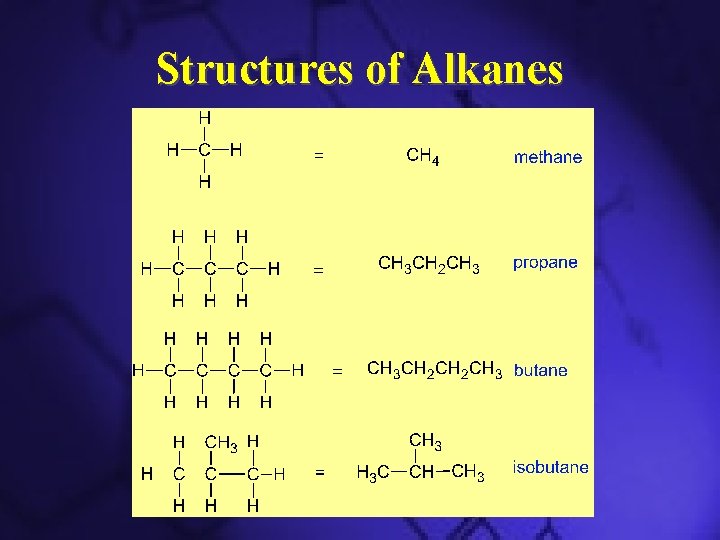
Structures of Alkanes
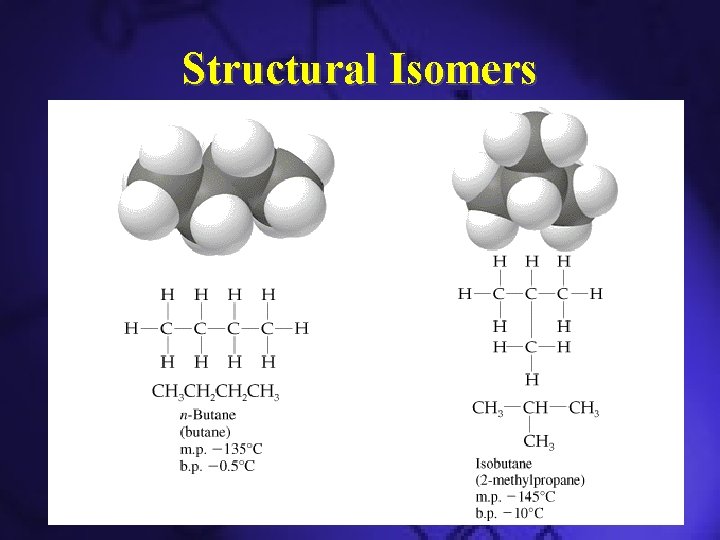
Structural Isomers
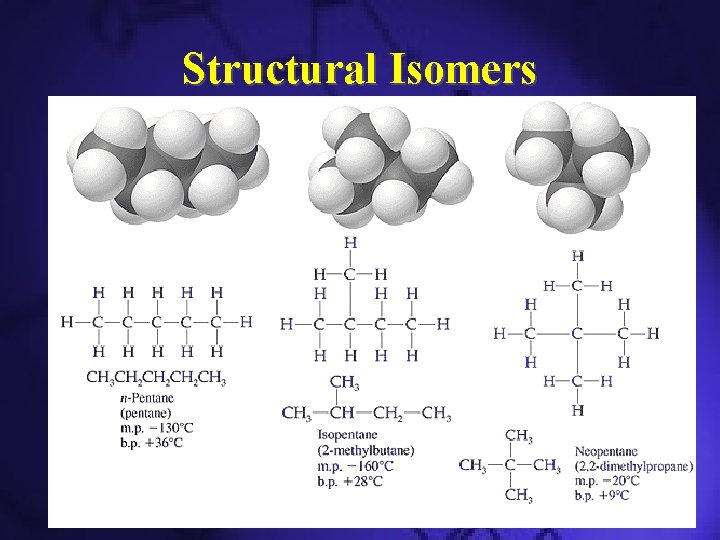
Structural Isomers
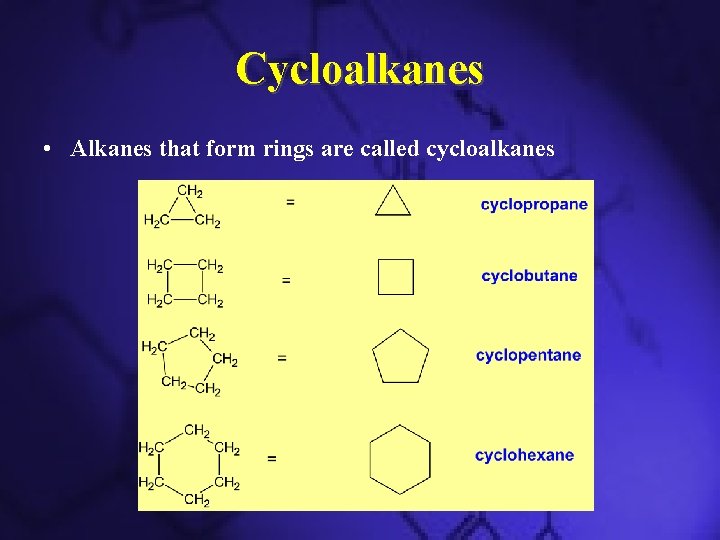
Cycloalkanes • Alkanes that form rings are called cycloalkanes
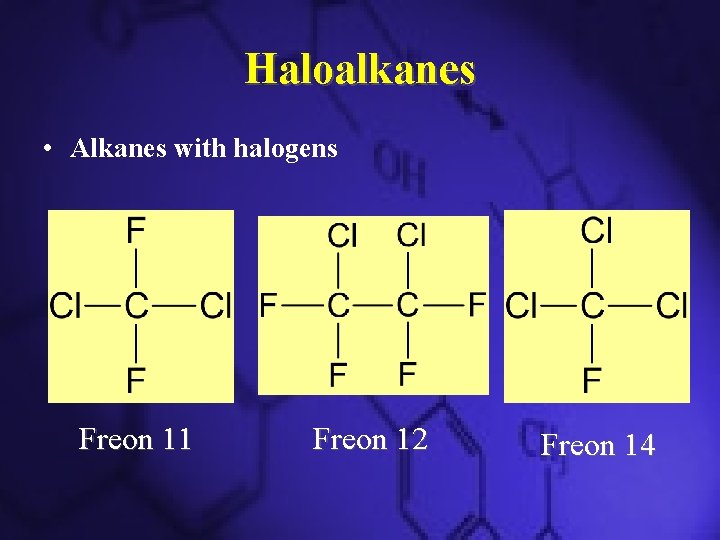
Haloalkanes • Alkanes with halogens Freon 11 Freon 12 Freon 14

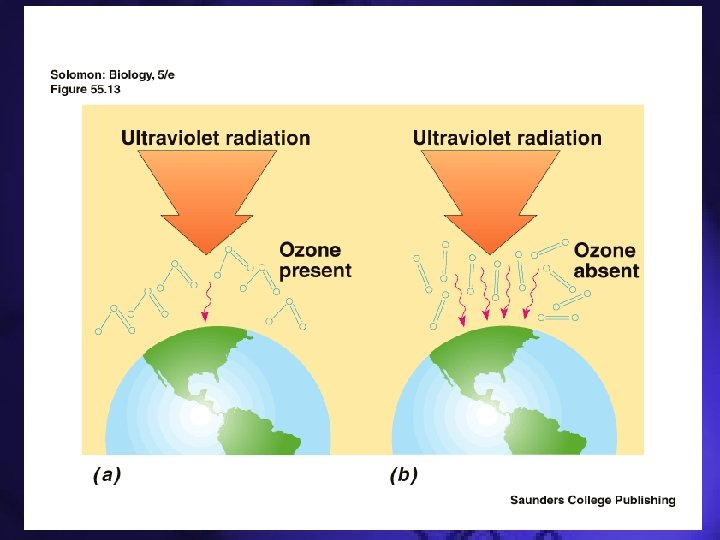
Chlorofluorocarbons • Insoluble in water and are unreactive towards substances • Unreactiveness lets them reach the stratosphere and react with the ozone layer
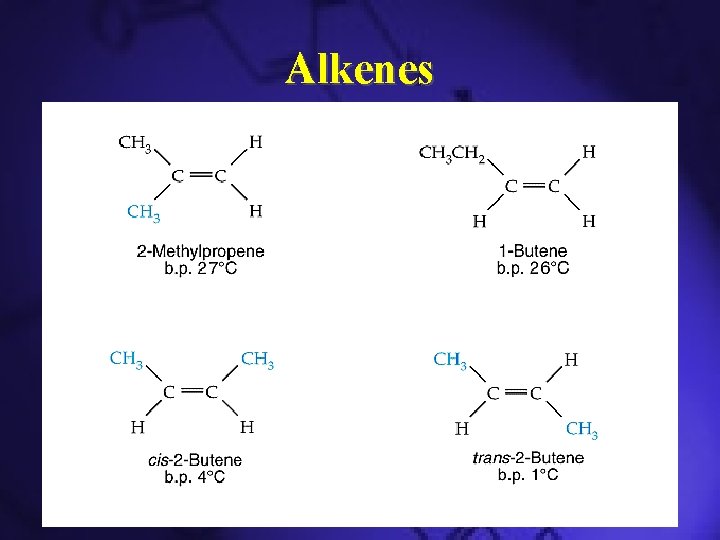
Alkenes
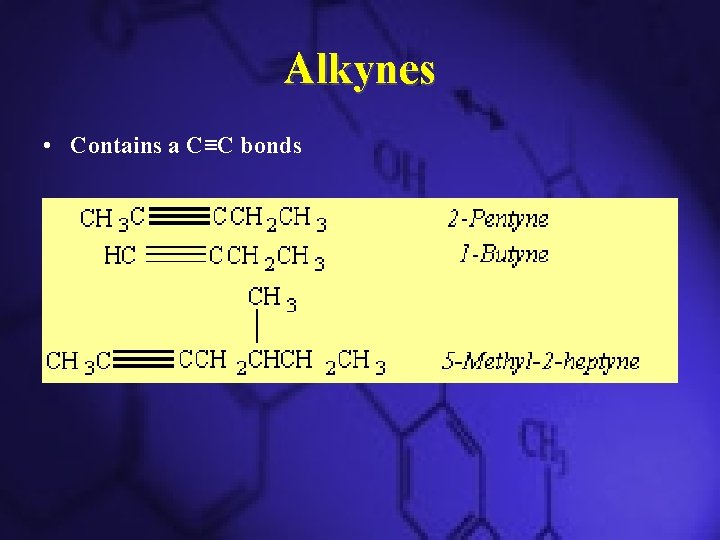
Alkynes • Contains a C≡C bonds

August Kekule and the Dream • C 6 H 6

• • "I fell into a reverie, and lo, the atoms were gamboling before my eyes! Whenever, hitherto, these diminutive beings had appeared to me, they had always been in motion; but up to that time, I had never been able to discern the nature of their motion. Now, however, I saw how, frequently, two smaller atoms united to form a pair; how a larger one embraced the two smaller ones; how still larger ones kept hold of three or even four of the smaller; whilst the whole kept whirling in a giddy dance. I saw how the larger ones formed a chain, dragging the smaller ones after them, but only at the ends of the chain. . . The cry of the conductor: “Clapham Road, ” awakened me from my dreaming; but I spent part of the night in putting on paper at least sketches of these dream forms. This was the origin of the Structural Theory. " ". . . I was sitting writing on my textbook, but the work did not progress; my thoughts were elsewhere. I turned my chair to the fire and dozed. Again the atoms were gamboling before my eyes. This time the smaller groups kept modestly in the background. My mental eye, rendered more acute by the repeated visions of the kind, could now distinguish larger structures of manifold conformation; long rows sometimes more closely fitted together all twining and twisting in snake-like motion. But look! What was that? One of the snakes had seized hold of its own tail, and the form whirled mockingly before my eyes. As if by a flash of lightning I awoke; and this time also I spent the rest of the night in working out the consequences of the hypothesis. "
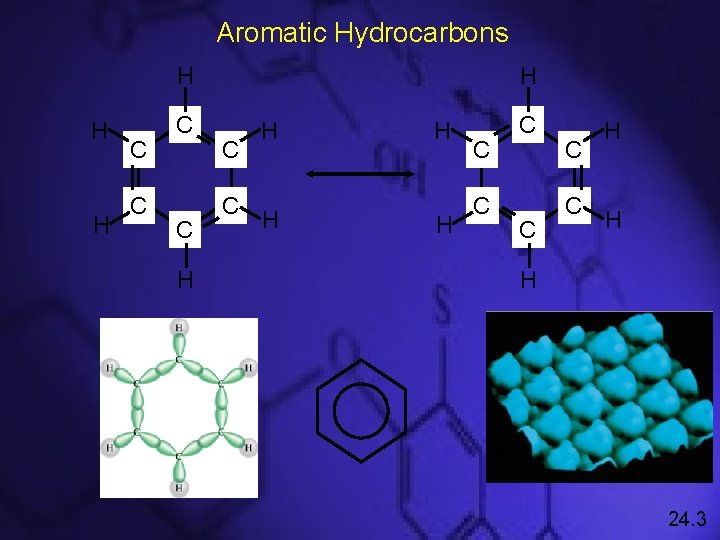
Aromatic Hydrocarbons H H C C H H C C C H H H 24. 3
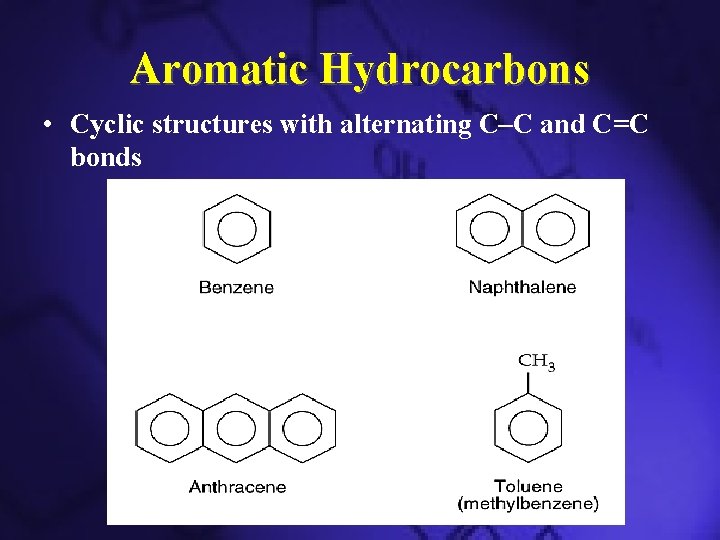
Aromatic Hydrocarbons • Cyclic structures with alternating C–C and C=C bonds

Polycyclic Aromatic Hydrocarbons 24. 3

Functional Groups • Determines the properties of the organic compound – Compounds with the same functional group will react similarly • Alkenes: –C=C– bond • Alkynes: –C≡C– bond • Aromatic hydrocarbons: cyclic structure with alternating –C–C– and –C=C– bonds

Alcohols • has the general structure R–OH – derived from hydrocarbons and contain -OH groups • Polar molecules and are soluble in water (Why? )

Some Common Alcohols • CH 3 OH (methanol) – Used as an industrial solvent – Possible replacement for gasoline in automobiles • CH 3 CH 2 OH (ethanol) – Made from fermentation of grain or other sugar materials – Made by reaction of ethylene with water (denatured alcohol) – Used for beverages and fuels
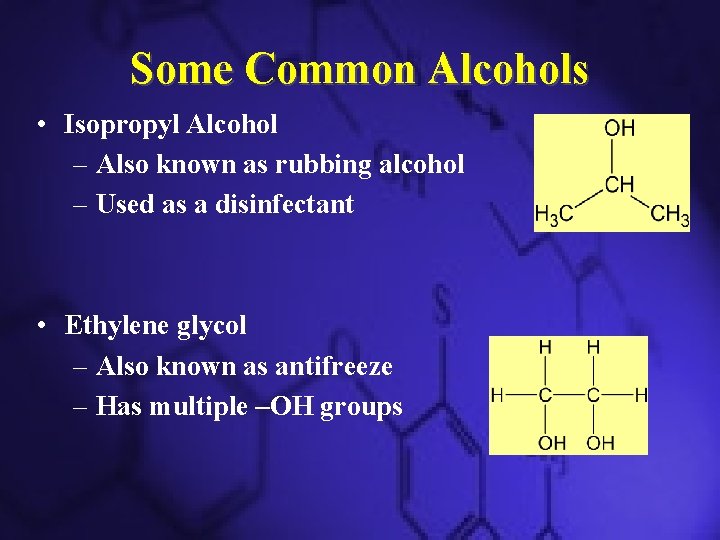
Some Common Alcohols • Isopropyl Alcohol – Also known as rubbing alcohol – Used as a disinfectant • Ethylene glycol – Also known as antifreeze – Has multiple –OH groups
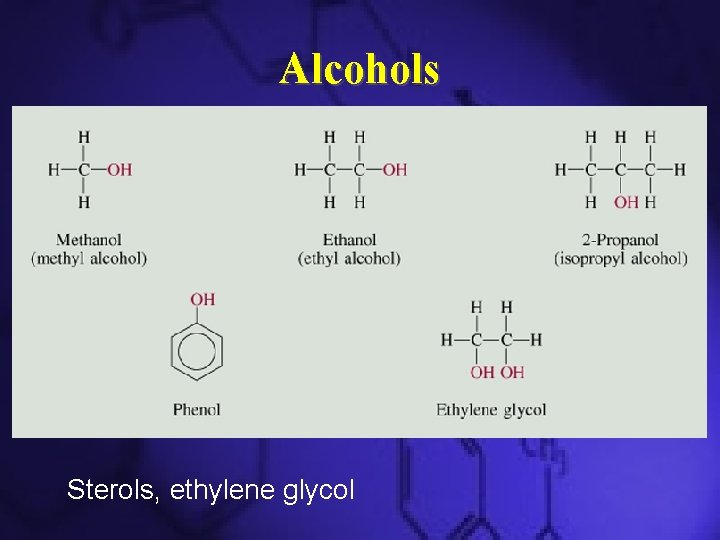
Alcohols Sterols, ethylene glycol
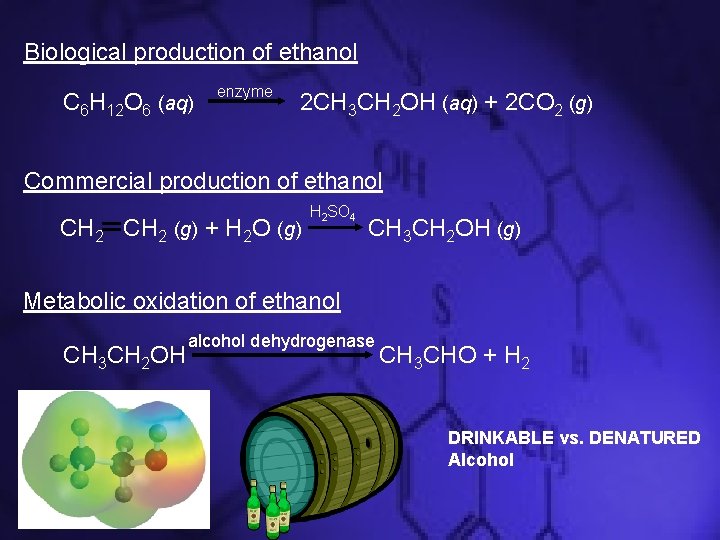
Biological production of ethanol C 6 H 12 O 6 (aq) enzyme 2 CH 3 CH 2 OH (aq) + 2 CO 2 (g) Commercial production of ethanol CH 2 (g) + H 2 O (g) H 2 SO 4 CH 3 CH 2 OH (g) Metabolic oxidation of ethanol CH 3 CH 2 OH alcohol dehydrogenase CH 3 CHO + H 2 DRINKABLE vs. DENATURED Alcohol

Ethers • Has the R-O-R’ general structure • Compounds in which two hydrocarbons linked by an oxygen are called ethers. • Used as general anesthetic • Used as solvents • Less soluble in water than alcohols
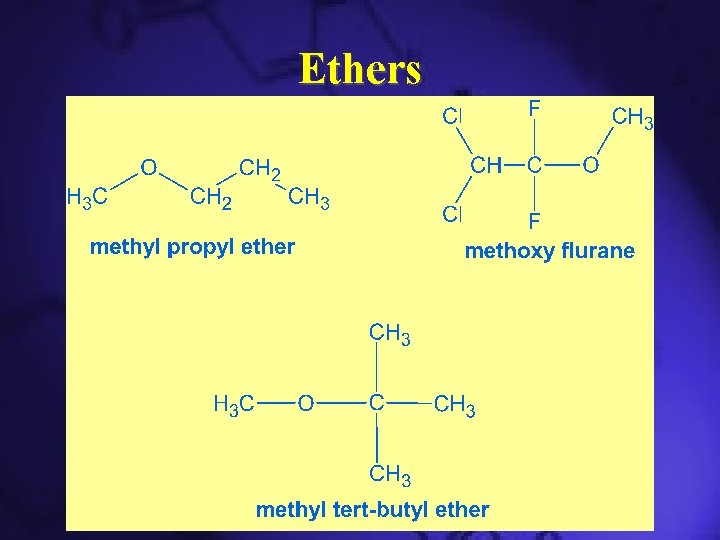
Ethers

Compounds with a Carbonyl Group • Carbonyl functional group is C=O • Types of different carbonyl compounds – Aldehydes – Ketones – Carboxylic Acids – Esters – Amides
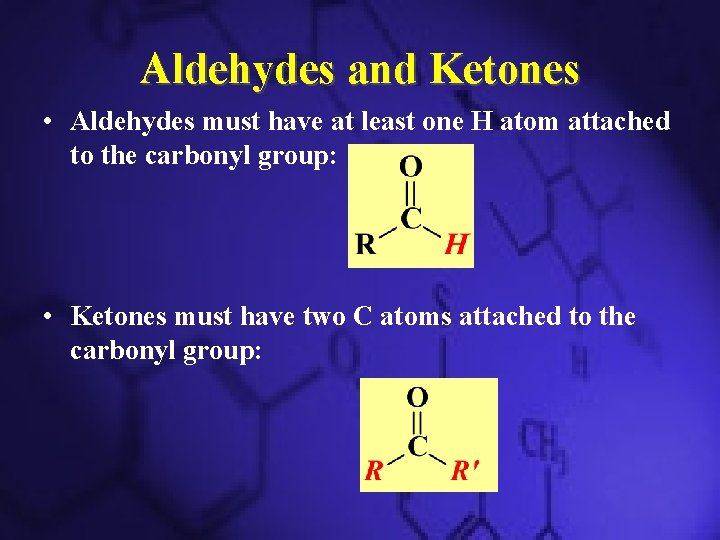
Aldehydes and Ketones • Aldehydes must have at least one H atom attached to the carbonyl group: • Ketones must have two C atoms attached to the carbonyl group:
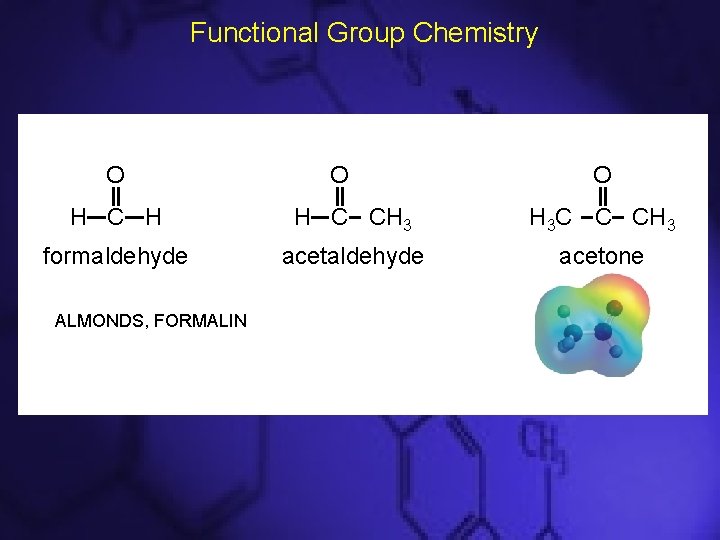
Functional Group Chemistry O O O H C H H C CH 3 H 3 C C CH 3 formaldehyde acetone ALMONDS, FORMALIN
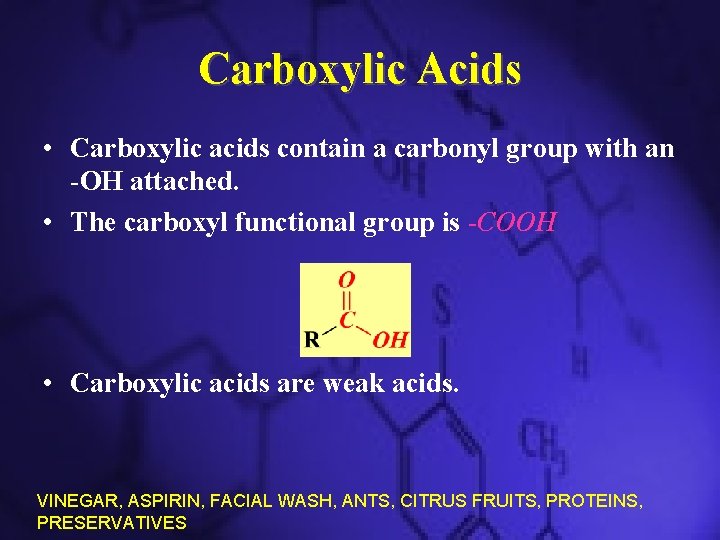
Carboxylic Acids • Carboxylic acids contain a carbonyl group with an -OH attached. • The carboxyl functional group is -COOH • Carboxylic acids are weak acids. VINEGAR, ASPIRIN, FACIAL WASH, ANTS, CITRUS FRUITS, PROTEINS, PRESERVATIVES
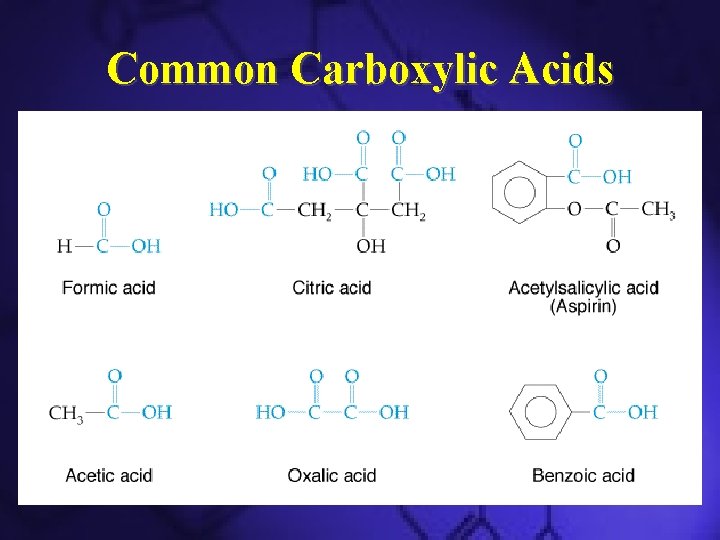
Common Carboxylic Acids

Esters • Esters contain -COOR groups: • Usually have fruity odors and tastes
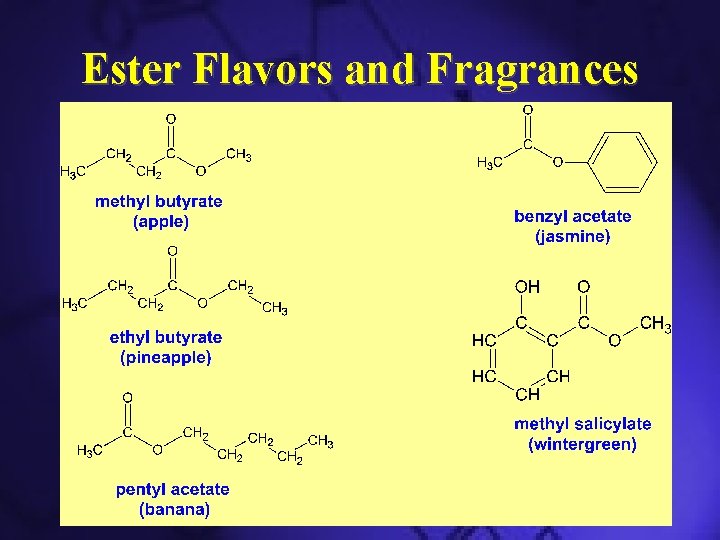
Ester Flavors and Fragrances

Functional Group Chemistry Esters have the general formula R’COOR, where R is a hydrocarbon group. O CH 3 COOH + HOCH 2 CH 3 C O CH 2 CH 3 + H 2 O ethyl acetate Banana: isopentyl acetate Pineapple: ethyl butanoate Apple: methyl butanoate Pear: propyl ethanoate Orange: Octyl ethanoate Raspberry: 2 -Methylpropyl ethanoate Oil of Wintergreen: methyl salycilate

Esters Galore! allyl caproate pineapple amyl acetate apple, banana amyl butyrate apricot, pear, ethyl pelargonate pineapple geranyl acetate amyl caproate apple, pineapple geranyl butyrate amyl valerate apple geranyl valerate benzyl acetate pear, strawberry bornyl acetate pine tree flavor linalyl acetate linalyl butyrate iso-butyl acetate cherry, raspberry, linalyl formate strawberry menthyl acetate ethyl acetate peach, pineapple, methyl benzyl acetate raspberry methyl cinnamate ethyl butyrate banana, pineapple, strawberry methyl phenyl acetate ethyl caproate strawberry ethyl cinnamate cinnamon methyl salicylate ethyl formate lemon, strawberry methyl anthranilate ethyl heptoate grape, pineapple nonyl caprylate ethyl isovalerate apple octyl butyrate ethyl heptanoate apricot, cherry, grape, raspberry terpenyl butyrate ethyl lactate grape geranium cherry apple lavender, sage peach apple, peach peppermint cherry strawberry honey wintergreen grape, jasmine orange parsnip cherry

HOW TO MIMIC NATURE: Making synthetic flavoring • • A good cherry is supposed to be tough to formulate. Here is an example of a *minimum* synthetic cherry flavoring: ethyl methyl p-tolyl glycidate 16. 0 % iso-amyl acetate 12. 0 % iso-butyl acetate 12. 0 % p-methyl benzyl acetate 11. 0 % benzaldehyde 8. 0 % vanillin 7. 0 % benzyl alkcohol 5. 5 % piperonal 5. 0 % ethyl caprate 4. 0 % cinnamic aldehyde dimethyl acetal 3. 0 % p-tolyl aldehyde 3. 0 % cinnamyl anthranilate 3. 0 % ethyl caproate 2. 0 % geranyl butyrate 2. 0 % terpenyl butyrate 0. 5 %
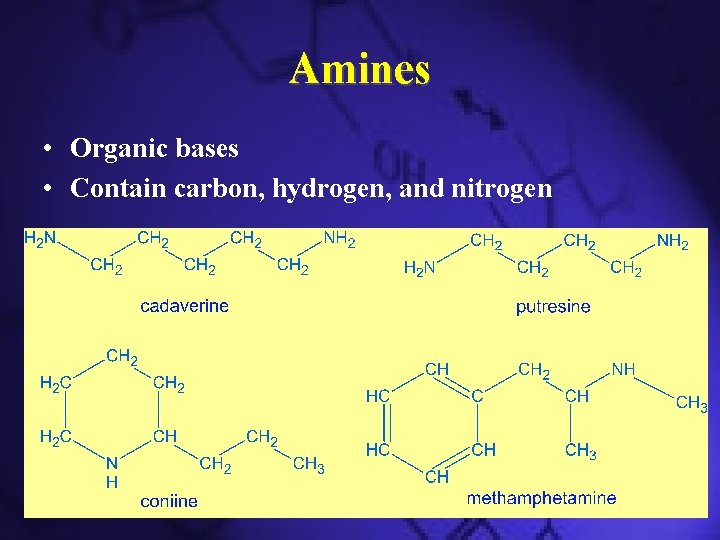
Amines • Organic bases • Contain carbon, hydrogen, and nitrogen
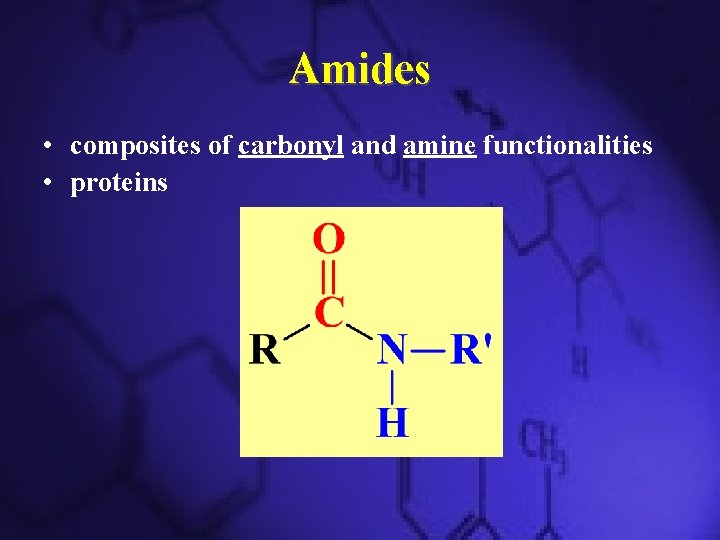
Amides • composites of carbonyl and amine functionalities • proteins
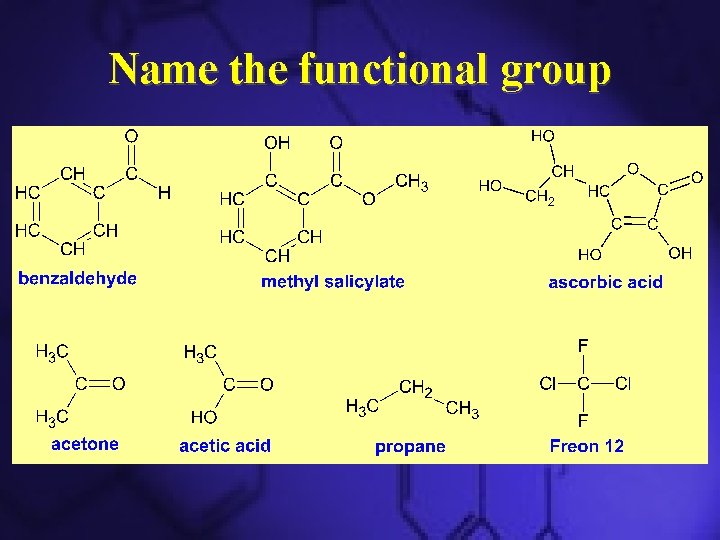
Name the functional group

Compounds with multiple functional groups

Mango

Would you eat this? a-Terpinolene, Ethyl butanoate, 3 -Carene, Ethyl acetate, Ethyl 2 -butenoate, a-Terpinene, a-Thujene, Dimethyl sulfide, Limonene, b-Phellandrene, Myrcene, p-Cymen -8 -ol, b-Caryophyllene, cis-3 -Hexene-1 -ol, hexadecyl acetate, 5 -Butyldihydro-3 H-2 -furanone, trans-2 hexenal, Ethyl tetradeconaoate, a-Humulene, sabinene, 2 -Carene, Camphene, Ethyl octanoate, 4 Isopropenyl-1 -methylbenzene 1 -Hexanol, a-terpinene, hexanal, Ethyl hexadecanoate, a-Copaene, Hexadecanal, Ethanol, Ethyl propionate, Dihydro-5 hexyl-3 H-2 -furanone, Carveol, Geranial, Ethyl decanoate, Furfural, Butyl acetate, Methyl butanoate, 2, 3, Pentanedione, 1, 1, diethoxyethane, pentadecanal, Butyl formate, 1 -Butanol, 5 -Methylfurfural, Ethyl dodecanoate, 2 -Acetylfuran, 2 Methyl-1 -butanol, 4 Methylacetophenoen, Acetaldehyde, Cyclohexane

FOSSIL FUELS • Coal, Petroleum, Natural Gas • From decayed organic matter • Makes up 90% of energy used

Coal • Main element is carbon, but contains small% of other elements • Complete combustion: – C(s) + O 2(g) CO 2(g) • Limited supply, strip mining

Natural Gas • Principally methane • Formed ages ago probably by heat, pressure, and action of bacteria on organic matter.

Chemistry In Action: The Petroleum Industry Crude Oil

The Guimaras Oil Spill

Gasoline: C 5 -C 12 alkanes • C 5 -C 12 alkanes, some sulfur and nitrogen-containing hydrocarbons • >C 15, difficult to combust, causes knock • OCTANE Rating: Isooctane (CH 3)3 CCH 2 CH(CH 3)2 = 100 Heptane = 0 • Some cpds improving octane rating: tetraethyllead Diesel • Runs on engine designed by Engineer Rudolf Diesel • Petrodiesel: It is a hydrocarbon mixture, (C 10 -C 15) obtained in the fractional distillation of crude oil between 250 °C and 350 °C. • Biodiesel: Methyl esters of fats from natural oils
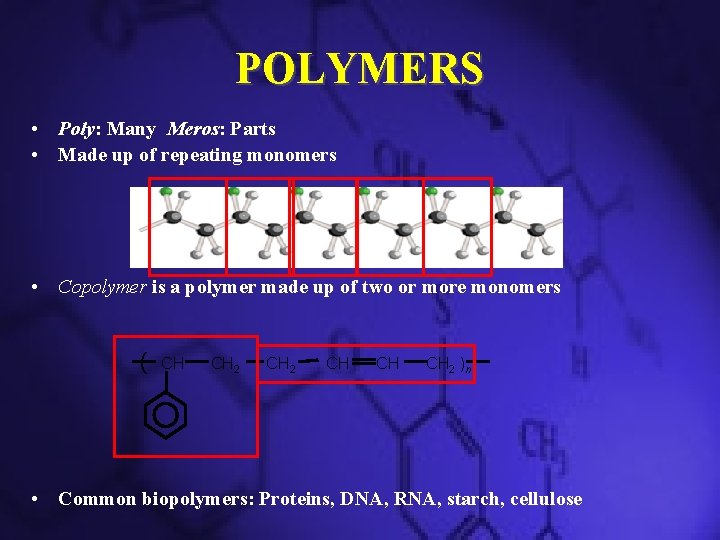
POLYMERS • Poly: Many Meros: Parts • Made up of repeating monomers • Copolymer is a polymer made up of two or more monomers ( CH CH 2 CH CH CH 2 )n • Common biopolymers: Proteins, DNA, RNA, starch, cellulose

Some commercial polymers

• PET: Polyethylene terephthalate – Plastic bottles • Polytetrafluoroethylene – Teflon™ • Polypropylene • Polystyrene • Polyvinyl chloride • Nylon (synthetic polyamide, similar to bonds in proteins) http: //www. nationalgeographic. com/education/plastics/index. html

References: q Hill, JW and Kolb DK. Chemistry for the Changing Times, 7 th ed. Prentice Hall International. 1992. q Brown, TL. Le May, Jr. , HE, and Bursten, BE. Chemistry: The Central Science. 1997. q Schmid, G. Organic Chemistry. Mosby-Year Book, Inc. 1996. q http: //chemed. chem. purdue. edu/genchem/ (accessed on July 28, 2003) q Partial list of ingredients for the mango from Snyder, C. H. “The Extraordinary Chemistry of Ordinary Things”, J. Wiley & Sons, 1998. p. 54 q Picture of Mango from http: //www. globalgourmet. com/ggt 0598/art/mango. jpg (accessed on June 6, 2003)
- Slides: 63