Organic Chemistry Organic Chemistry The chemistry of carbon

Organic Chemistry

Organic Chemistry • The chemistry of carbon compounds • Sometimes called carbon chemistry

Organic Chemistry • The backbone element in the molecules of all living things is carbon

Carbon Chemistry • Carbon is one of the only elements that can form long chains • Carbon can form 4 covalent bonds

Organic Chemistry • Carbon forms covalent bonds with hydrogen, oxygen, nitrogen, phosphorus, sulfur, and the halogens

Hydrocarbons

Hydrocarbons • Compounds whose molecular structures contain only carbon & hydrogen

Hydrocarbons CH 4 C 3 H 8 C 6 H 6 C 5 H 10 C 18 H 36 Cx. H 2 y

Nomenclature • Naming compounds • Rules made by IUPAC • International Union of Pure & Applied Chemistry

Nomenclature • Prefix: Indicates # of carbons in the group • Suffix: Indicates the type of organic compd

Organic Prefixes • 1 = • 2 = • 3 = • 4 = • 5 = methethpropbutpent- 6= 7= 8= 9= 10 = hexheptoctnondec-

Organic Suffixes • -ane • -ene • -yne • -ol • -one Sat hydrocarbon DB hydrocarbon TB hydrocarbon Alcohol Ketone etc

Alkanes

Alkanes • Hydrocarbons containing only single covalent bonds

Alkanes • All hydrocarbons with no multiple bonds end with the suffix -ane • Prefix: Alk- any length carbon chain

Alkanes 1 = methane 2 = ethane 3 = propane 4 = butane 5 = pentane 6 = hexane 7 = heptane 8 = octane 9 = nonane 10 = decane

Drill • Name & give the molecular formula for alkanes with 1, 2, 3, 4, & 5 carbons

Alkane Chemical Formula Cn. H 2 n+2

Name each of the following: C 5 H 12 C 3 H 8 C 8 H 18

Saturated Hydrocarbon • A hydrocarbon with the maximum number of hydrogens possible

Unsaturated Hydrocarbon • A hydrocarbon with less than the maximum number of hydrogens possible

Formula Writing • Molecular Formula C 4 H 10

Formula Writing • Condensed structural formula, C-C & C-H bonds understood CH 3 CH 2 CH 3

Formula Writing • Condensed structural formula, only C-H bonds understood CH 3 -CH 2 -CH 3

Formula Writing • Condensed structural formula, bonds understood & repeats in () CH 3(CH 2)2 CH 3

Formula Writing • Carbon skeleton, C-H bonds omitted C-C-C-C

Formula Writing • Complete structural formula HHHH H C C H HHHH

Stick or minimal Structures

Stick Structures • Line ends & joints represent carbons • Hydrogens understood • Others draw

Draw the following • Propane • hexane • butane • heptane octane pentane methane

Draw the following 5 -butyl-3 -ethyl-4 cyclohexyl-, 2, 2 dimethyl-6 propylnonane

Name the following

Branched Alkanes

Branched Chains • When an alkane chain gets another hydrocarbon group substituted onto it • Branches: sidechains

Branched Chains C C C C C

Naming Branched Chains

1) Find the longest continuous carbon chain & name it as the main chain. The final name will end with the naming of the main chain
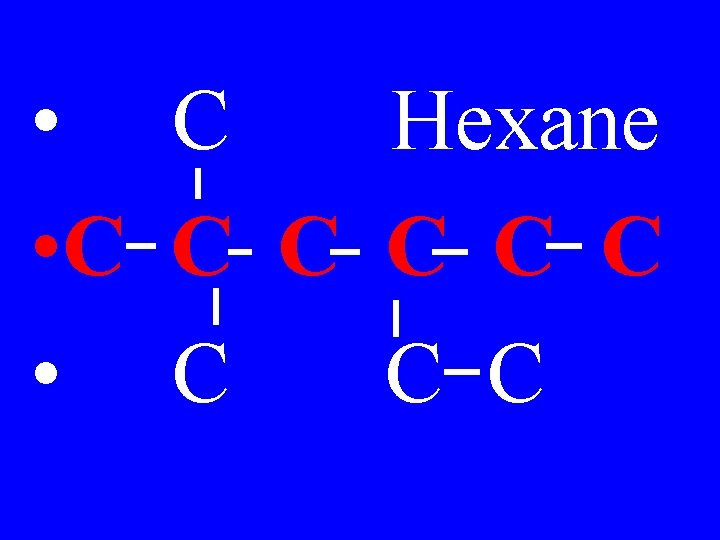
• C Hexane • C C C

2) Number each carbon in the chain; so that, the side chains will be connected to the lowest possible numbers on the carbons

• C Hexane 1 2 3 4 5 6 • C C C

3) Name the side chains or substituted groups, & precede them with the # of the carbon they are connected to & a dash
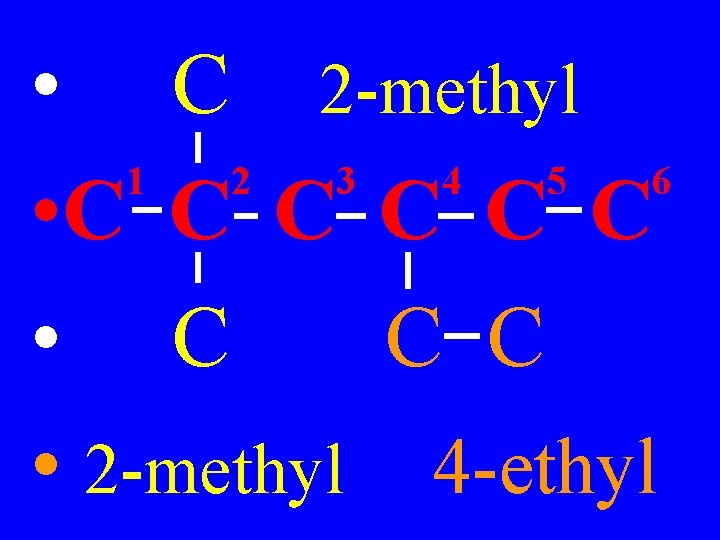
• C 2 -methyl 1 2 3 4 5 6 • C C C • 2 -methyl 4 -ethyl

4) If identical groups appear more than once, use prefixes: di for 2, tri for 3, tetra for 4, penta for 5, etc Examples: dimethyl, tripropyl, etc
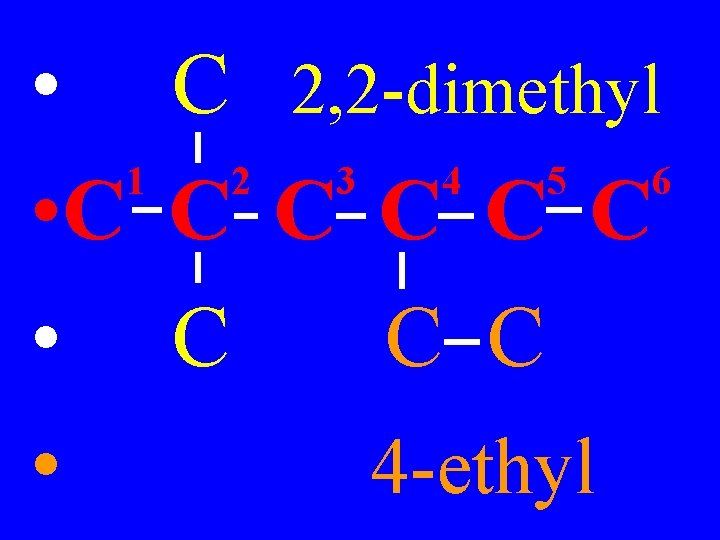
• C 2, 2 -dimethyl 1 2 3 4 5 6 • C C C • 4 -ethyl

5) Name the sidechains or groups in alphabetical order, but ignore the prefixes when doing so · Examples: ethyl comes before dimethyl, e < m

6) Separate numbers with commas, use hyphens between words & numbers, and write the alkanes as one word · Example: dimethylhexane
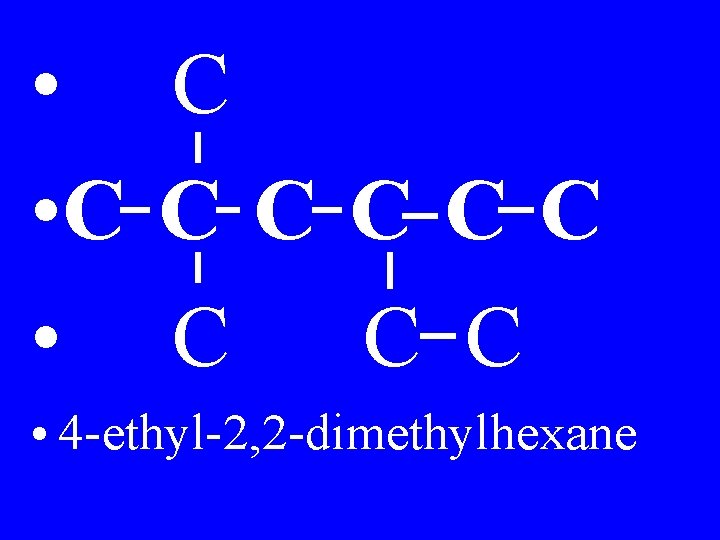
• C C C C • 4 -ethyl-2, 2 -dimethylhexane

Drill: Name CH 3 CH 2 H 3 C-CH-CH 2 -CH-CH-CH 3 CH 2 CH 3

Deriving Structures from Names • Draw the main chain first –Look the name ending • Draw the branches –Look the # & group name

Draw: • 3, 4 -diethyl-2 -methyloctane • 2, 3, 5 -trimethylhexane • 5 -butyl-3 -ethyl-2, 6, 8 trimethyl-4 -propyldecane

Drill: Draw: 5 -butyl-3 -ethyl-2, 6, 8, 9 -tetramethyl-4, 7 dipropyldecane

Isomers • Compounds that have the same chemical formula, but different shapes
- Slides: 52