Introduction to Industrial Chemistry Dr Ramy Y Morjan





































- Slides: 40

Introduction to Industrial Chemistry Dr. Ramy Y. Morjan Faculty of Science The Islamic University-Gaza 2014

Introduction v What is Industrial Chemistry? The development, optimization and monitoring of fundamental chemical processes used in industry. It Deals with transforming raw materials and precursors into useful commercial products for society

Introduction v Industrial Chemists study and apply the physical and chemical properties of substances to determine their composition. They use this information to develop new substances, processes and products and to increase scientific knowledge.

Characteristics of the Chemical Industry 1) Basic objective - make a profit 2) Very competitive 3) Highly dependent on science and technology 4) Spends large amounts of its money on R&D 5) Large capital requirements - to construct, expand maintain production facilities 6) Low labor requirements - BUT needs highly qualified personnel 7) Industry Growth - generally through integration rather than diversification

The Chemical Sector at a Glance Chemicals and chemical products: • account for ~10% of total world trade in all commodities • are the 2 nd largest single item of global trade (road vehicles being the 1 st). World Chemicals Output (2002): $1. 6 Trillion USD • Europe 31% USA 28% Asia/Pacific 27% Other*14%. North American Chemicals Output (2002): $505 Billion USD • USA 92% ($467 Billion USD) • Canada 5% ($23 Billion USD) • Mexico 3% ($15 Billion USD)

Laboratory Chemistry vs. Industrial Chemistry There are fundamental differences between the design of a chemical synthesis for industry and that for a research laboratory. Students should be able to 1) explain how industrial synthetic approaches differ from laboratory synthesis methods. 2) evaluate possible reaction schemes based on thermodynamic, economic, and other considerations.

. g. , formation of ethyl alcohol by hydration of ethylene Laboratory Scale • bubble ethylene into 98% H 2 SO 4 • dilute and warm the reaction mixture to hydrolyze the resultant sulfate ester

Laboratory Objectives • synthesize the product in the most convenient manner considering: 1) chemist’s time 2) equipment available (usually must use glassware) 3) conditions achievable

ßIndustrial Scale A stream of ethylene is mixed with steam at 325°C and 1000 psi and passed over a solid catalyst consisting of phosphoric acid Unreacted ethylene is recovered and recycled to the feed stream.

ßIndustrial Objectives ß • produce the product at minimum total cost on a scale that will generate the maximum economic return. may use: 1) large range of temperatures and pressures 2) batch process or continuous operation 3) reactants in vapor phase or liquid phase

Evaluation of a Reaction (process) 1) Evaluation of the reaction 2) Economic feasibility 3) Technical feasibility 4)Other considerations: environmental issues,

Evaluation of a Reaction The chemist must consider not only the well-known, obvious approaches, but also unknown or untested approaches. e. g. , the manufacture of ethylamine

Economic Feasibility Estimate the difference between the market value of the products and the reactants. First approximation, assume: 1) 100% yield 2) no costs of solvents or catalysts 3) no value for co-products These assumptions must be reassessed further on in the development stage.

Technical feasibility There are two basic questions that a chemist or chemical engineer must ask concerning a given chemical reaction: (1) How far does it go, if it is allowed to proceed to equilibrium? (Does it go in the direction of interest at all? ) (2) How fast does it progress?

Other considerations: environmental issues

Unit operation In transforming matter from inexpensive raw materials to highly desired products, chemical engineers became very familiar with the physical and chemical operations necessary in this transformation. Examples of this include: Filtration, Drying distillation, Crystallization Grinding, Sedimentation Combustion, Catalysis, heat exchange, Coating, and so on.

Body Care Products

About the skin ßHuman skin is a highly complex, highly important organ, serving many different functions within the body and being absolutely essential for human. ßLargest organ of the body There about 2 m 2 of skin covering the average human body. (2. 7 kilograms). Nearly 15% of total body weight

About the skin ßControls body temperature ßAllows us to have the sense of touch through nerve endings ßSkin can become infected with bacteria, viruses and fungi ßMends itself when damaged ßCovers and protects everything inside the body

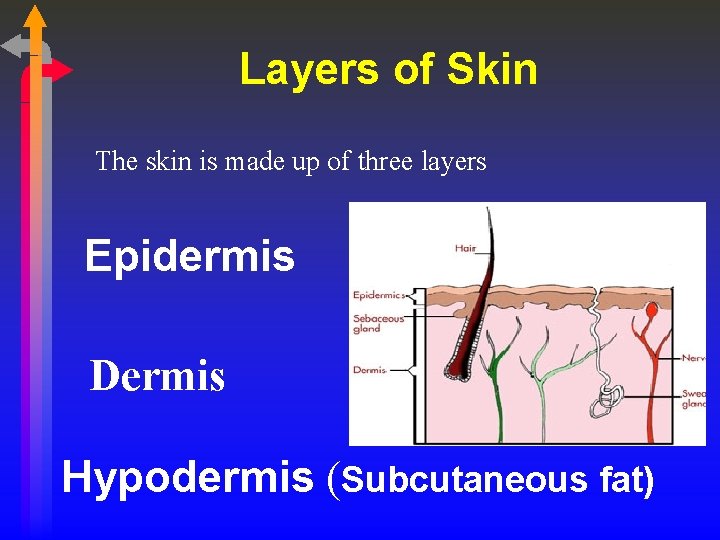
Layers of Skin The skin is made up of three layers Epidermis Dermis Hypodermis (Subcutaneous fat)

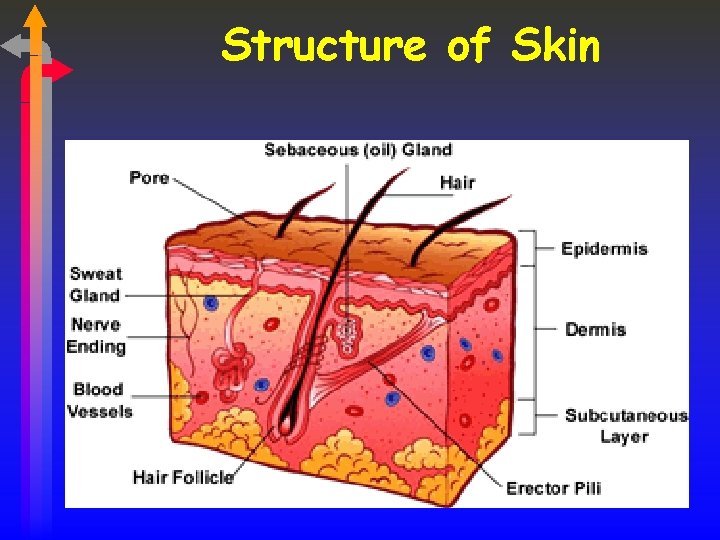
Structure of Skin

What skin looks like Skin layers may look perfectly smooth State of the very top layer (epidermis) Amount of pigment in the layers beneath State of the tissues Blood vessels in the deeper skin layers

Epidermis The top layer of the skin, The epidermis is the part of your skin you can see The epidermis does not contain any blood vessels but gets its oxygen and nutrients from the deeper layers of the skin. Epidermis is hard at work. At the bottom of the epidermis new skin cells are forming. When the cells are ready, they start moving toward the top of your epidermis. This trip takes about 2 weeks to a month. As newer cells continue to move up, older cells near the top die and rise to the surface of your skin. What you see on your hands are really dead skin cells

These old cells are tough and strong, just right for covering your body and protecting it. But they only stick around for a little while. Soon, they'll flake off. Though you can't see it happening, every minute of the day we lose about 30, 000 to 40, 000 dead skin cells off the surface of our skin. You've probably lost about 40, 000 cells. That's almost 4 kilograms of cells every year! But don't think your skin might wear out someday. Your epidermis is always making new skin cells that rise to the top to replace the old ones. Most of the cells in your epidermis (95%) work to make new skin cells The other 5% tissues make a substance called melanin

At the bottom of the epidermis is a very thin membrane , called the basement membrane. Function of the basement membrane 1) Attaches the epidermis firmly, though not rigidly, to the layer below (dermis layer) 2) The second function of the basement membrane is that of a protective barrier against foreign objects

Dermis The second layer of the skin is called Dermis contains blood vessels, nerves, hair roots and sweat glands. Connective tissue Binds structure of skin Elastic fibres Makes the skin flexible Capillaries Tiny blood vessels Muscle fibres Move the hairs Sensory cells Touch, pressure, heat, cold, pain Nerve fibres Activate muscles and glands, send messages to brain Sweat gland Goes up to the surface Hair follicles Hair grows through to the epidermis Sebaceous gland Produce oil to keep skin waterproof

Dermis This part of the skin is sandwiched on top of the Hypodermis (subcutaneous fat) and the epidermis Dermis is a vitally important area of the skin since it is here where the fibroblast cells form the network of fibers of the skin. it is also referred to as the connective tissue. The connective tissue is mainly made-up of Collagen and to a far lesser degree Elastin. C and E are complex proteins responsible for the support and elasticity of the skin and enables the skin to regain its shape after being pulled, stretched or pushed.

Dermis is fed by blood circulating through tiny arteries, veins and capillaries to bring nutrition and oxygen to the Cells whilst removing waste products. The dermis also contains the sensitive nerve endings, sweat glands, hair follicles and sebaceous glands (oil glands The connective tissue of the dermis is made up of Collagen and Elastin fibers immersed in glycosaminoglycans.

What is collagen? Collagen is a protein and is found in all body parts, including the largest organ of the body - the skin. Collagen accounts for up to 75% of the weight of the dermis, and is responsible for the resilience and elasticity of the skin It is made up of three strands of repeating amino acids coiling into the unique collagen triple helix.

The collagen and elastin are responsible for the strength and elasticity of the skin – and degradation of the collagen leads to aging and accompanying wrinkles Collagen has an amino acid composition, which differentiates it from other natural proteins. It contains around 25% glycine, 10% alanine, 12% proline and 10% hydroxyproline, which is the amino acids that characterizes collagen

What damages collagen ? 1) Sun exposure: free radicals (normal body functions (oxidation). & Tempreature 2) Inflammation 3) fat filled diet 4) Smoking: kills off ascorbic acid molecule 5) Pollution and toxins in your environment Vitamin A, Vitamin E , Green Tea , Centella Hyaluronic Acid , Aldenine® , Antarcticine® Serilesine®

Hypodermis (subcutaneous fat) This is the deepest layer of the skin and is manufactured by specialist cells. It is composed mainly of fat (adipose tissue). The thickness of this layer varies from person to person and also from one body area to the next. The hypodermis in women is thicker than in men, which helps to form the rounded curves in women.

Function of Hypodermis This layer acts as insulation and protects the internal organs from temperature variations. acts as an energy reserve from which the body can draw as required. The sebaceous glands These glands produce oil, or sebum, which is a mixture of waxes and fats. The glands empty through minute tubes called ducts. Sebaceous glands occur in the skin of every part of the body except on the palms and soles.

Skin Types 1. Normal Skin 2. Dry Skin 3. Oily Skin 4. Combination Skin 5. Sensitive Skin

1. Normal Skin In skin that belongs to a normal type, the tissue will not reveal any traces of oil. feels neither tight nor greasy Normal skin should feel vibrant, elastic, and supple. It looks clean and smooth and has a good circulation and healthy complexion with a minimum care, it will look good well into your advanced age. Normal skin is the least problematic type.

2. Dry Skin Feels tight and irritable often looks flaky often develops fine lines around the eyes Tightens after washing with soaps or detergents or prolonged exposure to low humidity Can easily develop a sallow tone, wrinkles, and fine pores, and it is very prone to aging and irritating. It might still great on a young person, but, to keep it healthy, you should apply thorough care and use regular skin natural masks and moisturizers.

3. Oily Skin (seborrhoeic skin) This type of skin is particularly common in adolescents and young adults. Oily skin is problematic – it usually looks greasy, thick, coarse, and shiny, has enlarged pores, and tends to break into acne. At this age there is in both sexes a dramatic increase in sebum production under the influence of sex hormones The epidermis tends to thicken, due to increased keratin production, and the pores dilate. As a result the skin feels rough and irregular. Oily skin is not prone much to aging and wrinkling

4. Combination Skin Has patches of both dry and oily skin. Traces of oil coming from nose and forehead, but will be clean in the areas that touched cheeks. Response to many external stimuli by becoming red and blotchy. Feel very tight after washing, be prone to developing dry flaky patches

5. Sensitive Skin Usually very dry, tends to feel tight, and becomes inflamed and irritated easily. Develops reddish and scaly areas, can be itchy, and is prone to breaking into spots. It is the most problematic and fragile type of skin, which needs a very special type of care.
