ELECTROCHEMISTRY Chapter 21 redox reactions electrochemical cells electrode







![Cells at Non-standard Conditions (2) Eo only applies to [ ] = 1 M Cells at Non-standard Conditions (2) Eo only applies to [ ] = 1 M](https://slidetodoc.com/presentation_image_h/d29053ad0026930229820079822bf0e7/image-11.jpg)















- Slides: 35

ELECTROCHEMISTRY Chapter 21 • redox reactions • electrochemical cells • electrode processes • construction • notation Electric automobile • cell potential and Go • standard reduction potentials (Eo) • non-equilibrium conditions (Q) • batteries • corrosion 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 1

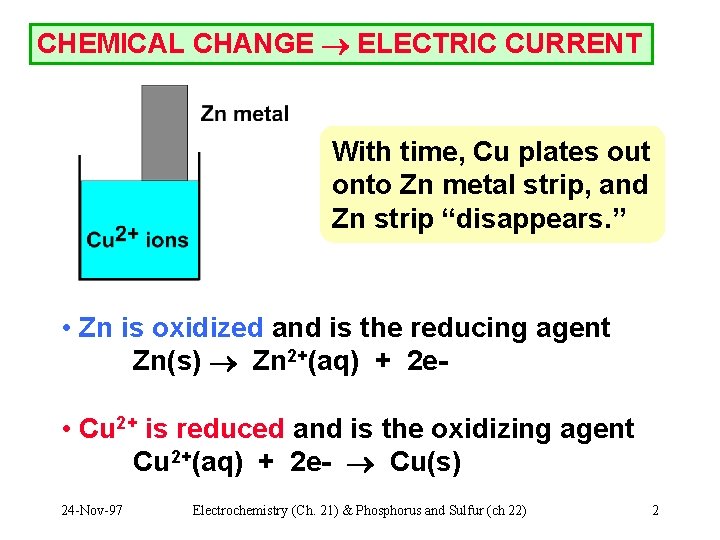
CHEMICAL CHANGE ELECTRIC CURRENT With time, Cu plates out onto Zn metal strip, and Zn strip “disappears. ” • Zn is oxidized and is the reducing agent Zn(s) Zn 2+(aq) + 2 e • Cu 2+ is reduced and is the oxidizing agent Cu 2+(aq) + 2 e- Cu(s) 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 2

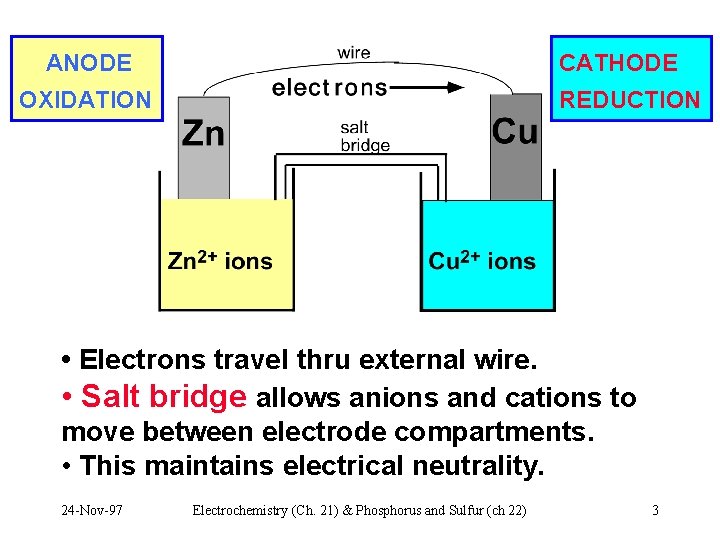
ANODE CATHODE OXIDATION REDUCTION • Electrons travel thru external wire. • Salt bridge allows anions and cations to move between electrode compartments. • This maintains electrical neutrality. 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 3

CELL POTENTIAL, Eo For Zn/Cu, voltage is 1. 10 V at 25°C and when [Zn 2+] and [Cu 2+] = 1. 0 M. • This is the STANDARD CELL POTENTIAL, Eo • Eo is a quantitative measure of the tendency of reactants to proceed to products when all are in their standard states at 25 °C. 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 4

Eo and Go Eo is related to Go, the free energy change for the reaction. Go = - n F Eo • F = Faraday constant = 9. 6485 x 104 J/V • mol • n = the number of moles of electrons transferred. Zn / Zn 2+ // Cu 2+ / Cu n for Zn/Cu cell ? 24 -Nov-97 n=2 Michael Faraday 1791 -1867 Discoverer of • electrolysis • magnetic props. of matter • electromagnetic induction • benzene and other organic chemicals Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 5

Eo and Go (2) Go = - n F Eo • For a product-favored reaction – battery or voltaic cell: Chemistry electric current Reactants Products Go < 0 and so Eo > 0 (Eo is positive) • For a reactant-favored reaction - electrolysis cell: Electric current chemistry Reactants Products Go > 0 and so Eo < 0 (Eo is negative) 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 6

STANDARD CELL POTENTIALS, Eo • Can’t measure half- reaction Eo directly. Therefore, measure it relative to a standard HALF CELL: the Standard Hydrogen Electrode (SHE). 2 H+(aq, 1 M) + 2 e. Eo = 0. 0 V 24 -Nov-97 H 2(g, 1 atm) Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 7

STANDARD REDUCTION POTENTIALS Oxidizing ability of ion Half-Reaction Cu 2+ + 2 e- Cu + 0. 34 2 H+ + 2 e- H 2 0. 00 Zn 2+ + 2 e- Zn -0. 76 BEST Oxidizing agent Cu ? ? 2+ BEST Reducing agent ? Zn ? 24 -Nov-97 Eo (Volts) Reducing ability of element Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 8

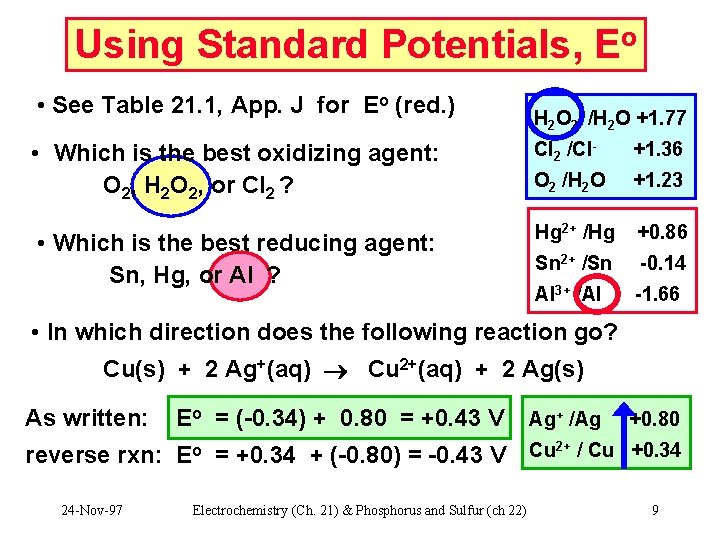
Using Standard Potentials, Eo • See Table 21. 1, App. J for Eo (red. ) • Which is the best oxidizing agent: O 2, H 2 O 2, or Cl 2 ? • Which is the best reducing agent: Sn, Hg, or Al ? H 2 O 2 /H 2 O +1. 77 Cl 2 /Cl- +1. 36 O 2 /H 2 O +1. 23 Hg 2+ /Hg +0. 86 Sn 2+ /Sn -0. 14 Al 3+ /Al -1. 66 • In which direction does the following reaction go? Cu(s) + 2 Ag+(aq) Cu 2+(aq) + 2 Ag(s) As written: Eo = (-0. 34) + 0. 80 = +0. 43 V Ag+ /Ag +0. 80 2+ reverse rxn: Eo = +0. 34 + (-0. 80) = -0. 43 V Cu / Cu +0. 34 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 9

Cells at Non-standard Conditions For ANY REDOX reaction, • Standard Reduction Potentials allow prediction of direction of spontaneous reaction If Eo > 0 reaction proceeds to RIGHT (products) If Eo < 0 reaction proceeds to LEFT (reactants) • Eo only applies to [ ] = 1 M for all aqueous species • at other concentrations, the cell potential differs • Ecell can be predicted by Nernst equation 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 10
![Cells at Nonstandard Conditions 2 Eo only applies to 1 M Cells at Non-standard Conditions (2) Eo only applies to [ ] = 1 M](https://slidetodoc.com/presentation_image_h/d29053ad0026930229820079822bf0e7/image-11.jpg)
Cells at Non-standard Conditions (2) Eo only applies to [ ] = 1 M for all aqueous species at other concentrations, the cell potential differs Ecell can be predicted by Nernst equation E = Eo - RT ln (Q) n. F n = # e- transferred F = Faraday’s constant = 9. 6485 x 104 J/V • mol Q is the REACTION QUOTIENT (recall ch. 16, 20) Go, Eo At equilibrium refer to ALL REACTANTS relative to G = 0 E= 0 Q=K ALL PRODUCTS 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 11

Example of Nernst Equation E = Eo - RT ln (Q) n. F Q. Determine the potential of a Daniels cell with [Zn 2+] = 0. 5 M and [Cu 2+] = 2. 0 M; Eo = 1. 10 V A. Zn / Zn 2+ (0. 5 M) // Cu 2+ (2. 0 M) / Cu Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s) [Zn 2+] Q=? [Cu 2+] E = 1. 10 - (0. 0257) ln ( [Zn 2+]/[Cu 2+] ) 2 E = 1. 10 - (-0. 018) = 1. 118 V 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 12

Nernst Equation (2) E = Eo RT ln (Q) n. F Q. What is the cell potential and the [Zn 2+] , [Cu 2+] when the cell is completely discharged? A. When cell is fully discharged: • chemical reaction is at equilibrium • E=0 G = 0 • Q=K and thus 0 = Eo - (RT/n. F) ln (K) Determine Kc = e Kc from Eo by (n. FEo/RT) or Eo = (RT/n. F) ln (K) or ln (K) = n. FEo/RT = (n/0. 0257) Eo at T = 298 K So. . . K = e 24 -Nov-97 (2)(1. 10)/(. 0257) = 1. 5 x 1037 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 13

Primary (storage) Batteries Anode (-) Zn 2+ + 2 e- Cathode (+) 2 NH 4 + 2 e- 2 NH 3 + H 2 + Common dry cell (Le. Clanché Cell) Anode (-) Zn (s) + 2 OH- (aq) Zn. O (s) + 2 H 2 O + 2 e- Cathode (+) Mercury Battery (calculators etc) 24 -Nov-97 Hg. O (s) + H 2 O + 2 e Hg (l) + 2 OH- (aq) Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 14

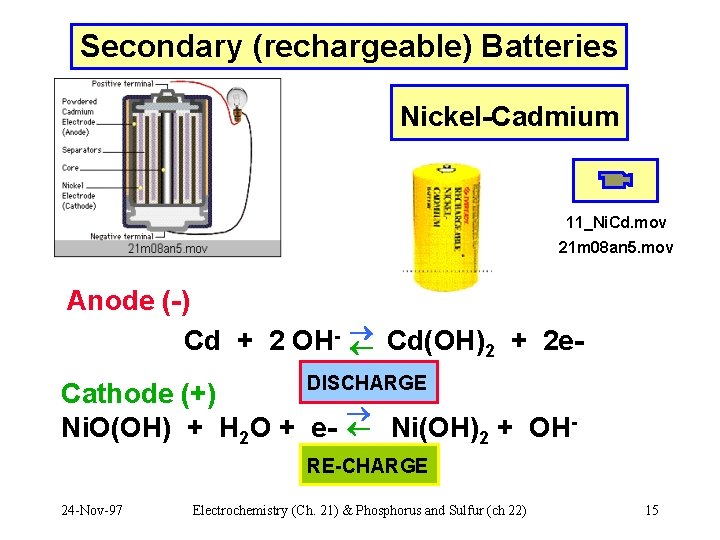
Secondary (rechargeable) Batteries Nickel-Cadmium 11_Ni. Cd. mov 21 m 08 an 5. mov Anode (-) Cd + 2 OH- Cd(OH)2 + 2 e. DISCHARGE Cathode (+) Ni. O(OH) + H 2 O + e- Ni(OH)2 + OHRE-CHARGE 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 15

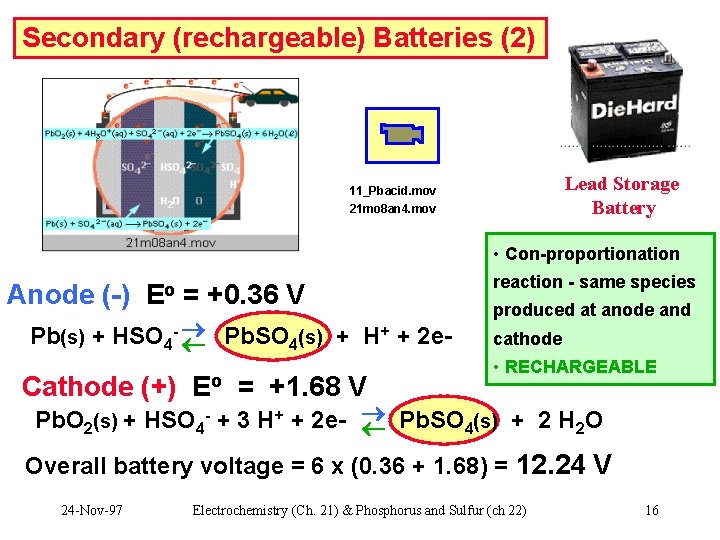
Secondary (rechargeable) Batteries (2) Lead Storage Battery 11_Pbacid. mov 21 mo 8 an 4. mov • Con-proportionation Anode (-) Eo = +0. 36 V + Pb(s) + HSO 4 - Pb. SO 4(s) + H + 2 e- Cathode (+) Eo = +1. 68 V reaction - same species produced at anode and cathode • RECHARGEABLE Pb. O 2(s) + HSO 4 - + 3 H+ + 2 e- Pb. SO 4(s) + 2 H 2 O Overall battery voltage = 6 x (0. 36 + 1. 68) = 12. 24 V 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 16

Corrosion - an electrochemical reaction Electrochemical or redox reactions are tremendously damaging to modern society e. g. - rusting of cars, etc: EOX = +0. 44 anode: Fe - Fe 2+ + 2 e. ERED = +0. 40 cathode: O 2 + 2 H 2 O + 4 e- 4 OHnet: 2 Fe(s) + O 2 (g) + 2 H 2 O (l) 2 Fe(OH)2 (s) Ecell = +0. 84 Mechanisms for minimizing corrosion • sacrificial anodes (cathodic protection) (e. g. Mg) • coatings - e. g. galvanized steel • - Zn layer forms (Zn(OH)2. x. Zn. CO 3) • this is INERT (like Al 2 O 3); if breaks, Zn is sacrificial 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 17

Electrolysis of Aqueous Na. OH Electric Energy Chemical Change Anode : Eo = -0. 40 V 4 OH- O 2(g) + 2 H 2 O + 2 e- Cathode : Eo = -0. 83 V 11_electrolysis. mov 21 m 10 vd 1. mov 4 H 2 O + 4 e- 2 H 2 + 4 OH- Eo for cell = -1. 23 V since Eo < 0 , Go > 0 - not spontaneous ! - ONLY occurs if Eexternal > 1. 23 V is applied 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 18

ELECTROCHEMISTRY Chapter 21 • redox reactions • electrochemical cells • construction • electrode processes • notation Electric automobile • cell potential and Go • standard reduction potentials (Eo) • non-equilibrium conditions (Q) • batteries • corrosion 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 19

Phosphorus and Sulfur Chemistry Kotz, Ch 22 • the elements • physical properties • chemical reactions • redox chemistry • acid/base chemistry 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 20

Elemental Sulfur - Obtained from: - free element in volcanic vents ‘mined’ by Frasch process - minerals : Fe. S 2 (pyrite), Pb. S 2 (galena) Cu 2 S (chalcocite) (S produced as by-product of metal extraction) - natural gas and oil processing desulfurization: 2 H 2 S (g) + SO 2 (g) 3 S (s) + 2 H 2 O (g) 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 21

Elemental Phosphorus - not found free in nature - too easily oxidized “phosphate rock” Ca 3 (PO 4)2 Ca 5 (PO 4)3 F Ca 5 (PO 4)3 OH Ca 5 (PO 4)3 Cl calcium phosphate fluoro apatite hydroxy apatite (teeth etc) chloro apatite • Isolate phosphorus from these ‘rocks’ by burning with charcoal and sand 2 Ca 3 (PO 4)2 (l) + 6 Si. O 2 (s) P 4 O 10 (g) + 6 Ca. Si. O 3 (l) P 4 O 10 (g) + 10 C (s) P 4 + 10 CO (g) 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 22

Structure of P P 4 - white (or yellow) phosphorus (m. p. 44 o. C) Pn - red or black phosphorus m. p. > 400 o. C Allotropes : - different structural forms of the same element or compound OTHER EXAMPLES ? ? C (diamond, graphite, fullerene) 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 23

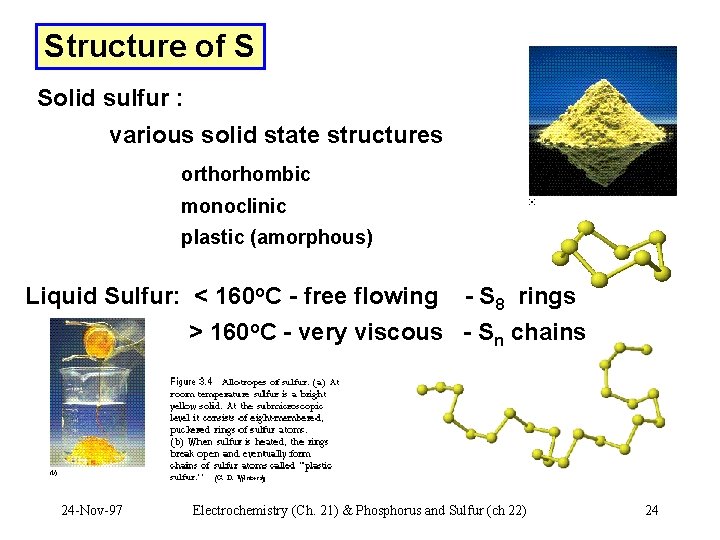
Structure of S Solid sulfur : various solid state structures orthorhombic monoclinic plastic (amorphous) Liquid Sulfur: < 160 o. C - free flowing - S 8 rings > 160 o. C - very viscous - Sn chains 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 24

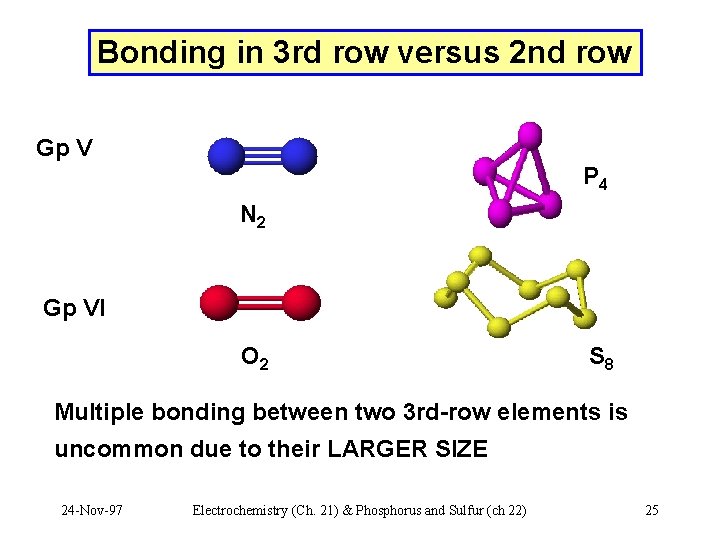
Bonding in 3 rd row versus 2 nd row Gp V P 4 N 2 Gp VI O 2 S 8 Multiple bonding between two 3 rd-row elements is uncommon due to their LARGER SIZE 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 25

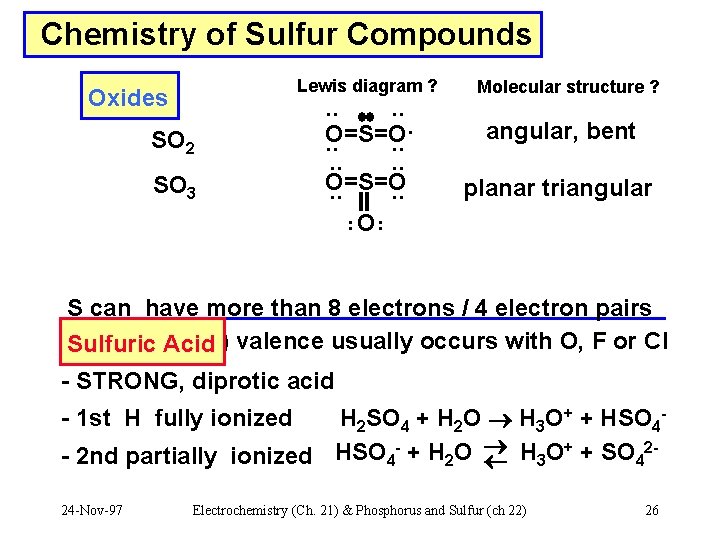
Chemistry of Sulfur Compounds Lewis diagram ? Oxides SO 2 SO 3 . . O=S=O. . . O. . Molecular structure ? angular, bent planar triangular S can have more than 8 electrons / 4 electron pairs expanded (>4) valence usually occurs with O, F or Cl Sulfuric Acid - STRONG, diprotic acid H 2 SO 4 + H 2 O H 3 O+ + HSO 4+ 2 - 2 nd partially ionized HSO 4 - + H 2 O H 3 O + SO 4 - 1 st H fully ionized 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 26

Reactions of Sulfuric Acid 1. Strong acid Na. NO 3 + H 2 SO 4 HNO 3 + Na. HSO 4 2. Dehydrating agent C 11 H 22 O 11 + H 2 SO 4 12 C + 11 H 3 O+ 11 HSO 43. Strong oxidizing agent 2 Br- + 2 H 2 SO 4 (conc. ) 2 Br 2 + SO 42 - + SO 2 + 2 H 2 O 4. Useful solvent : m. p. 10 o. C b. p. 338 o. C 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 27

Oxidation States of Sulfur and Phosphorus Both S and P have many oxidation states - and lots of redox chemistry Sulfur O. N. e. g. -2 H 2 S 0 name -3 Al. P S 8 0 P 4 +2 SCl 2 +3 PCl 3, H 3 PO 3 phosphorus +4 SF 4, H 2 SO 3 sulfurous PO 33 - phosphite SO 32 - sulfite PF 5, H 3 PO 4 phosphoric SF 6, H 2 SO 4 sulfuric PO 43 - phosphate SO 42 - sulfate +6 24 -Nov-97 sulfide Phosphorus O. N. e. g. name +5 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) phosphide 28

Redox chemistry of sulfur compounds Compounds in intermediate oxidation states S(2) or S(4) can act as both oxidizing and reducing agents SO 2 can act as a reducing agent. . . SO 2 (g) + Br 2 (aq) + 6 H 2 O 2 Br-(aq)+ SO 42 - (aq) + 4 H 3 O+ (aq) 5 SO 2 (g) + 2 Mn. O 4 - (aq) + 6 H 2 O 5 SO 42 - (aq) + 2 Mn 2+ (aq) + 4 H 3 O+ (aq) and can act as an oxidizing agent: SO 2 (g) + 2 H 2 S (g) 3 S(s) + 2 H 2 O Water is both CATALYST and product ! - autocatalysis 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 29

Chemistry of phosphorus compounds OXIDES P 4 + 3 O 2 P 4 O 6 P 4 + 5 O 2 P 4 O 10 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 30

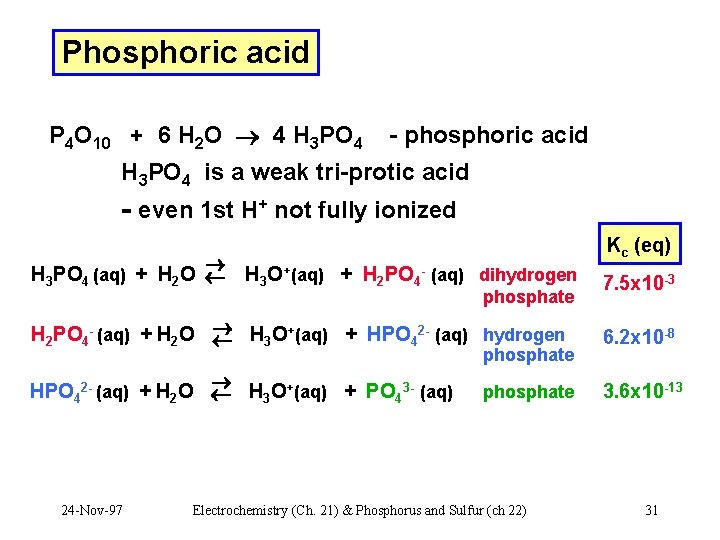
Phosphoric acid P 4 O 10 + 6 H 2 O 4 H 3 PO 4 - phosphoric acid H 3 PO 4 is a weak tri-protic acid - even 1 st H+ not fully ionized H 3 PO 4 (aq) + H 2 O Kc (eq) H 3 O+(aq) + H 2 PO 4 - (aq) dihydrogen phosphate 7. 5 x 10 -3 + 2 H 2 PO 4 - (aq) + H 2 O H 3 O (aq) + HPO 4 (aq) hydrogen 6. 2 x 10 -8 HPO 42 - (aq) + H 2 O H 3 O+(aq) + PO 43 - (aq) 3. 6 x 10 -13 phosphate 24 -Nov-97 phosphate Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 31

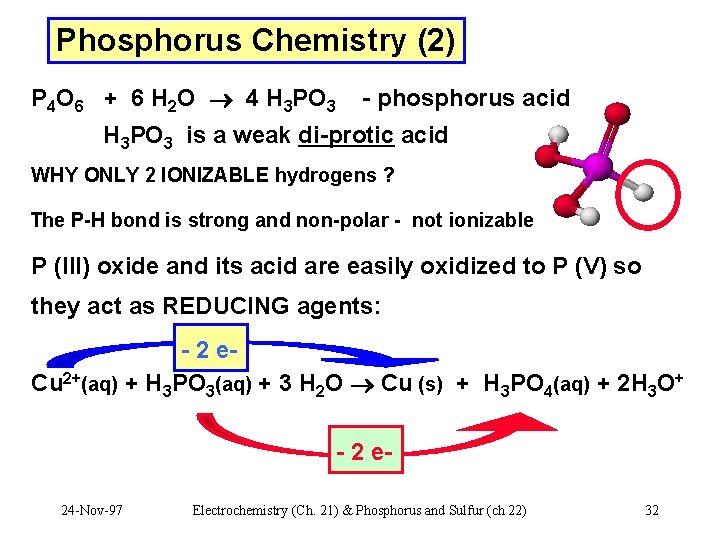
Phosphorus Chemistry (2) P 4 O 6 + 6 H 2 O 4 H 3 PO 3 - phosphorus acid H 3 PO 3 is a weak di-protic acid WHY ONLY 2 IONIZABLE hydrogens ? The P-H bond is strong and non-polar - not ionizable P (III) oxide and its acid are easily oxidized to P (V) so they act as REDUCING agents: - 2 e. Cu 2+(aq) + H 3 PO 3(aq) + 3 H 2 O Cu (s) + H 3 PO 4(aq) + 2 H 3 O+ - 2 e 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 32

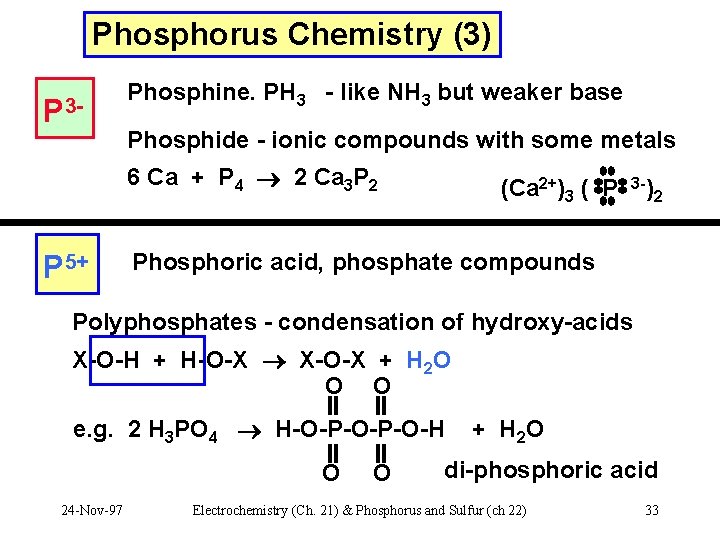
Phosphorus Chemistry (3) P 3 - Phosphine. PH 3 - like NH 3 but weaker base Phosphide - ionic compounds with some metals 6 Ca + P 4 2 Ca 3 P 2 P 5+ (Ca 2+)3 ( P 3 -) 2 Phosphoric acid, phosphate compounds Polyphosphates - condensation of hydroxy-acids X-O-H + H-O-X X-O-X + H 2 O O O e. g. 2 H 3 PO 4 H-O-P-O-H O 24 -Nov-97 O + H 2 O di-phosphoric acid Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 33

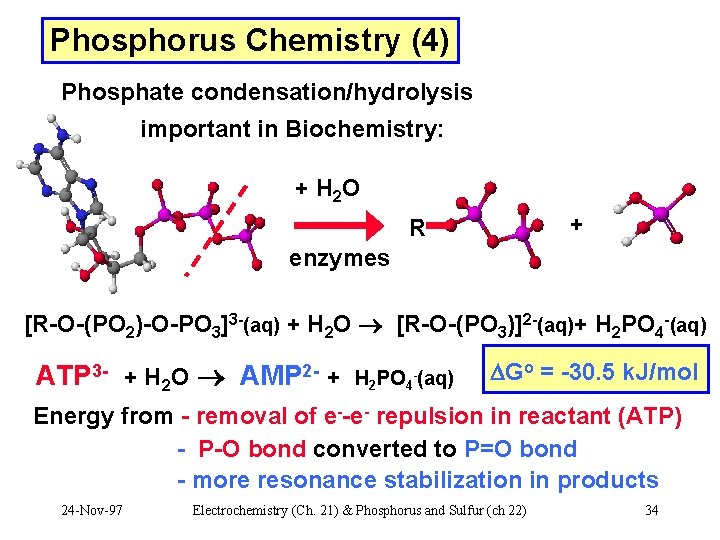
Phosphorus Chemistry (4) Phosphate condensation/hydrolysis important in Biochemistry: + H 2 O + R enzymes [R-O-(PO 2)-O-PO 3]3 -(aq) + H 2 O [R-O-(PO 3)]2 -(aq)+ H 2 PO 4 -(aq) ATP 3 - + H 2 O AMP 2 - + H 2 PO 4 -(aq) Go = -30. 5 k. J/mol Energy from - removal of e--e- repulsion in reactant (ATP) - P-O bond converted to P=O bond - more resonance stabilization in products 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 34

P and S Chemistry Kotz, Ch 22 • Physical properties • Chemical reactions • redox chemistry • acid/base chemistry 24 -Nov-97 Electrochemistry (Ch. 21) & Phosphorus and Sulfur (ch 22) 35