Development of Atomic Models Democritus l Greek philosopher







































- Slides: 53

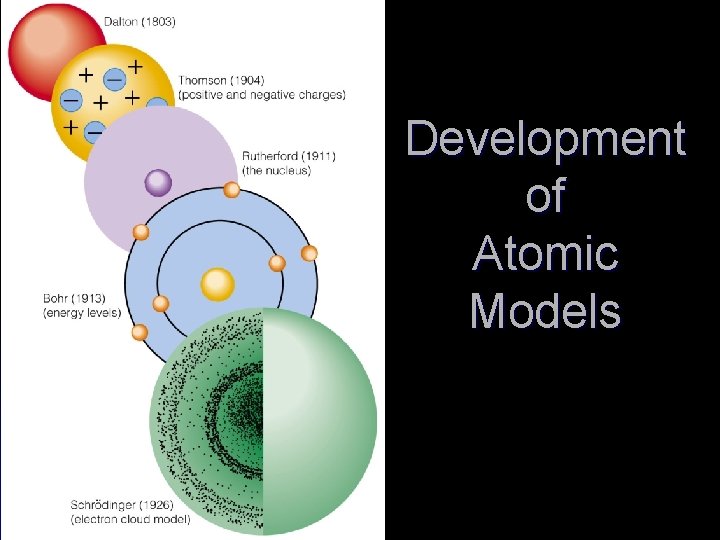
Development of Atomic Models

Democritus l Greek philosopher (400 BC) Matter can’t be divided forever Eventually, a piece is “indivisible” l “Atomos, ” = “not to be cut” l l

Dalton’s Atomic Theory (early 1800’s) l Matter made of tiny indivisible particles called “atoms”. l Each element’s atoms are different.

l Compounds form when atoms combine in fixed proportions. l Chemical reactions involve rearrangement of atoms. l Atoms are conserved in chemical reactions.

Page from Dalton’s Journal

Dalton’s Atomic Theory called “Hard Spheres Model”

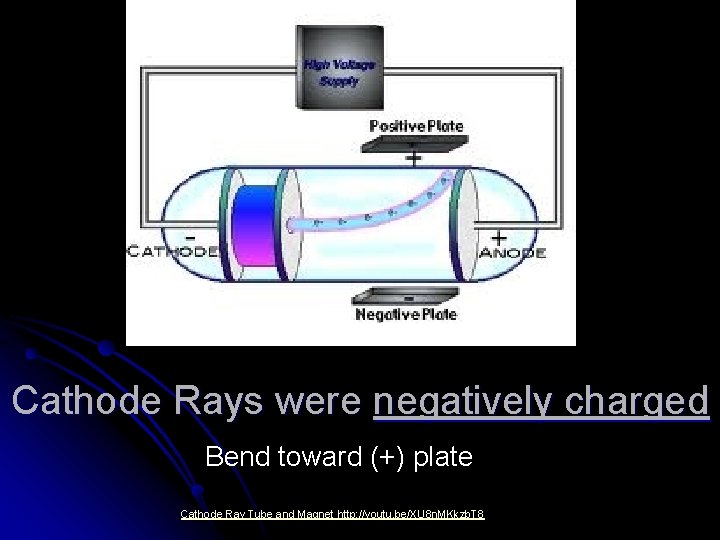
JJ Thomson (1897) l Studied “cathode rays” (electric current) in a “Crooke’s Tube”. l Fluorescent screen, shows how cathode ray behaved in a magnetic field. Lets draw a typical Crooke’s Tube in our notes.

Cathode Rays were negatively charged Bend toward (+) plate Cathode Ray Tube and Magnet http: //youtu. be/XU 8 n. MKkzb. T 8

Cathode Rays were particles They couldn’t pass through matter.

Thomson’s Conclusions l Negative “Cathode Ray” particles must come from within atoms. l Discovered first subatomic particle l Negative electron However, matter is neutral so a positive charge must exist to balance the negative!!

Thomson’s Theory: l Atoms are positively charged spheres with negatively charged particles scattered throughout. Called the “Plum Pudding Model” Brian Cox: Thompson and Discovery of Electron http: //youtu. be/Id. Tx. GJj. A 4 Jw

Ernest Rutherford (1908) l Physicist working in new field of radioactivity.

Found 3 Types of Radiation l Used magnetic field to isolate three types of radiation by their charge. l l l Alpha (α) Beta (β) Gamma (γ)

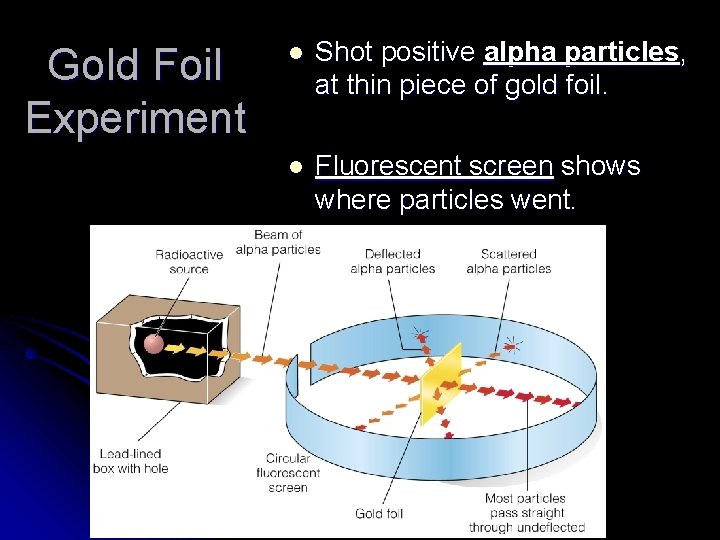
Gold Foil Experiment l Shot positive alpha particles, at thin piece of gold foil. l Fluorescent screen shows where particles went.

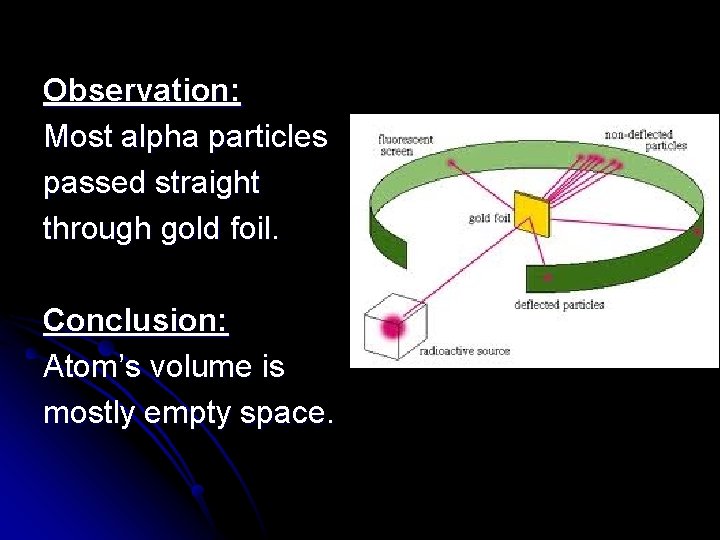
Observation: Most alpha particles passed straight through gold foil. Conclusion: Atom’s volume is mostly empty space.

Observation: Some alpha particles deflected at an angle or bounced back. Conclusion: Atoms have a very small, dense positively charged nucleus. http: //www. kentchemistry. com/moviesfiles/Units/Atomic. Structure/Ruthe rford 3. htm

Nucleus is extremely small compared to the size of the atom as a whole. Deflections happened rarely (1/8000). Modern Example of Gold Foil Experiment in Action http: //youtu. be/XBq. Hkraf 8 i. E

Rutherford’s Model is called the “Nuclear Model” Brian Cox: Rutherford and the Nucleus http: //youtu. be/wz. ALbz. Tdnc 8

Comparison to Thomson l Positive charge only in nucleus. l Negative particles scattered outside nucleus. l Charge not disbursed evenly. Simulator: https: //phet. colorado. edu/sims/html/rutherfordscattering/latest/rutherford-scattering_en. html

Niels Bohr (1913) l Came up with the “Planetary Model”

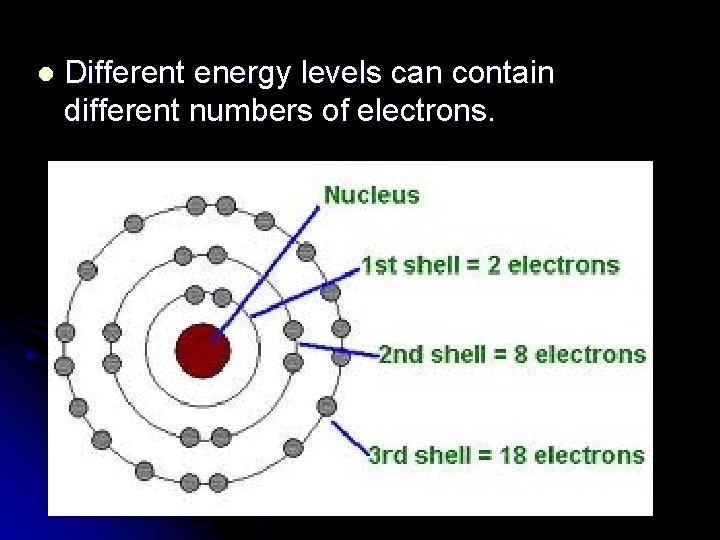
l Electrons circle nucleus in specific energy levels or “shells”. l The higher the “energy level, ” the higher the electron’s energy.

l Different energy levels can contain different numbers of electrons.

l n = the number of the energy level 2 2 n = maximum number of electrons an energy level can hold. Ex: Level 3 can hold 2(3)2 = 18 electrons

Energy Level configurations of neutral atoms are below its symbol. l Shows # of electrons in each occupied energy level l Ex: Bromine 2 -8 -18 -7

Draw a Bohr Atom Ex: Fluorine Atom (F) l Protons =9 l Neutrons = 10 l Electrons = 9 l How many energy levels do you draw? l How many electrons in each level?

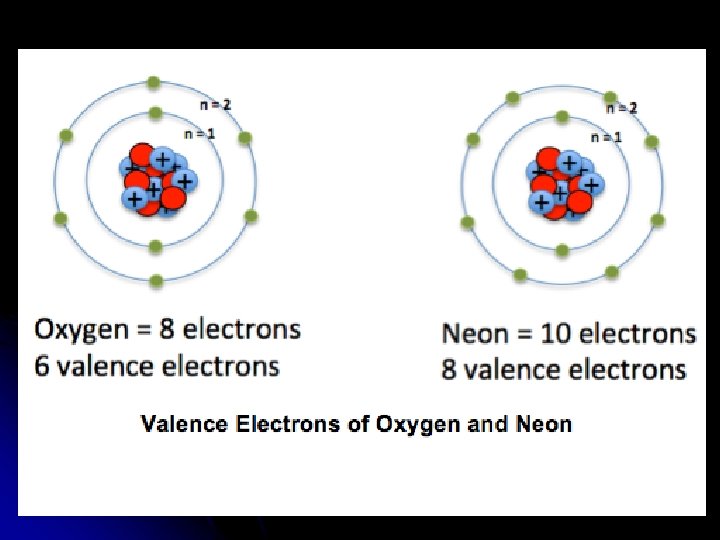
Outermost Electrons Valence Electrons: l Electrons found in highest occupied energy level (furthest from nucleus) l Involved in forming bonds with other atoms l “Stable Octet” of valence is desirable l What Group on the Table is stable as atoms and doesn’t need to form bonds?


l Noble Gases: (Group 18) l “Inert” l Ex: and unreactive (have stable octet) Argon 2 -8 -8, Neon 2 -8 Note: Helium is a Noble Gas too! It only has 2 electrons but its one energy level is complete and stable

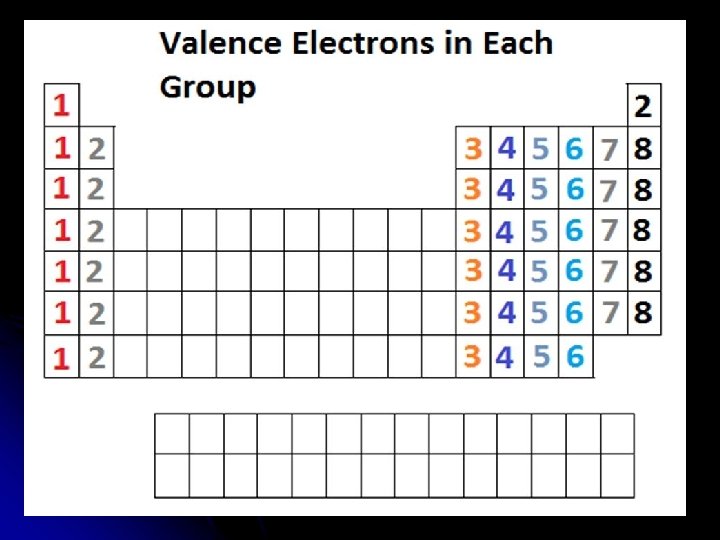
Valence Electrons l Look at last number in atom’s electron configuration to determine number of valence electrons. l Ex: l Al 2 -8 -3 3 valence l. F 2 -7 7 valence

Looking for Trends l Where do we see a similar valence electron pattern on the Periodic Table? l Down a Group? (column) l Across a Period? (row)


Lewis Dot Diagrams l Shows number of valence electrons an atom has as “dots” around atom’s symbol. Phosphorus is 2 -8 -5

Kernel Nucleus and non-valence electrons l Inner part of atom not involved directly in reactions l l Ex: l Al 2 -8 -3 has 10 kernel electrons and 3 valence electrons

Draw a Bohr Ion l One or more electrons gets added or taken out of the outer energy level. Ex: Magnesium Ion (Mg+2) l Protons = 12 l Neutrons = 12 l Electrons = 10

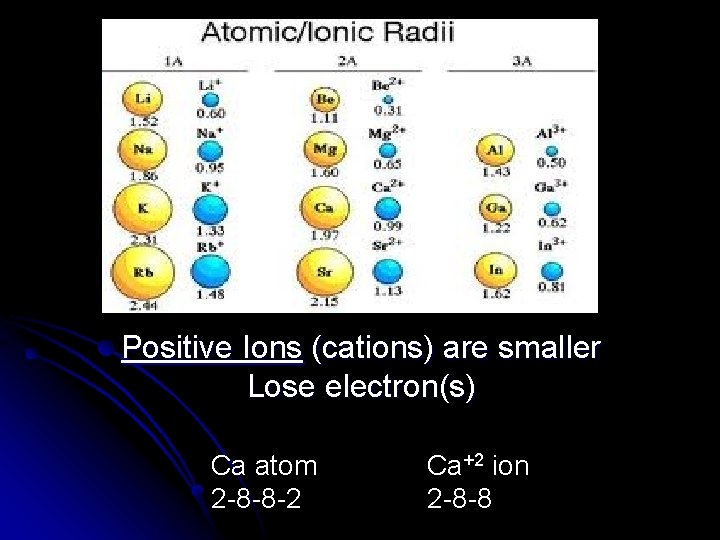
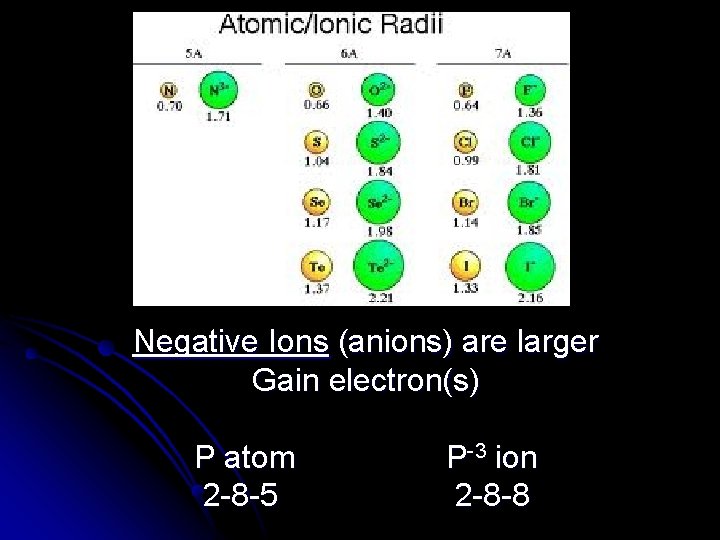
Pause for Thought l What do you think happens to the size of an atom when it gains or loses electrons and becomes an ion?

Positive Ions (cations) are smaller Lose electron(s) Ca atom 2 -8 -8 -2 Ca+2 ion 2 -8 -8

Negative Ions (anions) are larger Gain electron(s) P atom 2 -8 -5 P-3 ion 2 -8 -8

How Did Bohr Come Up With His Model? l Studied spectral lines emitted by various elements

What are Spectral Lines? l Energy absorbed by an atom causes it to emit a unique set of colored lines.

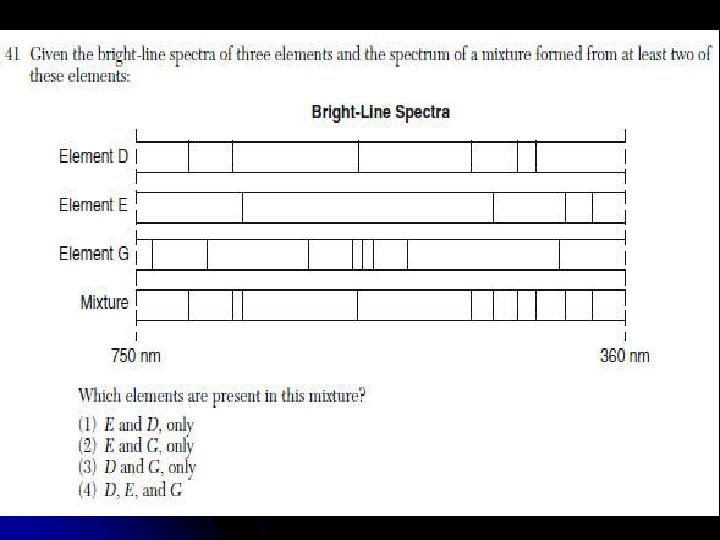
Spectral lines are different for each element l Used to identify elements present in a sample. (“Fingerprint”) l


What Causes Spectral Lines? Electrons jumping between energy levels l Electrons absorb energy then emit it l https: //phet. colorado. edu/en/simulation/legacy/discharge-lamps Video of Line Spectra of Hydrogen http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/linesp 16. swf

Ground State l Electrons normally exist in lowest energy level possible called “ground state”. (stable) l “Ground state” e- configurations are written on periodic table l Ex: Aluminum is 2 -8 -3 Calcium is 2 -8 -8 -2

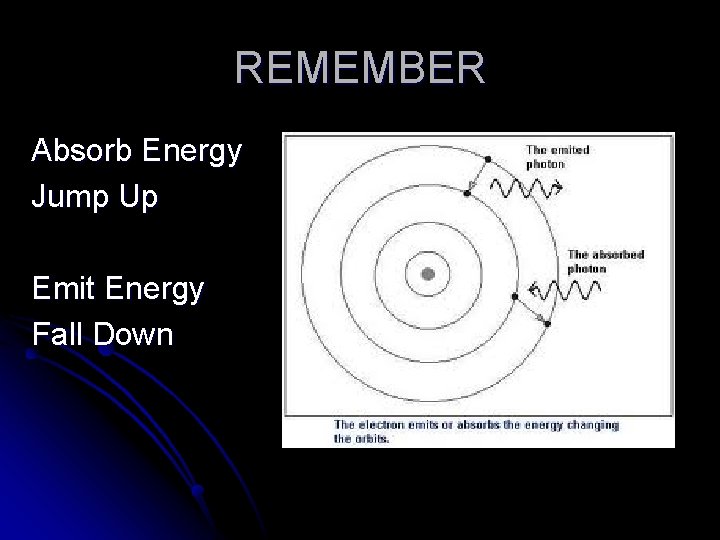
Excited State Electrons absorb a photon (or “quanta”) of energy and “jump up” to a higher energy level. This is called “excited state”. (unstable)

Jumping Electrons l Electrons quickly “fall back down” to ground state. (stable) l They emit a photon (or “quanta”) of energy that corresponds to how far they jumped.

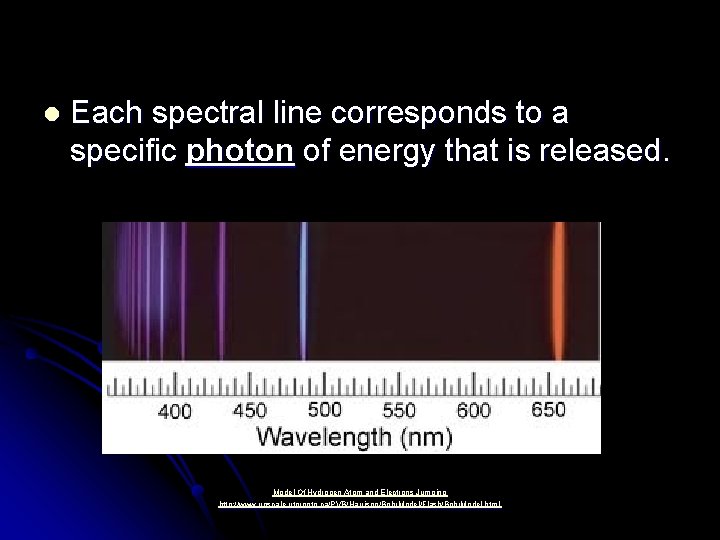
l Each spectral line corresponds to a specific photon of energy that is released. Model Of Hydrogen Atom and Electrons Jumping http: //www. upscale. utoronto. ca/PVB/Harrison/Bohr. Model/Flash/Bohr. Model. html

REMEMBER Absorb Energy Jump Up Emit Energy Fall Down

Recognizing Excited State l Count total electrons given and see if the configuration matches the on the table. l Ex: 2 -8 -7 -3 = 20 electrons l Calcium l So (atomic # 20) is 2 -8 -8 -2 this must be showing one of the ways calcium could be in the excited

Modern Atomic Model l Sometimes called: l l Crash Course: History of Atomic Theory (9: 45) https: //www. youtube. com/watch? v=thn. Dx. Fdkz. Zs Charge Cloud Model Wave Mechanical Model Quantum Theory created to help describe spectral line signatures of multielectron elements

How is it Different from the Planetary Model? l We can’t tell exactly where an electron is!! Heisenberg’s Uncertainty Principle: We can’t know exact location and momentum of an electron at the same time.

l Electrons exist in “orbital clouds” l Denser the cloud region the higher the probability of finding an electron there. l Can’t tell exactly where an electron is, but can tell region of higher probability of finding it.

Excited Atoms and Charge Cloud l Excited electrons shift to probability regions further from the nucleus

Comparison to Bohr’s Electron Model l Charge Cloud = area of highest probability of finding electron, but can’t know exact location l Bohr Model = electrons in fixed orbit like planet around the sun at fixed distance from nucleus