Atomic Theory Democritus Greek Philosopher Expressed the belief

Atomic Theory: Democritus • Greek Philosopher • Expressed the belief that matter consisted of very small, _______, particles, which he named _______. • At first, this idea was not accepted by many other scientists.

Atomic Theory: John Dalton • English scientist and school teacher. • ________________ ________________.

John Dalton’s Atomic Theory • Elements are composed of extremely small particles called atoms. • All atoms of a given element are identical, having the same size, mass, and chemical properties. The atoms of one element are different from the atoms of all other elements. • Compounds are composed of atoms of more than one element. • A chemical reaction involves only the separation, combination, or rearrangement of atoms. It does not result in their creation or destruction.
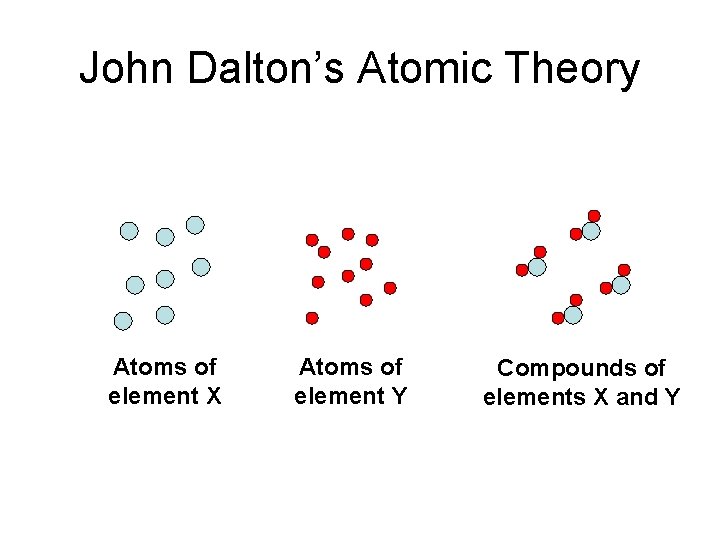
John Dalton’s Atomic Theory Atoms of element X Atoms of element Y Compounds of elements X and Y

Although Dalton’s theory was much more detailed that Democritus’ theory, Dalton made no attempt to describe the structure or composition of the atom. Dalton’s Atom

The Structure of the Atom can be defined as ______ of an _____ that can enter into chemical combination
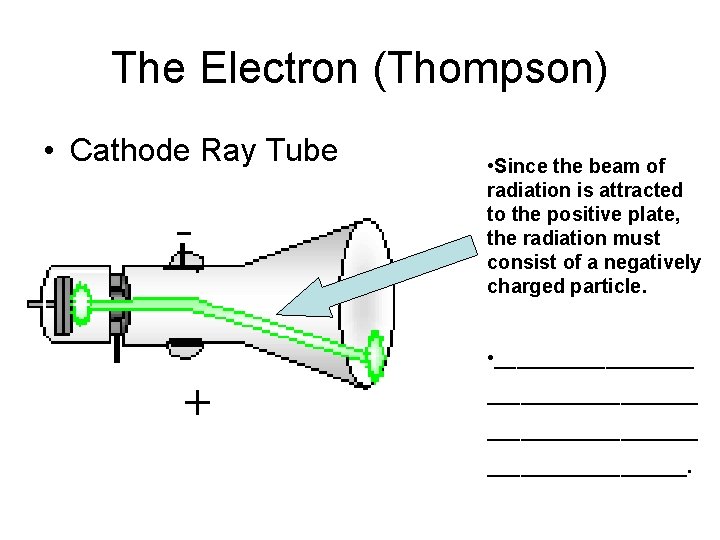
The Electron (Thompson) • Cathode Ray Tube • Since the beam of radiation is attracted to the positive plate, the radiation must consist of a negatively charged particle. • ___________________.

The Proton & the Nucleus • By the early 1900’s, two features of atoms had become clear. – They contain _______. – But, they are electrically ______. – Therefore, there must be a positive particle to balance out the negative charge.

J. J. Thompson: __________ • Thomson proposed that an atom could be thought of as a uniform, positive sphere of matter in which electrons are embedded like raisins in a cake.
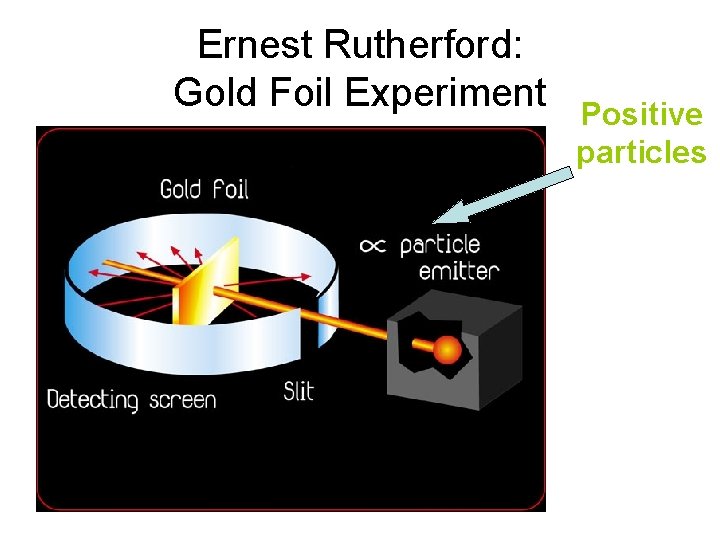
Ernest Rutherford: Gold Foil Experiment Positive particles

Ernest Rutherford: Gold Foil Experiment • The atoms positive charges, are all concentrated in the ________. – A dense central core within the atom. • Whenever an alpha particle came close to a nucleus, it experienced a large repulsive force and therefore a large deflection.

Ernest Rutherford: Gold Foil Experiment • The positively charged particles in the nucleus are called _______.

The Neutron • Simplest atom, Hydrogen. • Contains ____. • Helium • Contains _______. • Since the mass of an electron is MUCH smaller than the mass of a proton, the ratio would be 2: 1.

Chadwick: The Neutron • Ratio is actually found to be 4: 1. (Helium to Hydrogen) • Chadwick discovered a third atomic particle and called them neutrons, because they proved to be electrically neutral. ___________________
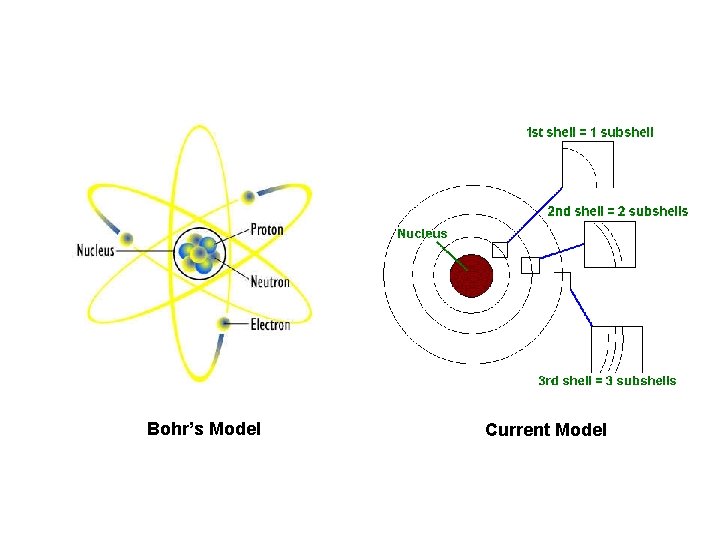
Bohr’s Model Current Model

Atomic Number & Atomic Mass Number of protons and neutrons, combined
- Slides: 16