The History of the Atom Democritus c 460










- Slides: 11

The History of the Atom

Democritus (c. 460 -c. 370 BCE) • Democritus was a philosopher in ancient Greece. • He thought that all matter was made of tiny particles that could not be divided. • The Greek word atomos means ‘indivisible’.

John Dalton (1766 -1844) • Dalton was an English scientist. • He developed modern atomic theory. • His model of the atom is sometimes called the ‘billiard-ball model’.

Dalton’s Billiard-Ball Model • All matter is made of tiny particles called atoms. • Atoms of the same element are identical. • Atoms can combine to form compounds. • Chemical reactions change the grouping of atoms, but not the atoms themselves.

Sir J. J. Thomson (1856 -1940) • Thomson was a British physicist. • He did experiments on cathode rays and discovered the electron. • In 1906, he was awarded a Nobel prize for his discovery. • His model of the atom is called the ‘plum-pudding model’.

Thomson’s Plum-Pudding Model • Dalton realised that negatively charged electrons could come from an atom. • He proposed the ‘plumpudding’ model of the atom, suggesting that atoms consist of negatively-charged electrons in a ‘sea’ of positive charge.

Ernest Rutherford (1871 -1937) • Rutherford was a chemist from Nelson, New Zealand. • Based on the results of his goldfoil experiment, he proposed that most of the mass of an atom is concentrated in a central nucleus. • He won the Nobel Prize in Chemistry in 1908 for his work.

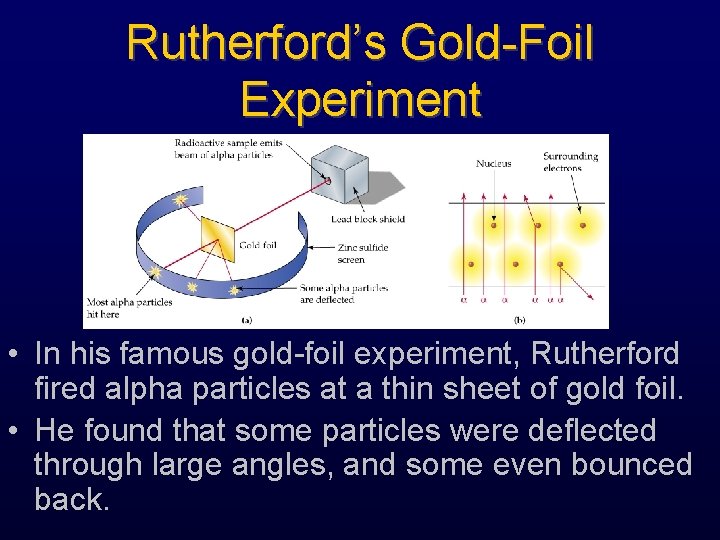
Rutherford’s Gold-Foil Experiment • In his famous gold-foil experiment, Rutherford fired alpha particles at a thin sheet of gold foil. • He found that some particles were deflected through large angles, and some even bounced back.

Rutherford’s Nuclear Model Rutherford concluded that: • most of the mass in an atom must be in a very small, positively-charged nucleus in the centre of the atom • electrons spin around this central nucleus • there was a basic unit of positive charge in the nucleus, called the proton.

• Niels Bohr (1885 -1962) realised that the electrons could only occupy fixed orbits around the nucleus. • Louis de Broglie (1892 -1987) proposed that electrons can be regarded as waves, resulting in an ‘electron cloud’ around the nucleus. • Sir James Chadwick (1891 -1974) discovered the neutron, a nuclear particle with similar mass to a proton but no electrical charge.

References Atom Image: http: //www. turbosquid. com/Full. Preview/Index. cfm/ID/197928 Democritus: http: //smccd. net/accounts/goth/Main. Pages/Chron/Democritus. jpeg Dalton: http: //en. wikipedia. org/wiki/John_Dalton Thomson: http: //en. wikipedia. org/wiki/J_J_Thomson Plum-Pudding Atom: https: //reich-chemistry. wikispaces. com/file/view/348 px. Plum_pudding_atom_svg. png Ernest Rutherford: http: //en. wikipedia. org/wiki/Earnest_Rutherford http: //nobelprize. org/nobel_prizes/chemistry/laureates/1908/rutherford-bio. html Gold-Foil Experiment: http: //wps. prenhall. com/wps/media/objects/602/616516/Media_Assets/Chapter 02/Text_I mages/FG 02_05. JPG Nuclear Atom: http: //upload. wikimedia. org/wikipedia/commons/thumb/e/e 1/Stylised_Lithium_Atom. svg/1 80 px-Stylised_Lithium_Atom. svg. png Electron Cloud: http: //www. csmate. colostate. edu/cltw/cohortpages/viney_old 1/atom. jpg