Chemical Bonding Chemical Bonding Chemical bonding refers to

Chemical Bonding

Chemical Bonding • Chemical bonding refers to the attractive forces that hold atoms together in compounds.

There are two major classes of bonding: • Ionic bonding results from electrostatic interactions among ions, which often results from the net transfer of one or more electrons from one atom to another -Atoms that lose electrons form positive ions called cations. -Atoms that gain electrons form negative ions called anions.

There are two major classes of bonding (continued): • Covalent bonding results from sharing one or more electron pairs between two atoms -Atoms that share electron pairs equally are referred to as nonpolar covalent bonds. -Atoms that share electron pairs unequally are referred to as polar covalent bonds.

All bonds between atoms of different elements have at least some degree of both ionic and covalent character. • Compounds containing predominantly ionic bonding are • • called ionic compounds. Compounds that are held together mainly by covalent bonds are called covalent compounds. Polyatomic ions are groups of covalently bonded atoms that together form ionic bonds with other atoms. Some nonmetallic elements, such as H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2 also involve covalent bonding. Many molecules found in nature consist of covalently bonded carbon-carbon or carbon-hydrogen bonds or both. These compounds are classified as organic compounds.

Properties of Ionic and Covalent Compounds • • • Ionic Compounds Crystalline solids High melting points (>400 o. C) Soluble in polar solvents such as water Molten compounds conduct electricity Aqueous solutions conduct electricity Usually formed between a metal and a nonmetal. • • • Covalent Compounds Gases, Liquids, or soft solids Low melting points (<300 o. C) Insoluble in polar solvents such as water Most are soluble in nonpolar solvents such as hexane and carbon tetrachloride Liquid, molten, and aqueous solutions do not conduct electricity Usually formed between nonmetals

Representing Compounds There are several methods used in representing compounds: • Chemical formula -gives the number of atoms of each type in the compound -Molecular formula gives the actual number of atoms of each type -Empirical formula gives the simplest ratio of atoms of each type • Structural formula -shows the order in which the atoms in a compound are connected

Naming Simple Compounds • Binary Ionic Compounds (Type I) 1. The cation is always named first and the anion second. 2. A monatomic cation takes its name from the name of the element. 3. A monatomic anion is named by taking the root of the element name and adding –ide.

What is the name of Al. Cl 3? 10 1. Aluminum trichloride 2. Aluminum chloride 3. Aluminum chlorine 4. Aluminide chloride 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Naming Simple Compounds • Binary Ionic Compounds (Type II)- Metals that can form more than one positive ion can form more than one type of compound with a given anion. 1. The charge on the metal ion must be specified by using a Roman numeral following the cation name. 2. The anion is named the same as with the Type I compounds.

What is the name of Cu. Cl? 10 1. Copper chloride 2. Copper (I) chloride 3. Copper (II) chloride 4. Copper chlorine 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Hg. O? 10 1. Mercury oxide 2. Mercury (I) oxide 3. Mercury (II) oxide 4. Mercury oxygen 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Fe 2 O 3? 10 1. Iron oxide 2. Iron (I) oxide 3. Iron (II) oxide 4. Iron (III) oxide 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Mn. O 2? 10 1. Manganese oxide 2. Manganese (I) oxide 3. Manganese (II) oxide 4. Manganese (IV) oxide 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Ga. I 3? 10 1. Gallium iodide 2. Gallium (I) iodide 3. Gallium (II) iodide 4. Gallium (III) iodide 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Naming Simple Compounds • Ionic Compounds with Polyatomic Ions 1. Polyatomic ions have specific names which must be memorized. 2. The other ion is named in the same manner as the Type I or Type II compounds.

What is the name of Na 2 SO 4? 10 1. Sodium sulfide 2. Sodium sulfate 3. Sodium sulfite 4. Sodium (II) sulfate 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Fe(NO 3)3? 10 1. Iron nitrate 2. Iron (III) nitrate 3. Iron nitrite 4. Iron (III) nitrite 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Cu. CO 3? 10 1. Copper carbonate 2. Copper (I) carbonate 3. Copper (II) carbonate 4. Copper (III) carbonate 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Na. OCl? 10 1. Sodium oxygen chloride 2. Sodium hypochlorite 3. Sodium chlorite 4. Sodium chlorate 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Naming Simple Compounds • Binary Covalent Compounds (Type III) 1. The first element in the formula is named first, using the full element name. 2. The second element is named as if it were an anion. 3. Prefixes are used to denote the numbers of atoms present.
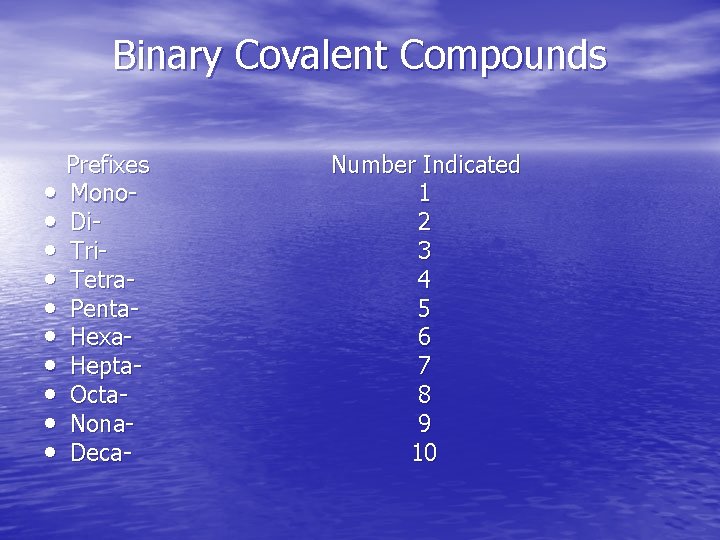
Binary Covalent Compounds • • • Prefixes Mono. Di. Tri. Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca- Number Indicated 1 2 3 4 5 6 7 8 9 10

What is the name of PCl 5? 10 1. Phosphorus chloride 2. Phosphorus (V) chloride 3. Monophosphorus pentachloride 4. Phosphorus pentachloride 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of N 2 O 3? 10 1. Nitrogen oxide 2. Nitrogen (II) oxide 3. Nitrogen trioxide 4. Dinitrogen trioxide 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Nb 2 O 5? 10 1. Diniobium pentaoxide 2. Niobium oxide 3. Niobium (II) oxide 4. Niobium (V) oxide 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

What is the name of Ti(NO 3)4? 10 1. Titanium nitrate 2. Titanium tetranitrate 3. Titanium (IV) nitrate 4. Titanium (XII) nitrate 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Writing Formulas • Given the name of a compound, the following rules are followed when writing the formula: 1. For ionic compounds, the sum of the charges must equal zero. Subscripts are added as needed in order to follow this rule. 2. For covalent compounds, subscripts are added according to the numerical prefixes given.

Energy of Chemical Bonds • Bond energy- the energy required to break a • • • bond. Bond energy can be used to indicate the strength of a bonding interaction. Ionic bonds form when an atom loses electrons relatively easily to an atom that has a high electron affinity. Ionic compounds result when a metal reacts with a nonmetal.

Coulomb’s Law • Coulomb’s law can be used to calculate the energy of • • • interaction between a pair of ions. E = 2. 31 x 10 -19 J. nm (Q 1 Q 2/ r) E is energy in joules, r is the radius between the ion centers in nanometers, and Q 1 and Q 2 are the numerical ion charges. Example: the distance between Na+ and Cl- ions is 0. 276 nm. What is the ionic energy per pair of ions? -8. 37 x 10 -19 J A negative sign indicates an attractive force. (The ion pair has lower energy than the separated ions and is therefore more stable). Coulomb’s law can also be used to calculate the repulsive force between two like charged ions. In this case, the sign will be positive.

Energy Effects in Binary Ionic Compounds • Lattice energy is the change in energy that takes place when separated gaseous ions are packed together to form an ionic solid. (Energy that is released when an ionic solid forms from its ions). • If energy is released, it represents an exothermic process. (Energy will have a negative sign).

Lattice Energy Calculations • A modified form of Coulomb’s Law can be used to calculate lattice energy. • Lattice energy = k(Q 1 Q 2 / r) • K is a constant that depends on the structure of the solid and electron configuration of the ions, Q 1 and Q 2 are the charges on ions, and r is the distance between the nuclei of the ions.

Which compound has the most 10 exothermic lattice energy? 1. Na. Cl 2. KCl 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Which compound has the most 10 exothermic lattice energy? 1. Li. F 2. Li. Cl 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Which compound has the most 10 exothermic lattice energy? 1. Mg(OH)2 2. Mg. O 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Which compound has the most 10 exothermic lattice energy? 1. Fe(OH)2 2. Fe(OH)3 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Which compound has the most 10 exothermic lattice energy? 1. Na. Cl 2. Na 2 O 1 2 3 4 5 6 7 8 9 10 21 22 23 24 25 26 27 28 29 30 11 12 13 14 15 16 17 18 19 20

Which compound has the most exothermic lattice energy? 10 1. Mg. O 2. Ba. S 1 21 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Which compound has the most 10 exothermic lattice energy? 1. Mg. O 2. Na. F 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Covalent Bonding • Electrons are shared between the nuclei of two atoms. • Polar covalent bonding occurs when there is unequal • • • sharing between the atoms. Unequal sharing results from electronegativity differences between atoms. Electronegativity is the ability of an atom in a molecule to attract shared electrons to itself. Electronegativity increases bottom to top and left to right across the periodic table. The fractional charge that results is called a dipole moment. The dipolar character of a molecule is represented by an arrow pointing to the negative charge center. See page 334 for table of electronegativities.

Which of the following pairs of elements forms a nonpolar covalent bond? 10 1. H-H 2. S-H 3. O-H 4. F-H 5. Cl-H 1 21 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Select the answer choice that lists the pairs of elements in order of increasing polarity. 10 1. H-H, O-H, Cl-H, S-H, F-H 2. F-H, Cl-H, O-H, S-H, H-H 3. H-H, O-H, S-H, Cl-H, F-H 4. H-H, S-H, Cl-H, O-H, F-H 1 21 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Polar and Nonpolar Molecules • A molecule is nonpolar if the dipoles cancel (opposed bond polarities cancel out). • This occurs when the bonds are symmetrically arranged within the molecule. • A molecule is polar if the dipoles do not cancel out.

Which of the following molecules is nonpolar? 10 1. H 2 O 2. CH 4 3. CH 3 Cl 4. HCl 1 21 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Which of the following molecules is polar? 10 1. CO 2 2. CO 3. C 6 H 6 4. O 2 1 21 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20

Types of Covalent Bonds • Single bond-One pair of electrons is shared between atoms • Double bond-two pairs of electrons are shared between atoms • Triple bond-three pairs of electrons are shared between atoms

Bond Energy and Bond Length • Bond energy increases as follows: single bond < double bond < triple bond • Bond length increases as follows: triple bond < double bond < single bond

Bonding Models • Localized Electron Bonding Model • Molecules consist of lone pairs and bonding pairs of electrons VSEPR Model The structure around a given atom is determined principally by minimizing electron pair repulsions. (Bonding and nonbonding pairs are positioned as far apart as possible)

Lewis Structures • The Lewis structure shows how the valence electrons are arranged among the atoms in a molecule. • For the formation of a stable compound, the atoms should achieve noble gas configuration.

Steps for Writing Lewis Structures • Find the total number of electrons needed if each • • • element had a complete octet. (remember hydrogen only needs two valence electrons to be stable) Determine the total number of valence electrons available. Find the difference between what is needed and what is available in order to determine the number of electrons shared. Every two electrons shared represents one bond.

Give the Lewis structure for each of the following. • H 2 O • CH 4 • HCl • NO+ • NH 3 • HCN • CO 2

Exceptions to the Octet Rule • Some compounds form in which one or more • • • atoms will have fewer than 8 electrons in the outer level (incomplete octet) Example: BF 3 Other compounds form in which one atom may have more than 8 electrons in the outer level (Exceeds the octet rule) Example: SF 6

Resonance • Resonance is invoked when more than one valid • • Lewis structure can be written for a molecule. The resulting electron structure of the molecule is given by an average of these resonance structures. This is supported by experimental observations in which the bond lengths and strengths are between those expected if the bonds were single and/or double.

Formal Charge • Molecules that exceed the octet rule can often have many nonequivalent Lewis structures, all which obey the rules for writing Lewis structures. • Through the determination of formal charges, the Lewis structures can be evaluated as to which best describes the bonding in the molecule.

Rules Governing Formal Charge • Determine the sum of the lone pair electrons and one • • • half of the shared electrons. This is the number of valence electrons assigned to the atom. Subtract the number of valence electrons assigned to the atom from the valence electrons on the free , neutral atom. The sum of the formal charges must equal the overall charge on the ion or molecule. If nonequivalent structures exist, those with formal charges closest to zero and with any negative formal charges on the most electronegative atoms are considered the best description of the bonding.
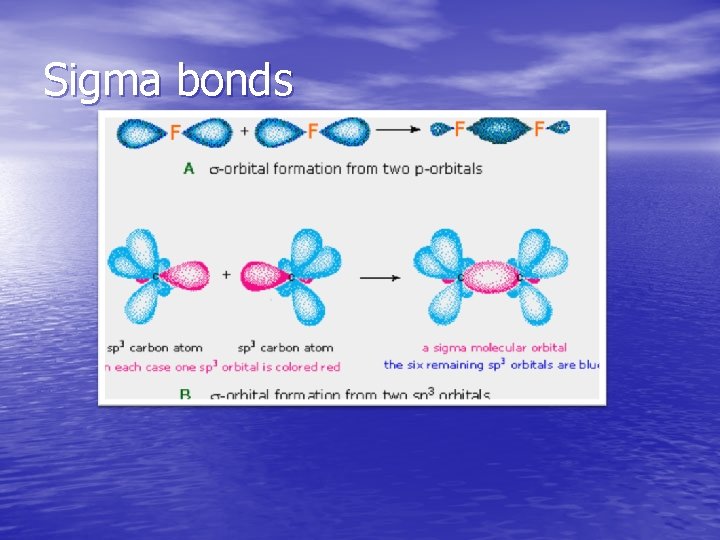
Sigma bonds
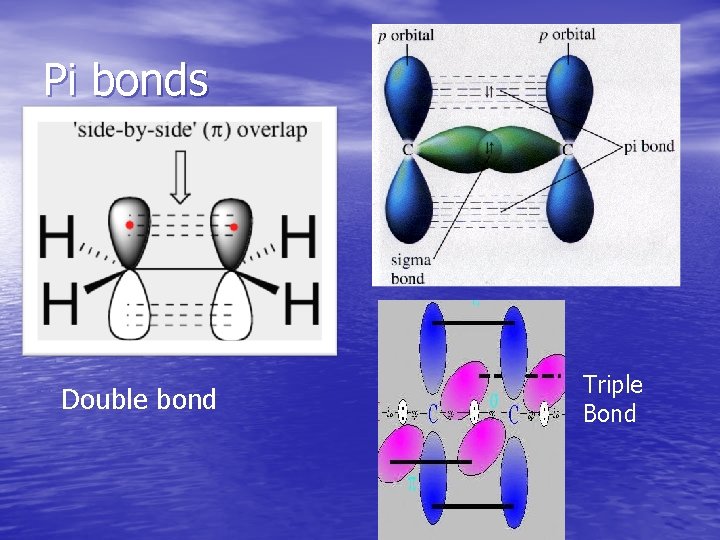
Pi bonds Double bond Triple Bond
- Slides: 56