CHEMICAL BONDING THE IONICBOND MODEL CHEMICAL BONDING The

CHEMICAL BONDING: THE IONICBOND MODEL

CHEMICAL BONDING: The Ionic BOND MODEL 4 -1 Chemical Bonds 4 -2 Valence Electrons and Lewis Symbols 4 -3 The Octet Rule 4 -4 The Ionic Bond Model 4 -5 The Sign and the Magnitude of Ionic Charge 4 -6 Lewis Structure for Ionic Compounds 4 -7 Chemical Formulas for Ionic Compounds 4 -8 The Structure of Ionic Compounds 4 -9 Recognizing and Naming Binary Ionic compounds 4 -10 Polyatomic Ions 4 -11 Chemical Formulas and Names for Ionic Compounds Containing Polyatomic Ions
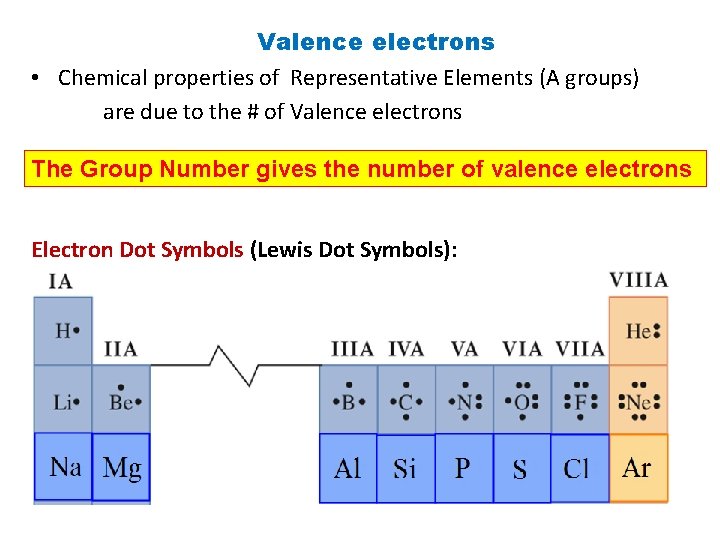
Valence electrons • Chemical properties of Representative Elements (A groups) are due to the # of Valence electrons The Group Number gives the number of valence electrons Electron Dot Symbols (Lewis Dot Symbols):
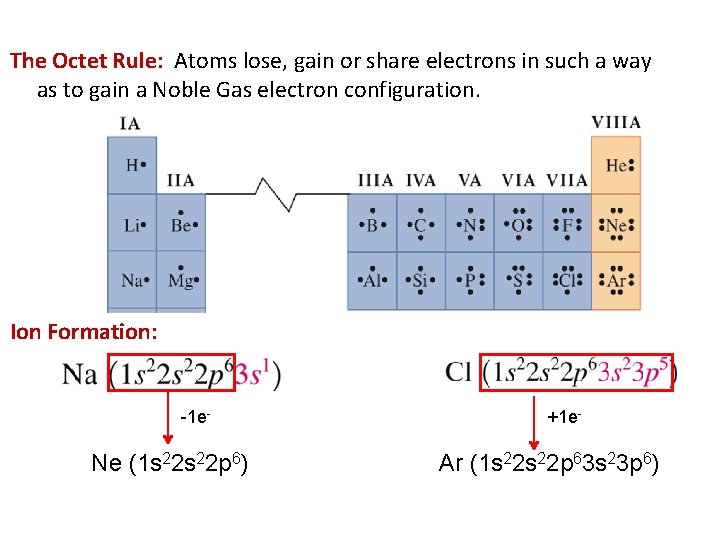
The Octet Rule: Atoms lose, gain or share electrons in such a way as to gain a Noble Gas electron configuration. Ion Formation: -1 e- Ne (1 s 22 p 6) +1 e- Ar (1 s 22 p 63 s 23 p 6)

Isoelectronic Species: Atoms/Ions that share the same electron configuration. Ions that are: Isoelectronic with Neon: Isoelectronic with Argon: Ne 1 s 22 p 6 Ar 1 s 22 p 63 s 23 p 6

IONS • IONS are atoms or groups of atoms with a positive or negative charge. • Taking away an electron from an atom gives a CATION with a positive charge • Adding an electron to an atom gives an ANION with a negative charge. • To tell the difference between an atom and an ion, look to see if there is a charge in the superscript! Examples: Na → Na+ Electron Configuration of Sodium Ion
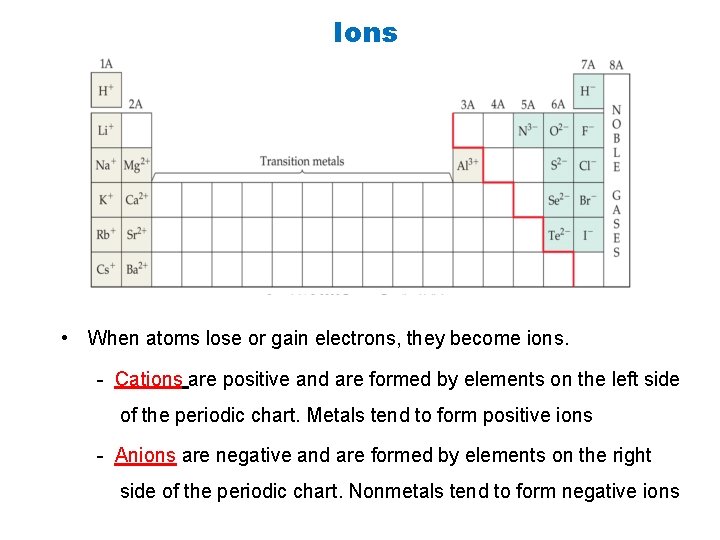
Ions • When atoms lose or gain electrons, they become ions. - Cations are positive and are formed by elements on the left side of the periodic chart. Metals tend to form positive ions - Anions are negative and are formed by elements on the right side of the periodic chart. Nonmetals tend to form negative ions
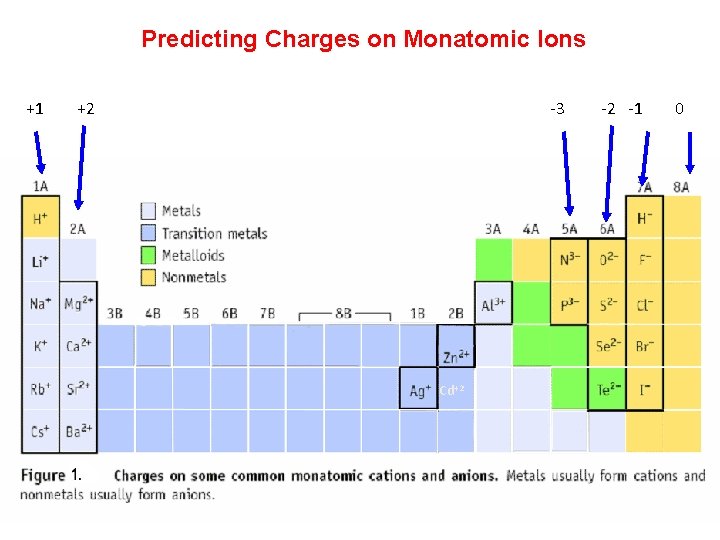
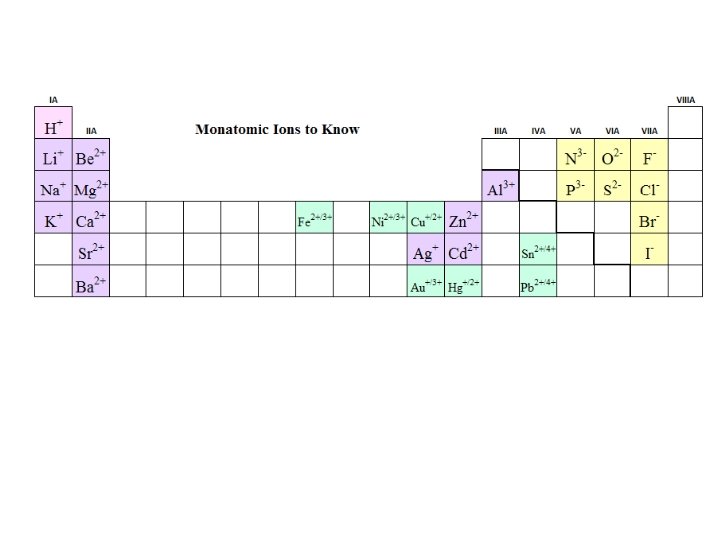
Predicting Charges on Monatomic Ions +1 +2 -3 Cd+2 -2 -1 0
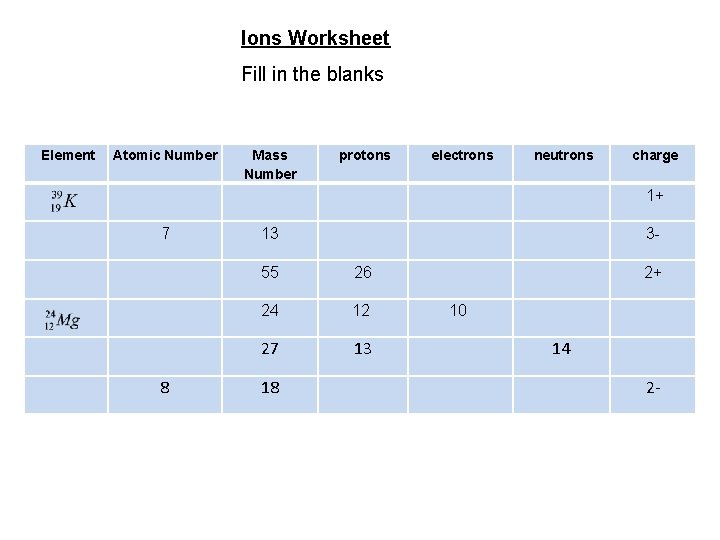
Ions Worksheet Fill in the blanks Element Atomic Number Mass Number protons electrons neutrons charge 1+ 7 8 13 3 - 55 26 24 12 27 13 18 2+ 10 14 2 -
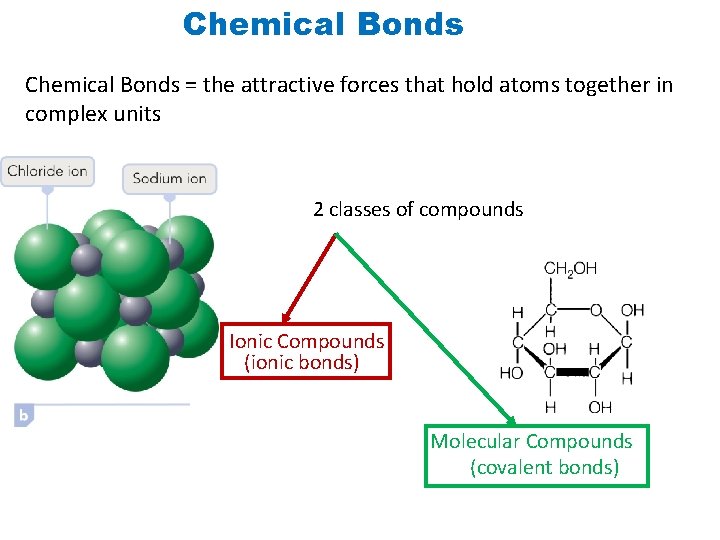
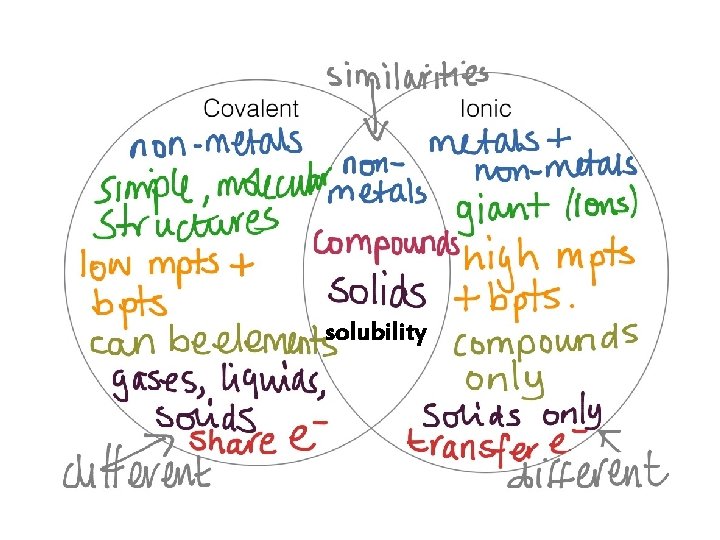
Chemical Bonds = the attractive forces that hold atoms together in complex units 2 classes of compounds Ionic Compounds (ionic bonds) Molecular Compounds (covalent bonds)
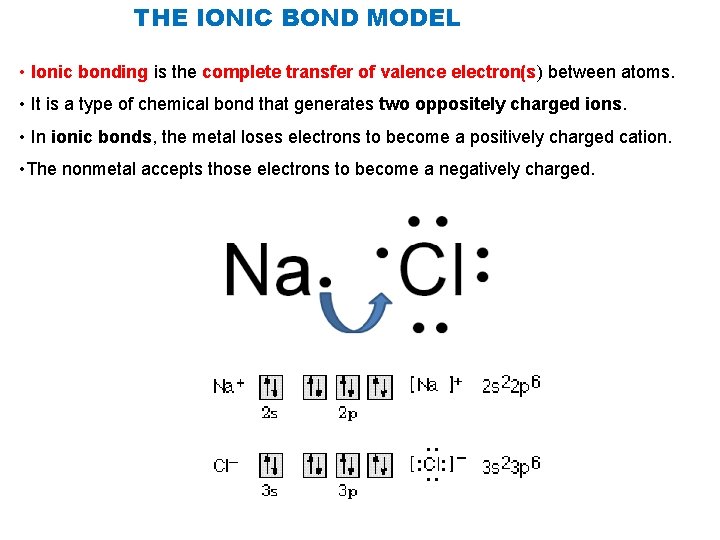
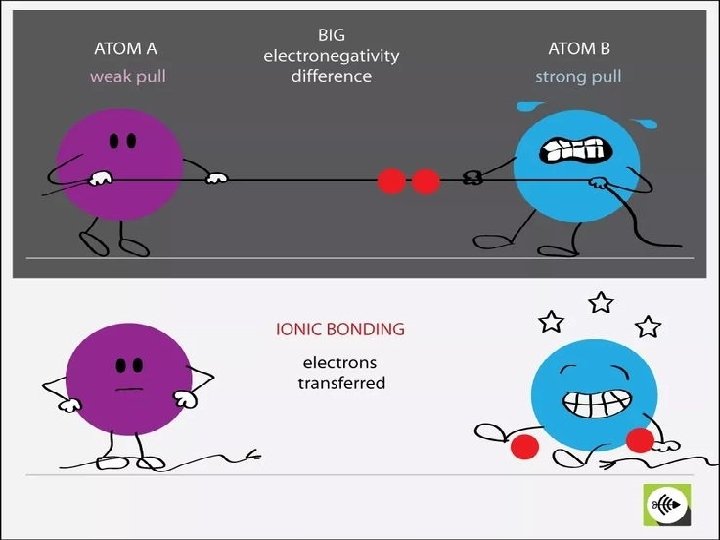
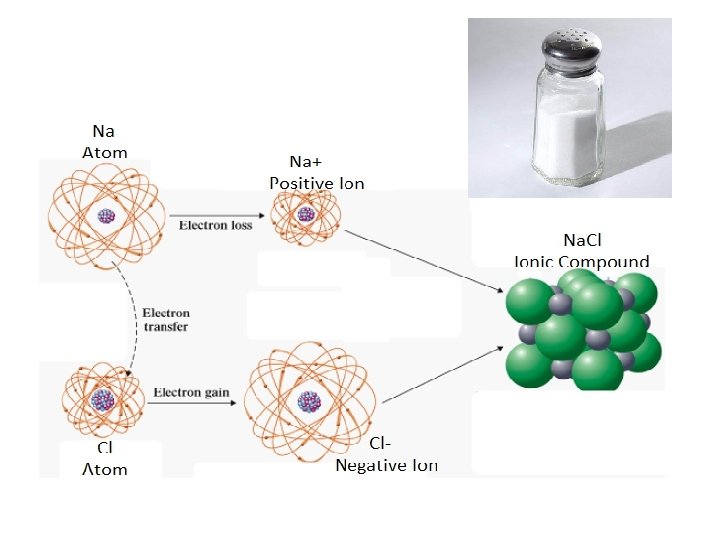
THE IONIC BOND MODEL • Ionic bonding is the complete transfer of valence electron(s) between atoms. • It is a type of chemical bond that generates two oppositely charged ions. • In ionic bonds, the metal loses electrons to become a positively charged cation. • The nonmetal accepts those electrons to become a negatively charged.
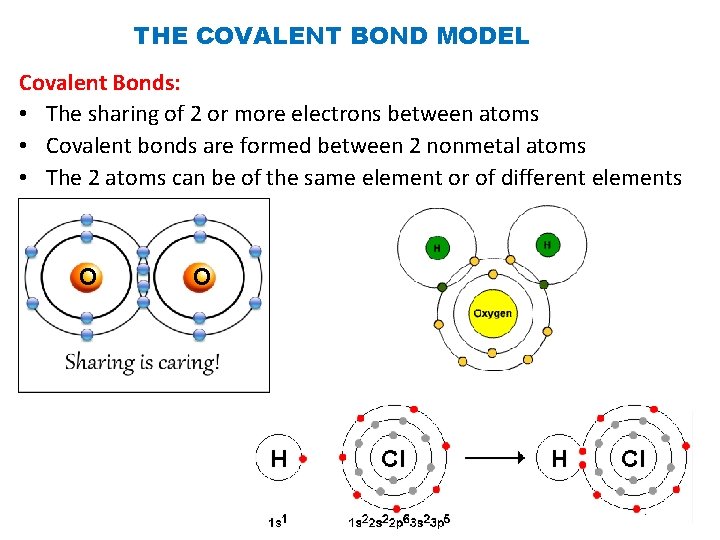
THE COVALENT BOND MODEL Covalent Bonds: • The sharing of 2 or more electrons between atoms • Covalent bonds are formed between 2 nonmetal atoms • The 2 atoms can be of the same element or of different elements O O
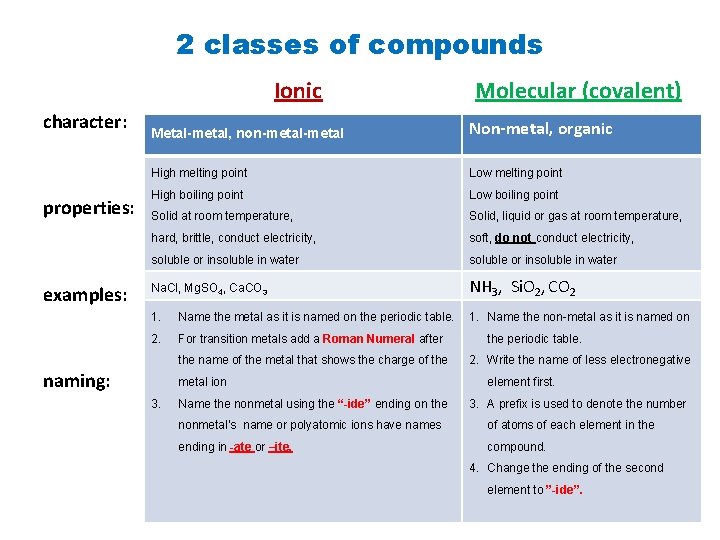
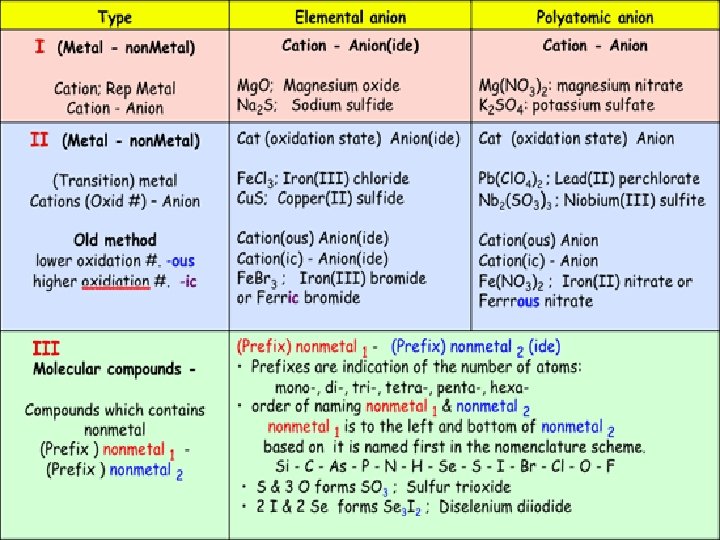
2 classes of compounds Ionic character: properties: examples: Molecular (covalent) Metal-metal, non-metal Non-metal, organic High melting point Low melting point High boiling point Low boiling point Solid at room temperature, Solid, liquid or gas at room temperature, hard, brittle, conduct electricity, soft, do not conduct electricity, soluble or insoluble in water Na. Cl, Mg. SO 4, Ca. CO 3 NH 3, Si. O 2, CO 2 1. Name the metal as it is named on the periodic table. 1. Name the non-metal as it is named on 2. For transition metals add a Roman Numeral after the name of the metal that shows the charge of the naming: metal ion 3. Name the nonmetal using the “-ide” ending on the periodic table. 2. Write the name of less electronegative element first. 3. A prefix is used to denote the number nonmetal’s name or polyatomic ions have names of atoms of each element in the ending in -ate or –ite. compound. 4. Change the ending of the second element to ”-ide”.
solubility

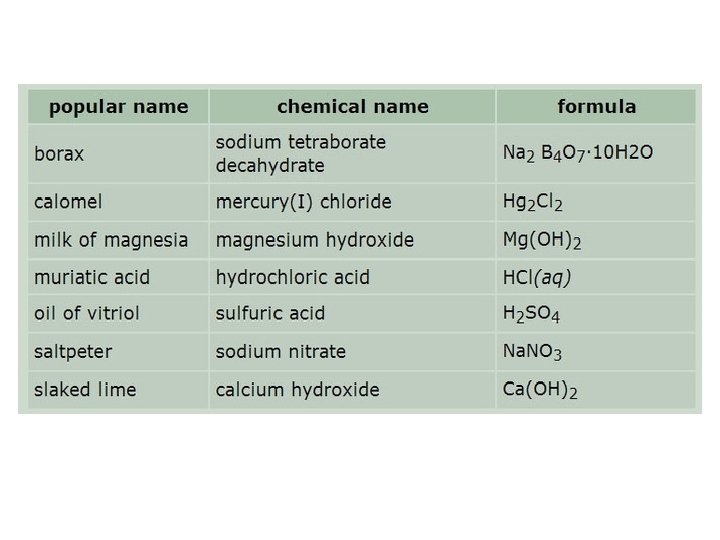
You must memorize the sheet of ions, including the formula, charge, and name of each ion, before you can name compounds successfully or write formulas for them. The best way to do this is make flash cards with the formula of the ion and its charge on one side and the name or names on the other side. This information will be needed for the rest of your chemistry course, so it is important to learn it now. http: //www. quia. com/quiz/1240133. html https: //quizlet. com/6143206/all-24 -polyanions-flash-cards/ http: //chemistry. about. com/od/testsquizzes/l/blionicnamesquiz. htm http: //chemistry. about. com/library/weekly/blcompnamequiz. htm http: //www. chem. purdue. edu/gchelp/nomenclature/simple_ionic. htm

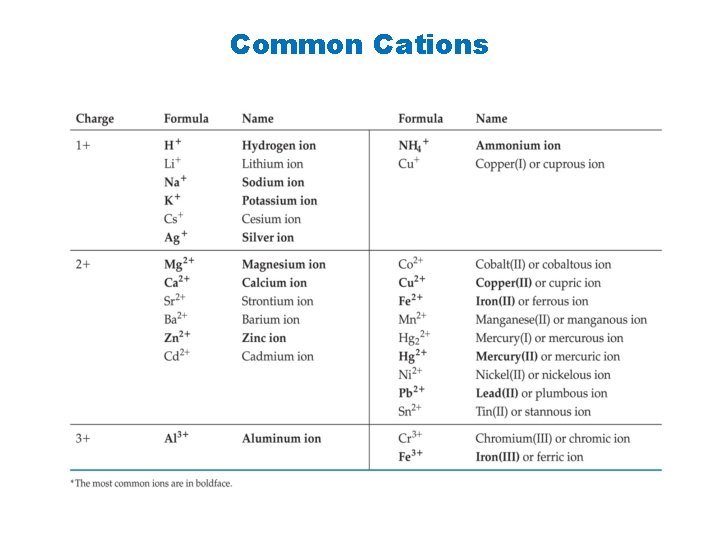
Common Cations
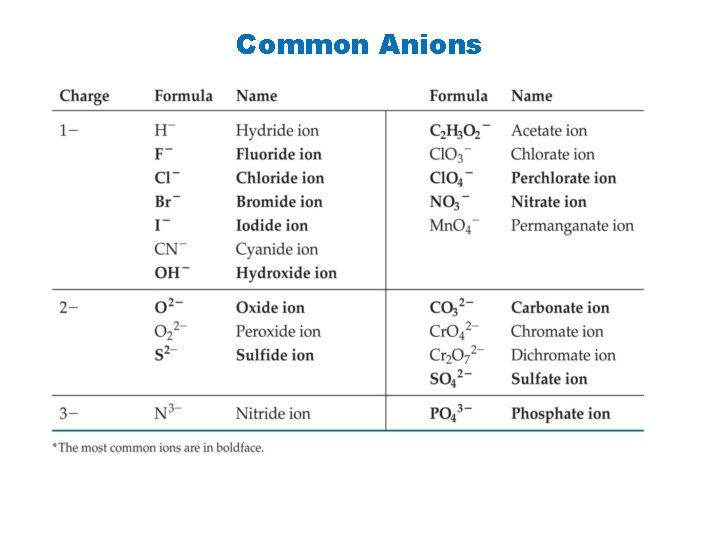
Common Anions

Writing Formulas of Compound from Ions There are three rules to remember when writing chemical formulas for ionic compounds. 1. The symbol for the positive ions (cations, metals) is always written first. 2. The charges on the ions that are present are not shown in formula, however ionic charges must be known to determine the formula. The charges are not explicitly shown in the formula. 3. The numbers in the formula (the subscripts) give the combining ratio for the ions.

Nonmetals named with “ide” ending • • • carbon nitrogen oxygen fluorine phosphorous sulfur chlorine selenium bromine iodine • • • carbide nitride oxide fluoride phosphide sulfide chloride selenide bromide iodide

Writing Formulas • Because compounds are electrically neutral, one can determine the formula of a compound this way: – The charge on the cation becomes the subscript on the anion. – The charge on the anion becomes the subscript on the cation. – If these subscripts are not in the lowest whole-number ratio, divide them by the greatest common factor. Example: Manganese (IV) Oxide.
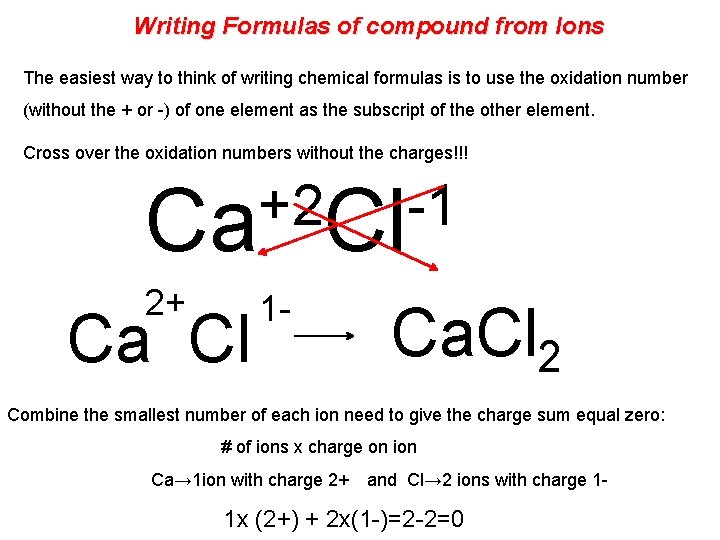
Writing Formulas of compound from Ions The easiest way to think of writing chemical formulas is to use the oxidation number (without the + or -) of one element as the subscript of the other element. Cross over the oxidation numbers without the charges!!! +2 -1 Ca Cl 2+ Ca Cl 1 - Ca. Cl 2 Combine the smallest number of each ion need to give the charge sum equal zero: # of ions x charge on ion Ca→ 1 ion with charge 2+ and Cl→ 2 ions with charge 1 - 1 x (2+) + 2 x(1 -)=2 -2=0

Writing Formulas of Compound from Ions Combine the smallest number of each ion need to give the charge sum equal zero: # of ions x charge on ion Ca→ 1 ion with charge 2+ and NO 3→ 2 ions with charge 1 - 1 x (2+) + 2 x(1 -)=2 -2=0

Writing Formulas of Compound from Ions Combine the smallest number of each ion need to give the charge sum equal zero: # of ions x charge on ion Pb→ 1 ion with charge 4+ and O→ 2 ions with charge 2 - 1 x (4+) + 2 x(2 -)=4 -4=0

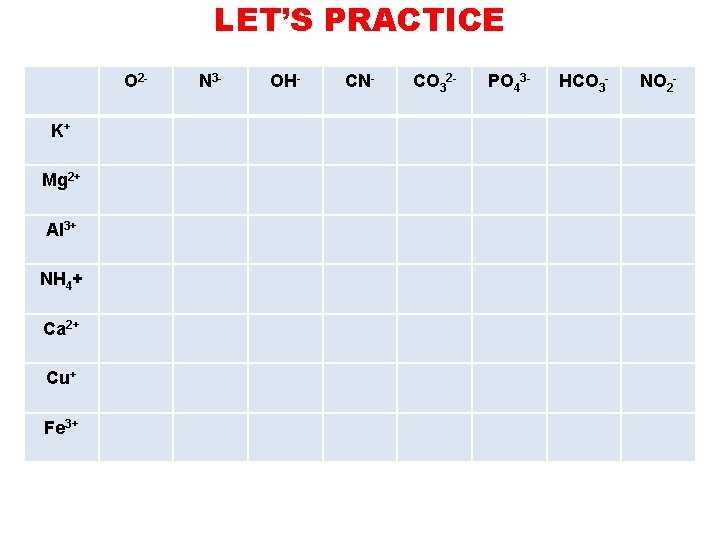
LET’S PRACTICE O 2 K+ Mg 2+ Al 3+ NH 4+ Ca 2+ Cu+ Fe 3+ N 3 - OH- CN- CO 32 - PO 43 - HCO 3 - NO 2 -

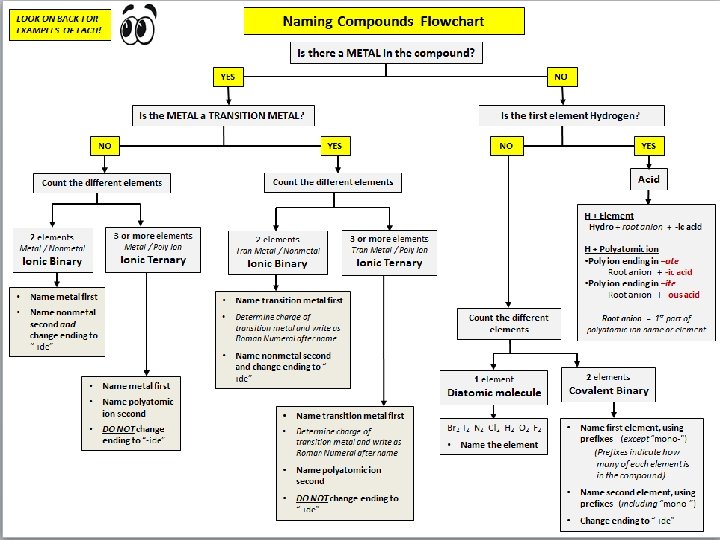
Naming Ionic Compounds • 1. Cation first, then anion • 2. Monatomic cation = name of the element • Ca 2+ = calcium ion • 3. Monatomic anion = root + -ide • Cl- = chloride • Ca. Cl 2 = calcium chloride

Compounds with Transition Metals (Binary) Iron II Sulfide • Step 1: Write Symbols » Fe S • Step 2: Determine charges (roman numeral gives the charge) » Fe+2 S-2 • Step 3: Crisscross charges to become subscripts » Fe. S

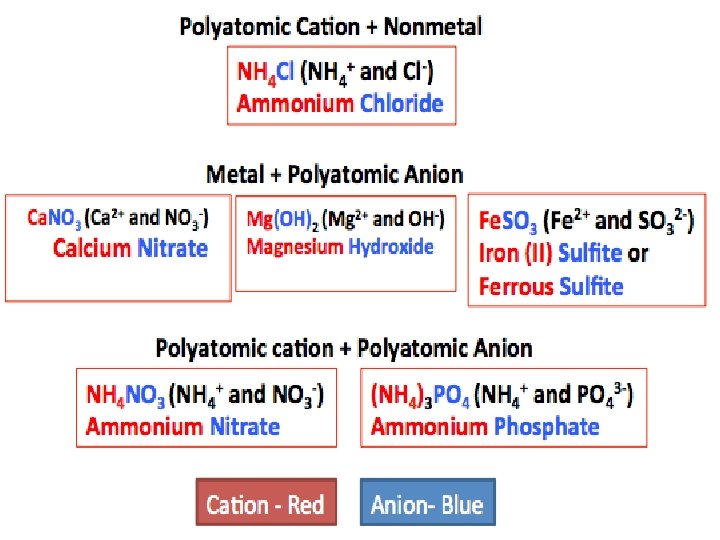
Steps for Naming Ionic Compounds Not a Binary Ionic compound • There are more than 2 elements present • There must be a POLYATOMIC ION in the compound! • Naming compounds with polyatomic ions is similar to naming other ionic compounds • If the Polyatomic is at the end of the compound, – name the metal and then name the Polyatomic Ion • You should note that compounds with polyatomic ions have names ending in -ate or -ite not -ide • Note that most are negative, except ammonium

Steps for Naming Ionic Compounds • Binary Compounds – These compounds contain only two elements – Name the metal as it is named on the periodic table – Name the nonmetal using the “ide” ending on the nonmetal’s name Examples: Na. Cl sodium chloride Zn. I 2 zinc iodide Al 2 O 3 aluminum oxide Na 3 N sodium nitride KBr potassium bromide Al 2 O 3 aluminum oxide Mg. S magnesium sulfide

Nomenclature of binary ionic compounds Some polyatomic anions that you must know: NO 3 - = nitrate NO 2 - = nitrite SO 4 2 - = sulfate SO 32 - = sulfite PO 43 - = phosphate PO 33 - = phosphite CO 32 - = carbonate HCO 31 - = hydrogen carbonate or bicarbonate OH- = hydroxide CN- = cyanide C 2 H 3 O 2 - = acetate C 2 O 42 - = oxalate

Writing Ionic Compound Formulas Ammonium Carbonate • Step 1: Write Symbols » NH 4 CO 3 • Step 2: Determine charges » NH 4+1 CO 3 -2 • Step 3: Crisscross charges to become subscripts » (NH 4)2 CO 3 Combine the smallest number of each ion need to give the charge sum equal zero: # of ions x charge on ion NH 4→ 2 ions with charge 1+ and CO 3→ 1 ion with charge 2 - 2 x (1+) + 1 x(2 -)=2 -2=0

Manganese IV Nitrate • Step 1: Write Symbols » Mn NO 3 • Step 2: Determine charges (roman numeral gives the charge) » Mn+4 NO 3+1 • Step 3: Crisscross charges to become subscripts » Mn(NO 3)4 Combine the smallest number of each ion need to give the charge sum equal zero: # of ions x charge on ion Mn→ 1 ion with charge 4+ and NO 3→ 4 ions with charge 1 - 1 x (4+) + 4 x(1 -) = 4 -4= 0

LET’S PRACTICE Na. Cl Cu. O Sodium Chloride Copper(II) oxide Ammonium chloride Carbon monoxide KBr Potassium Bromide Mg. F 2 (NH 4)Cl CO Pb. Cl 2 Ba(SO 4) CO 2 Sn. Br 4 Ca 3(PO 4)2 N 2 O 5 Lead(II) chloride Magnesium fluoride Al 2 O 3 Aluminium oxide Ag. I Silver iodide Barium sulfate Carbon dioxide Tin(IV) Bromide Calcium phosphate Dinitrogen pentoxide Fe. Cl 3 Iron(III) chloride Mn 2 O 3 Fe(NO 3)3 Iron(III) nitrate Na(OH) PCl 3 Phosphorus trichloride SO 2 Manganese(III) oxide Sodium hydroxide Sulfur dioxide

Transition Metals Many form 2 or more positive ions 1+ 2+ 1+ or 2+ Ag+ silver ion Cd 2+ Zn 2+ Cu+, Cu 2+ cadmium (II)ion copper(I) ion zinc (II)ion copper (II) ion 2+ or 3+ Fe 2+, Fe 3+ iron(II) ion iron(III) ion • Figure out the charge of the metal ion • Add a Roman Numeral after the name of the metal that shows the charge of the metal ion • Examples: Fe. Cl 3 Cu. Cl Sn. F 4 Pb. Cl 2 Fe 2 S 3 (Fe 3+) (Cu+ ) (Sn 4+) (Pb 2+) (Fe 3+) iron (III) chloride copper (I) chloride tin (IV) fluoride lead (II) chloride iron (III) sulfide

Nomenclature of Binary Compounds Non-Metals • The less electronegative atom is usually listed first. • A prefix is used to denote the number of atoms of each element in the compound (mono- is not used on the first element listed, however. )
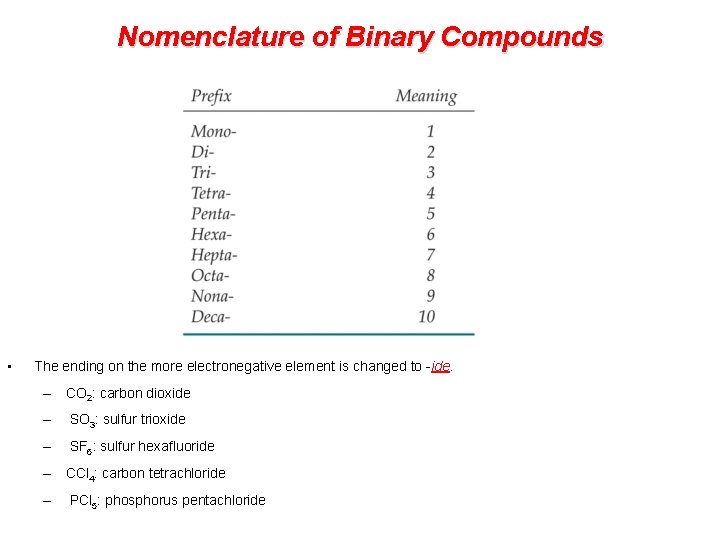
Nomenclature of Binary Compounds • The ending on the more electronegative element is changed to -ide. – CO 2: carbon dioxide – SO 3: sulfur trioxide – SF 6: sulfur hexafluoride – CCl 4: carbon tetrachloride – PCl 5: phosphorus pentachloride
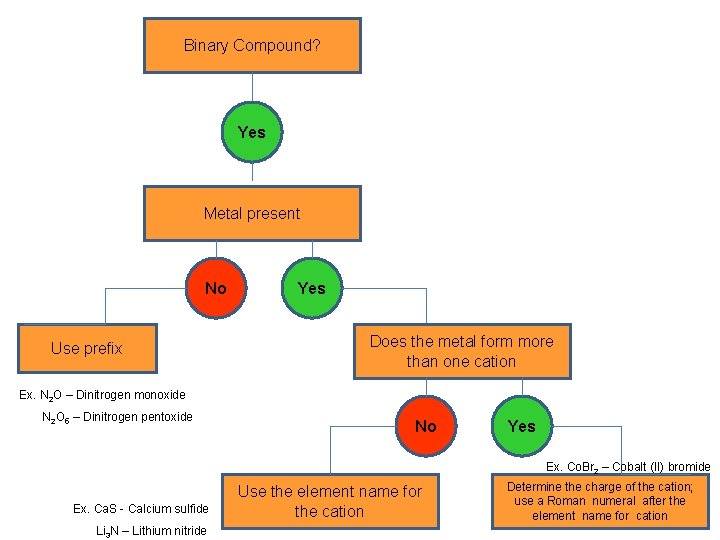
Binary Compound? Yes Metal present No Use prefix Yes Does the metal form more than one cation Ex. N 2 O – Dinitrogen monoxide N 2 O 5 – Dinitrogen pentoxide No Yes Ex. Co. Br 2 – Cobalt (II) bromide Ex. Ca. S - Calcium sulfide Li 3 N – Lithium nitride Use the element name for the cation Determine the charge of the cation; use a Roman numeral after the element name for cation

Patterns in Oxyanion Nomenclature • When there are two oxyanions involving the same element: – The one with fewer oxygens ends in -ite • NO 2− : nitrite; SO 32− : sulfite – The one with more oxygens ends in -ate • NO 3− : nitrate; SO 42− : sulfate

Mixed Practice 1. 2. 3. 4. 5. 6. 7. 8. 9. N 2 O K 2 S Cu(NO 3)2 Cl 2 O 7 Cr 2(SO 4)3 Fe 2(SO 3)3 Ca. O Ba. CO 3 ICl 1. 2. 3. 4. 5. 6. 7. 8. 9. Dinitrogen monoxide Potassium sulfide Copper (II) nitrate Dichlorine heptoxide Chromium (III) sulfate Ferric sulfite Calcium oxide Barium carbonate Iodine monochloride

Mixed Practice 1. 2. 3. 4. 5. 6. 7. 8. 9. Ba. I 2 P 4 S 3 Ca(OH)2 Fe. CO 3 Na 2 Cr 2 O 7 I 2 O 5 Cu(Cl. O 4)2 CS 2 B 2 Cl 4 1. 2. 3. 4. 5. 6. 7. 8. 9. Barium iodide Tetraphosphorus trisulfide Calcium hydroxide Iron (II) carbonate Sodium dichromate Diiodine pentoxide Cupric perchlorate Carbon disulfide Diboron tetrachloride
- Slides: 49