Chemical Bonding Chapter 8 Chemical Bonding Structure Molecular
Chemical Bonding Chapter 8

Chemical Bonding & Structure Molecular bonding and structure play the central role in determining the course of chemical reactions.

Bonds Forces that hold groups of atoms together and make them function as a unit.

Bond Energy - It is the energy required to break a bond. - It gives us information about the strength of a bonding interaction. - The stronger the bond, the higher the bond energy.

Bond Energies Bond breaking requires energy (endothermic). Bond formation releases energy (exothermic).
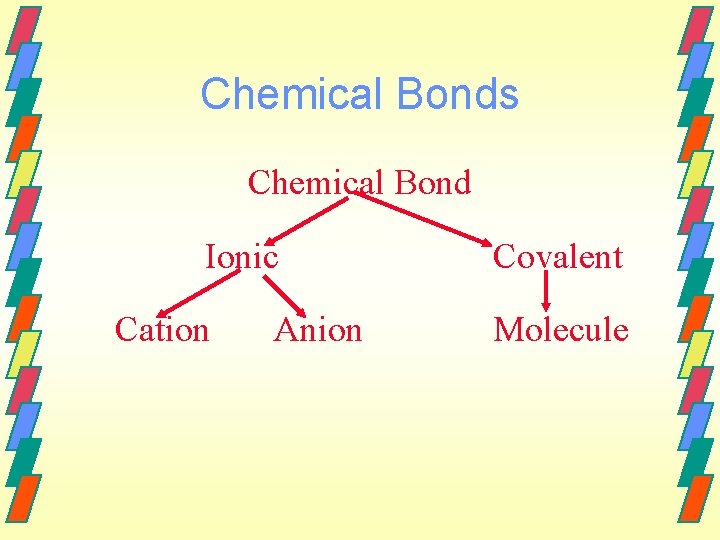
Chemical Bonds Chemical Bond Ionic Cation Anion Covalent Molecule

Ionic Bonds - Formed from electrostatic attractions of closely packed, oppositely charged ions. - Formed when an atom that easily loses electrons (metal) reacts with one that has a high electron affinity(nonmetal). - 2 Na(s) + Cl 2(g) ----> 2 Na+(aq) + 2 Cl-(aq)
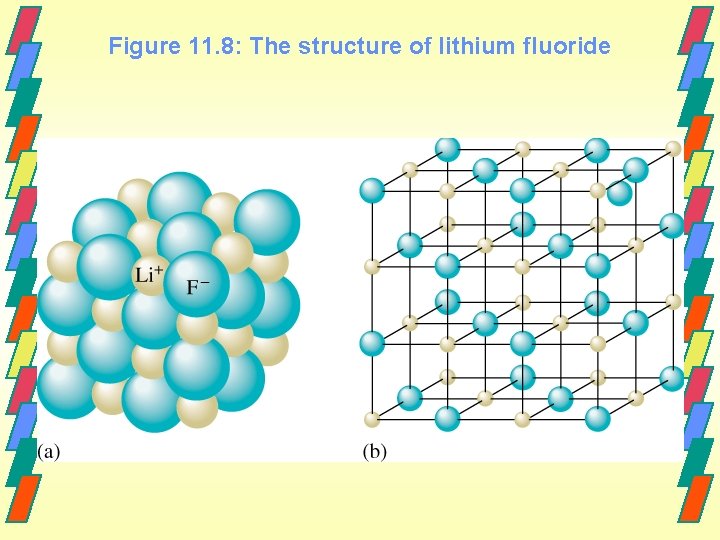
Figure 11. 8: The structure of lithium fluoride
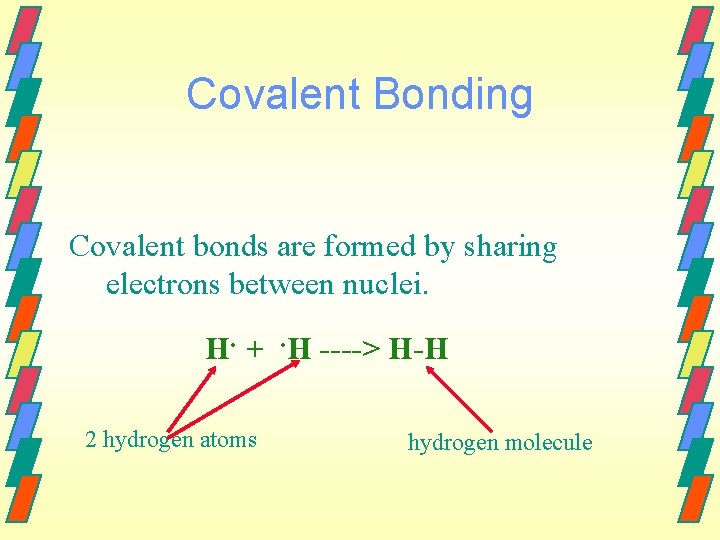
Covalent Bonding Covalent bonds are formed by sharing electrons between nuclei. H. +. H ----> H-H 2 hydrogen atoms hydrogen molecule
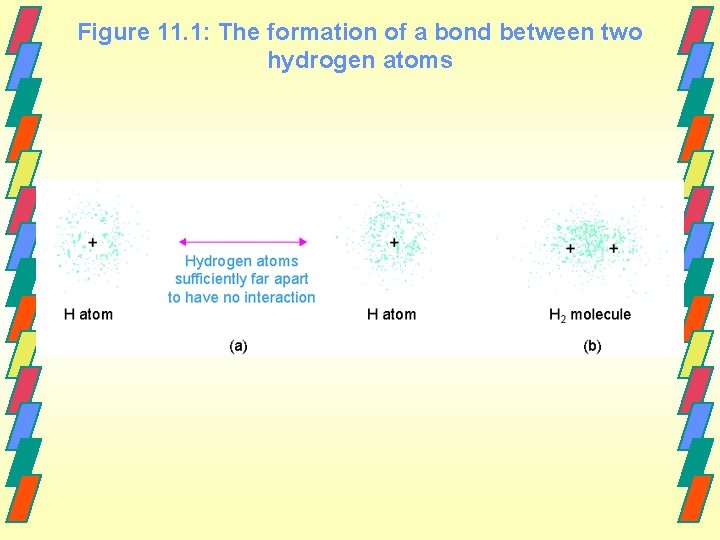
Figure 11. 1: The formation of a bond between two hydrogen atoms

Types of Covalent Bonds Polar covalent bond -- covalent bond in which the electrons are not shared equally because one atom attracts them more strongly than the other. A dipole moment exists. HOH, HCl, & CO Nonpolar covalent bond -- covalent bond in which the electrons are shared equally between both atoms. No dipole moment exists. CO 2, CH 4, & Cl 2

Electronegativity The ability of an atom in a molecule to attract shared electrons to itself. As electronegativity increases, the attraction for electrons increases. Fluorine has the highest value at 4. 0 and cesium and francium are lowest at 0. 7.

Pauling Electronegativity Values

Electronegativity values for selected elements. See Figure 8. 3 on page 334 in Zumdahl.

Homework #24 a. Rb < K < Na b. Ga < B < O c. Br < Cl < F d. S < O < F

Three Possible Types of Bonds Nonpolar Covalent (Electrons equally shared. ) Polar Covalent (Electrons shared unequally. ) Ionic (Electrons are transferred. )

Percent Ionic Character where x. A is the larger electronegativity and x. B is the smaller value. Watch significant figures!!! Ionic Bond Polar Covalent Nonpolar Covalent % IC > 50 % % IC 5 - 50 % % IC < 5 %

Percent Ionic Character What type of bonding & % ionic character does KCl have? Ionic

Percent Ionic Character What type of bonding & % ionic character does HOH have? Polar covalent

Percent Ionic Character What type of bonding & % ionic character does N 2 have? Nonpolar covalent
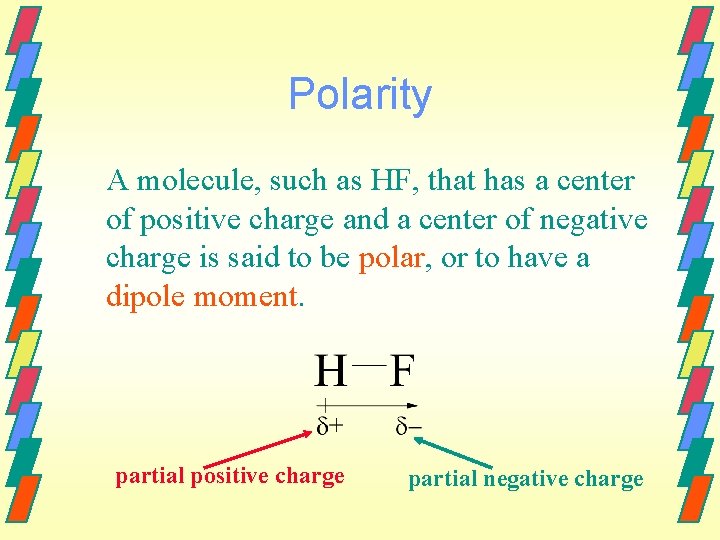
Polarity A molecule, such as HF, that has a center of positive charge and a center of negative charge is said to be polar, or to have a dipole moment. partial positive charge partial negative charge

Figure 11. 2: Probability representations of the electron sharing in HF

The Effect of an electric field on hydrogen fluoride molecules.

Dipole Moment for the water molecule.

Polar Water Molecule The polarity of water allows it to dissolve ionic materials which are essential for life. The polarity of the water molecule allows water molecules to attract each other strongly (hydrogen bonds). Because of this fact water remains as a liquid at room temperatures and allows the existence of life as we know it.

Dipole moment for the ammonia molecule.

Homework #30 a. b. P H is a pure covalent (nonpolar) bond since P and H have identical electronegativities. c. d. e.

Nonpolar molecule--zero dipole moment.

Cation Size Cations are always smaller than the parent atom because they have lost an entire electron shell. As well, the number of protons is greater than the number of electrons so the electrons are held tighter.

Anion Size Anions are always larger than the parent atom because they have added electrons which repel each other. As well, the number of protons is less than the number of electrons so they are not held as tightly.
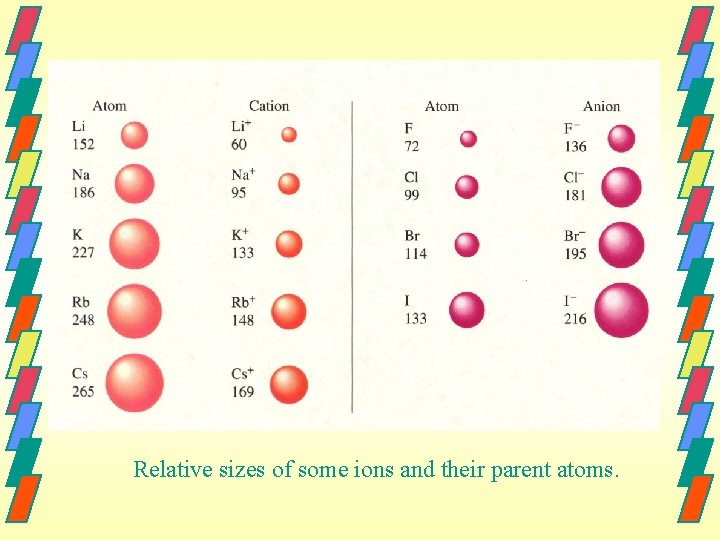
Relative sizes of some ions and their parent atoms.

Energy Changes & Chemical Bonding Most chemical reactions can be explained in terms of the rearrangements of bonds. • Energy must be added to break existing bonds in the reacting substances. • Energy is released when new bonds are formed in the products. • For the reactions of covalent compounds, the net energy of the reaction, ∆Hrxn, = (sum of energy added to break bonds (reactants) - (sum of energy released when bonds form(products).

Energy Changes & Chemical Bonding Consider the reaction for the formation of water from elemental hydrogen and oxygen, 2 H 2(g) + O 2(g) ---> 2 H 2 O(g). • First, write the Lewis dot structures for the reactants • and products, and determine which bonds must be broken and which bonds must be formed. Then use the average bond energy values found in Table 8. 4 p 351 of the book. to calculate the energy needed to break the bonds in the reactants and the energy released when the bonds form in the products.

Energy Changes & Chemical Bonding 2 H (g) + O (g) ---> 2 H O(g). 2 2 2 • To break the H-H bond requires 432 k. J/mole H 2(g). • • Since there are two moles of H 2(g) in the reaction, this value is doubled to 864 k. J. The oxygen atoms in the O 2(g) are held together by a double bond which requires 495 k. J/mole to break. For each water molecule produced, there are two O-H bonds formed. For the 2 H 2 O(g), the total energy released when four moles of O-H bonds form is: 4 moles O-H x 467 k. J/mole = 1868 k. J.

Energy Changes & Chemical Bonding 2 H (g) + O (g) ---> 2 H O(g). 2 2 2 • Therefore, the ∆Hrxn = (864 k. J to break H-H bonds + 495 k. J to break O=O bonds) – (1868 k. J released when O-H bonds formed) = -509 k. J. • The negative value indicates that the reaction releases energy and is exothermic (Note that bond energies are average values, values and can vary depending upon what other elements are bonded to the atoms which are being broken apart. )
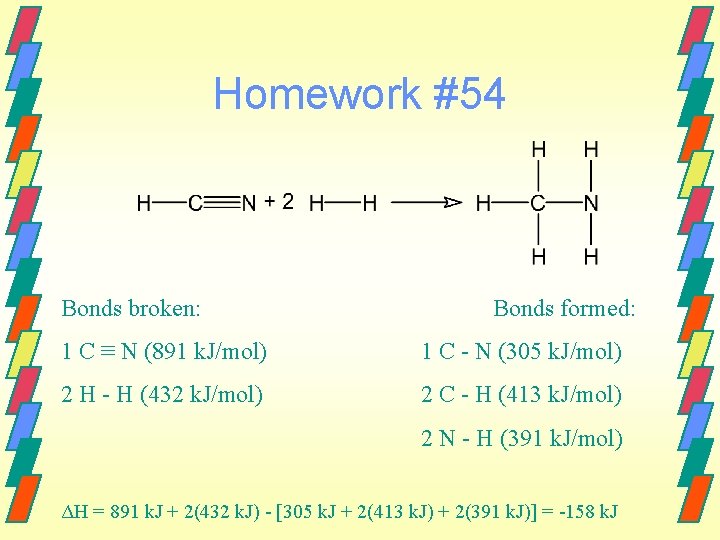
Homework #54 Bonds broken: Bonds formed: 1 C ≡ N (891 k. J/mol) 1 C - N (305 k. J/mol) 2 H - H (432 k. J/mol) 2 C - H (413 k. J/mol) 2 N - H (391 k. J/mol) ΔH = 891 k. J + 2(432 k. J) - [305 k. J + 2(413 k. J) + 2(391 k. J)] = -158 k. J
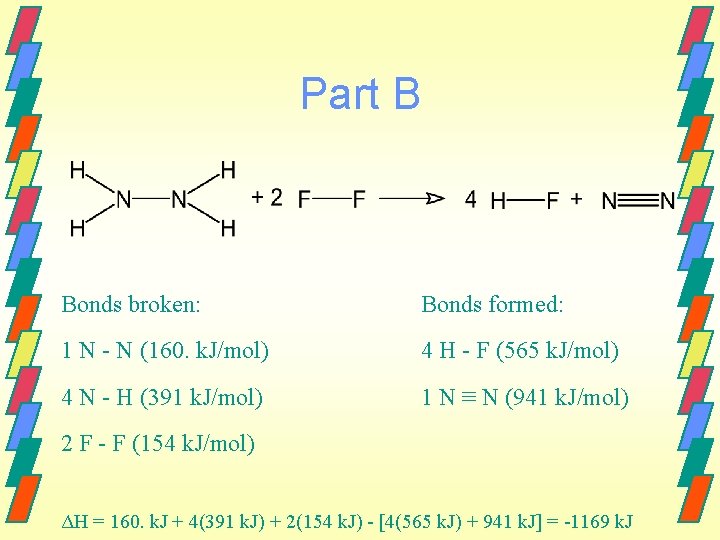
Part B Bonds broken: Bonds formed: 1 N - N (160. k. J/mol) 4 H - F (565 k. J/mol) 4 N - H (391 k. J/mol) 1 N ≡ N (941 k. J/mol) 2 F - F (154 k. J/mol) ΔH = 160. k. J + 4(391 k. J) + 2(154 k. J) - [4(565 k. J) + 941 k. J] = -1169 k. J

Energy Changes & Chemical Bonding For the formation of ionic compounds, compounds the ∆Hrxn is based upon all of the energy changes involved in the transfer of electrons between the metal and nonmetal. Consider the reaction for the formation of sodium chloride from elemental sodium and chlorine, 2 Na(s) + Cl 2(g) ---> 2 Na. Cl(s).

Energy Changes & Chemical Bonding • Ionization energy must be added to completely • • • remove electrons from the metal in the gaseous state. Since most metals are solids at room temperature, energy must be added to vaporize the metal before this ionization occurs. This energy value is called the heat of formation, formation ∆Hºf. Then energy is released when the nonmetal gains electrons, as measured by the electron affinity value. Energy is also released when the ions bond to form a crystal lattice structure, called the lattice energy

Ionic Compounds 2 Na(s) + Cl 2(g) ---> 2 Na. Cl(s) 2 Na(s) ---> 2 Na(g) add ∆Hºf = (108 k. J/mole Na x 2 moles Na) = 216 k. J 2 Na(g) ---> 2 Na 1+(g) + 2 e 1 add ionization energy = (496 k. J/mole Na x 2 moles Na) = 992 k. J Cl 2(g) ---> 2 Cl(g) add bond energy = 243 k. J/mole Cl 2 x 1 mole Cl 2) = 243 k. J 2 Cl(g) + 2 e 1 - ---> 2 Cl 1 -(g) release electron affinity = 349 k. J/mole Cl 1 - x 2 moles Cl 1 - = -698 k. J 2 Na 1+(g) + 2 Cl 1 -(g) ---> 2 Na. Cl(s) release lattice energy = 788 k. J/mole Na. Cl x 2 moles Na. Cl = -1576 k. J ∆Hrxn = 216 k. J + 992 k. J + 243 Kj + -698 k. J + -1576 k. J = -823 k. J per mole of reaction

Homework #50 50. Let us look at the complete cycle for Na 2 S. 2 Na(s) → 2 Na(g) 2 ΔHsub, Na = 2(109) k. J 2 Na(g) → 2 Na+(g) + 2 e 2 IE = 2(495) k. J S(s) → S(g) ΔHsub, S = 277 k. J S(g) + e → S (g) EA 1 = -200. k. J S-(g) + e → S 2 (g) EA 2 = ? 2 Na+(g) + S 2 (g) → Na 2 S LE = -2203 k. J ___________________________ 2 Na(s) + S(s) → Na 2 S(s) ΔH= 2 ΔH= -365 k. J + 2 IE + ΔHsub. S + EA 1 + EA 2 + LE, -365 = -918 + EA 2, EA 2 = 553 k. J For each salt: ΔH= 2 ΔHsub, M + 2 IE + 277 - 200. + LE + EA 2

# 50 continued K 2 S: -381 = 2(90. ) + 2(419) + 277 - 200. - 2052 + EA 2, EA 2 = 576 k. J Rb 2 S: -361 = 2(82) + 2(409) + 277 - 200. - 1949 + EA 2, EA 2 = 529 k. J Cs 2 S: -360. = 2(78) + 2(382) + 277 - 200. - 1850. + EA 2, EA 2 = 493 k. J We get values from 493 to 576 k. J. The mean value is: = 538 k. J

Energy Changes & Chemical Bonding The accepted values for the various energy changes that occur during bonding reactions have been carefully calculated using calorimeter experiments. In your textbook, • Ionization energies for some of the elements can be • • found in Table 7. 5 p. 310 & Figure 7. 6 p. 310. Electron affinity values are listed in Figure 7. 7, page 313. Lattice energies for ionic crystals are found in the problems or given data Values for the enthalpy of formation, formation ∆Hºf, are listed in Figure Appendix 4 pp. A 19 -A 22. Values for the enthalpy of vaporization, vaporization ∆Hºvap, are listed in The problems or given appendix.

Achieving Noble Gas Electron Configurations (NGEC) Two nonmetals react: They share electrons to achieve NGEC. A nonmetal and a representative group metal react (ionic compound): The valence orbitals of the metal are emptied to achieve NGEC. The valence electron configuration of the nonmetal achieves NGEC.

Noble Gas Configuration When a Group I, II, or III metal reacts with a nonmetal to form a binary ionic compound, the nonmetal gains electrons to obtain the configuration of the next noble gas. The metal loses electrons to gain the configuration of the previous noble gas. Na ----> Na+ + e- configuration of Ne Cl + e- ----> Cl- configuration of Ar

Lewis Structure - Shows how valence electrons are arranged among atoms in a molecule. - Reflects central idea that stability of a compound relates to noble gas electron configuration. - Developed by G. N. Lewis in 1902.
![Lewis Structures Na. sodium atom sulfur atom [Na]+ sodium ion [ ] sulfide ion Lewis Structures Na. sodium atom sulfur atom [Na]+ sodium ion [ ] sulfide ion](http://slidetodoc.com/presentation_image_h2/61c0063b4eca9ebcd12a7033dc3d588c/image-48.jpg)
Lewis Structures Na. sodium atom sulfur atom [Na]+ sodium ion [ ] sulfide ion

Drawing Lewis structures • Write the electron dot diagrams for each element in the compound. • Check the electronegativity difference between the elements to determine if electrons are transferred or shared. • If the electronegativity difference > 1. 67, the reaction forms ions. Remove the electrons from the metal and add them to the nonmetal

Drawing Lewis Structures Write the charges of the ions formed and use coefficients to show many of each ion are needed to balance the overall charge. + 2 - 2 Na , [ O ] Ionic sodium oxide

Drawing Lewis structures • If the electronegativity difference < 1. 67, then the atoms will share electrons. • Position shared electron pairs between the two atoms, and connect them with a single line to represent a covalent bond. • Place the extra pairs of electrons around atoms until each has eight • (Exception: For hydrogen or metallic elements use only the valence electrons that are available, so these atoms have less than an octet. )

Drawing Lewis structures • If an atom other than hydrogen or a metal has less than eight electrons, move unshared pairs to form multiple bonds • Add extra atoms, atoms if needed, to obtain the octets Atoms with positive oxidation numbers should be bonded to those with negative oxidation numbers • If extra electrons still remain, add them to the central atom. All oxidation numbers should add up to zero for a compound.

Lewis structures Example CO 2 Step 1 C-O-O O-C-O – Draw any possible structures You may want to use lines for bonds. Each line represents 2 electrons.

Lewis structures Step 2 – Determine the total number of valence electrons. – CO 2 1 carbon x 4 electrons = 4 2 oxygen x 6 electrons = 12 Total electrons = 16

Lewis structures Try the C-O-O structure C O O No matter what you try, there is no way satisfy the octet for all of the atoms.
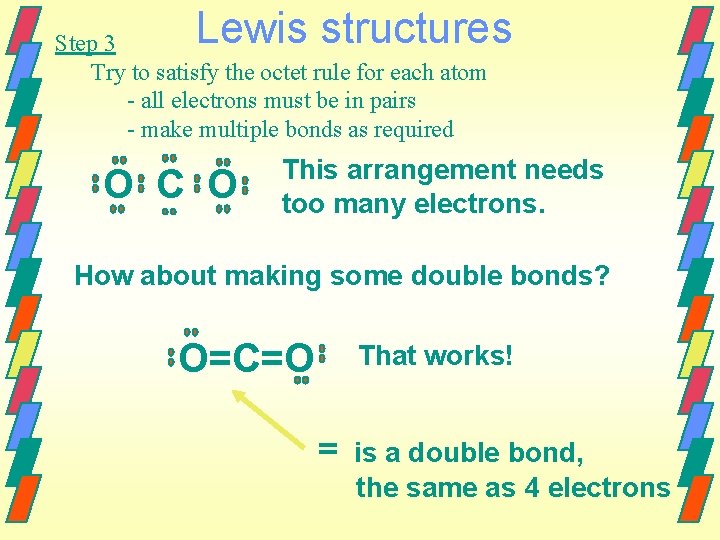
Lewis structures Step 3 Try to satisfy the octet rule for each atom - all electrons must be in pairs - make multiple bonds as required O C O This arrangement needs too many electrons. How about making some double bonds? O=C=O That works! = is a double bond, the same as 4 electrons

Step 1 Ammonia, NH 3 H H N H Step 2 3 e- from H 5 e- from N 8 e- total N has octet H has 2 electrons (all it can hold)

Lewis Structures NO+ • 5 e- + 6 e- - 1 e- = 10 e- • [: N O: ]+ • Each atom has an octet and is satisfied.

Are there any other ways? Yes, there is a simple calculation involving the number of shared electrons and number of bonding sites that tells you what kind of bonding is going on. Let S = N-A S= # of e¯ that need to be shared N=needed e¯ (either 2 or 8) A= available valence e¯ B=regions with e¯ Then S/2/B= kind of bonding

Caveat • Note this can be very helpful when it works (when the octet rule is satisfied) and really misleading when it doesn’t. I personally never use it. You might find it a good way to get started on learning this material when you are in doubt about resonance or number of double and triple bonds.

Examples S/2/B= 1 CCl 4 S= 40 -32=8 S/2/B= 8/2/4= 1 Single Bonds around C

Double bond example S/2/B=2 CO 2 Double bonds S=24 -16=8 S/2/B=8/2/2= 2 Double bonds around C

Greater Than 1= Resonance S/2/B=fraction greater than 1 SO 2 S=24 -18=6 S/2/B= 6/2/2=3/2 3 e¯ pairs shared between two bonding regions

Less Than one = Multiple around central atom S/2/B<1 IBr 2 S=24 -22 S/2/B=2/1/2=1/2 So need expanded for 5 e¯ pairs

Single, Double, & Triple Bonds Single bonds -- one shared pair of electrons. Double bonds -- two shared pairs of electrons. Triple bonds -- three shared pairs of electrons.

• Bond Strength = Triple > Double > Single –For bonds between same atoms, C N > C=N > C—N –Though Double not 2 x the strength of Single and Triple not 3 x the strength of Single • Bond Length = Single > Double > Triple –For bonds between same atoms, C—N > C=N > C N

Comments About the Octet Rule - 2 nd row elements C, N, O, F observe the octet rule. - 2 nd row elements B and Be often have fewer than 8 electrons around themselves - they are very reactive. - 3 rd row and heavier elements CAN exceed the octet rule using empty valence d orbitals. - When writing Lewis structures, satisfy octets first, then place electrons around elements having available d orbitals.

Four Failures of Lewis Structures cannot adequately explain: 1. electron-deficient molecules. 2. the paramagnetism of oxygen and other similar substances. 3. odd-electron molecules. 4. resonance.

Atoms with fewer than eight electrons Beryllium and boron will both form compounds where they have less than 8 electrons around them. : : . . : Cl Be Cl: . . . : F. . B F: . . : F: . .

Atoms with fewer than eight electrons Electron deficient. Species other than hydrogen and helium that have fewer than 8 valence electrons. They are typically very reactive species. Coordinate covalent bond forms when N atom donates both shared e. F H | | F - B + : N - H F-B-N-H | | F H BF 3 is called a Lewis acid because it accepts a pair of electrons and NH 3 is a Lewis base because it donates a pair of electrons.

Atoms with more than eight electrons Except for species that contain hydrogen, this is the most common type of exception. For elements in the third period and beyond, the d orbitals can become involved in bonding. Examples 5 electron pairs around P in PF 5 5 electron pairs around S in SF 4 6 electron pairs around S in SF 6

An example: SF 4 1. Write a possible – arrangement. F F S F F 2. Total the electrons. • 6 from S, 4 x 7 from F – total = 34 3. Spread the electrons – around. F | F - S- F | F

Homework # 68 a. POCl 3 has 5 + 6 + 3(7) = 32 valence electrons. This structure uses all 32 e while satisfying the octet rule for all atoms. This is a valid Lewis structure. SO 42 =32 valence electrons.
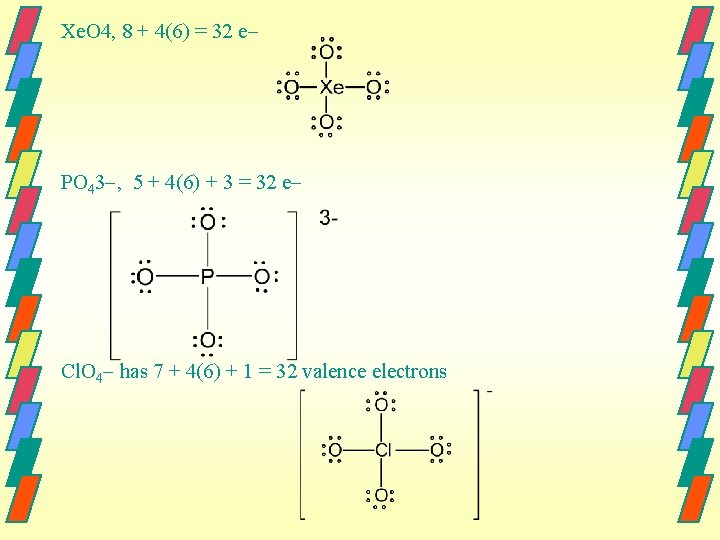
Xe. O 4, 8 + 4(6) = 32 e PO 43 , 5 + 4(6) + 3 = 32 e Cl. O 4 has 7 + 4(6) + 1 = 32 valence electrons
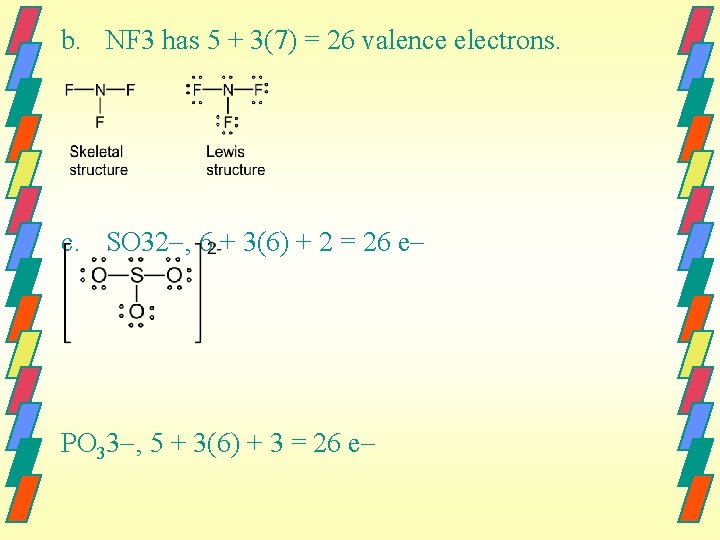
b. NF 3 has 5 + 3(7) = 26 valence electrons. c. SO 32 , 6 + 3(6) + 2 = 26 e PO 33 , 5 + 3(6) + 3 = 26 e

Cl. O 3 , 7 + 3(6) + 1 = 26 e c. Cl. O 2 has 7 + 2(6) + 1 = 20 valence SCl 2, 6 + 2(7) = 20 e PCl 2 , 5 + 2(7) + 1 = 20 e

d. Molecules ions that have the same number of valence electrons and the same number of atoms will have similar Lewis structures.

Odd-Electron Molecules NO 2 • contains 17 electrons. • cannot satisfy the octet rule. • a more sophisticated model is neededthe molecular orbital model.

Species with an odd total number of electrons A very few species exist where the total number of valence electrons is an odd number. This must mean that there is an unpaired electron which is usually very reactive. Radical - a species that has one or more unpaired electrons. They are believed to play significant roles in aging and cancer.

Species with an odd total number of electrons Example - NO Nitrogen monoxide is an example of a compound with an odd number of electrons. It is also known as nitric oxide. It has a total of 11 valence electrons: six from oxygen and 5 from nitrogen. : . : N O: The best Lewis structure for NO is:
Resonance Occurs when more than one valid Lewis structure can be written for a particular molecule. These are resonance structures. The actual structure is an average of the resonance structures called a resonance hybrid.

Resonance structures Sometimes we can have two or more equivalent Lewis structures for a molecule. O-S=O O=S–O They both - satisfy the octet rule - have the same number of bonds - have the same types of bonds Which is right?
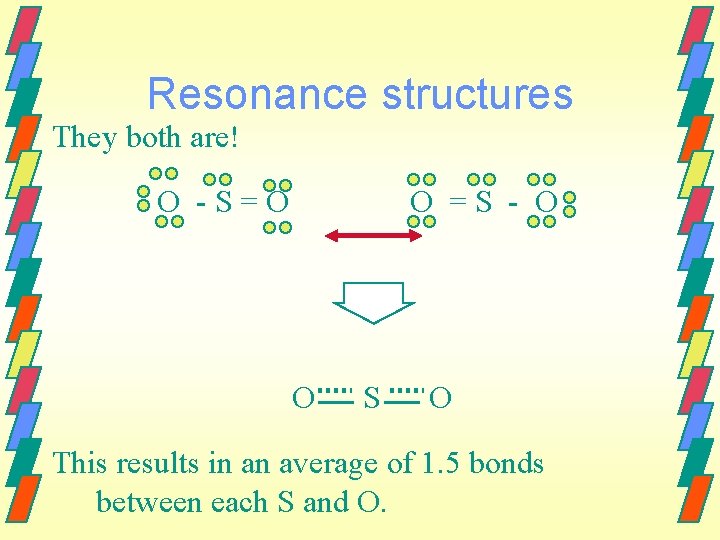
Resonance structures They both are! O -S=O O =S - O O S O This results in an average of 1. 5 bonds between each S and O.

Resonance structures Benzene, C 6 H 6, is an example of a compound for which resonance structures must be written. At each corner of the hexagonal ring, there is a carbon atom with a double bond to one C and a single bond to another C and to an H atom. All of the bonds are the same length. or

Stereochemistry The study of the threedimensional arrangement (molecular structure) of atoms or groups of atoms within molecules and the properties which follow such arrangement.

Formal Charges • A bookkeeping system for electrons that is used to predict which possible Lewis structure is more likely. • They are used to show the approximate distribution of electron density in a molecule or polyatomic ion.

Rules for Formal Charges 1. Assign each atom half of the electrons in each pair it shares. 2. Also give each atom all electrons from unshared pairs it has. 3. Subtract the number of electrons assigned to each atom from the number of valence electrons for an atom of the element.

0 Formal Charges 0 0 -1 0 +1 O=C=O O C=O Structure 1 Structure 2 For the single-bond oxygen For eachmost oxygen The likely Lewis structures are those which (6 e from unshared e + 1 e- from have: assigned from (4 electrons bond) = 7 total - from the bonds) unshared e- + 2 eobeying Formalrule, charge = 6 - 7 = -1 all atoms the octet rule = 6 total the triple-bond oxygen all atoms with a formal. For charge of zero, or (2 e from unshared e + 3 e from Formal 6 -6=0 the charge most =electronegative element with the bonds) = 5 total formal charge. Formal charge = 6 - 5 = +1 Fornegative carbon For carbon 4 e- assigned from the bonds = 4 4 e- from the bonds = 4 total Formal charge = 4 - 4 = 0 total • • •

Another Example of Formal 0 Charges 0 +1 -1 C=O For oxygen Structure 1 (4 electrons assigned from unshared e- + 2 e- from the Although Structure bonds) = 6 total C=O Structure 2 1 has. Foralloxygen atoms with a formal - from unshared e- + 3 e- from (2 e charge of zero, the carbon atom does not obtain bonds) = 5 total Formal charge =Therefore, 6 -6=0 an octet. Structure 2 is=the Formal charge 6 - 5 most = +1 all atoms obey the For carbon Forlikely carbon. Lewis structure since (2 electrons assigned from unshared octet rule (2 electrons assigned from unshared e- + 2 e- from the bonds) = 4 total Formal charge = 4 - 4 = 0 e- + 3 e- from the bonds) = 5 total Formal charge = 4 - 5 = -1

Homework #82 82. For SO 42 , Cl. O 4 , PO 43 and Cl. O 3 , only one of the possible resonance structures is drawn. a. Must have five bonds to P to minimize formal charge of P. The best choice is to form a double bond to O since this will give O a formal charge of zero and single bonds to Cl for the same reason. b. Must form six bonds to S to minimize formal charge of S.
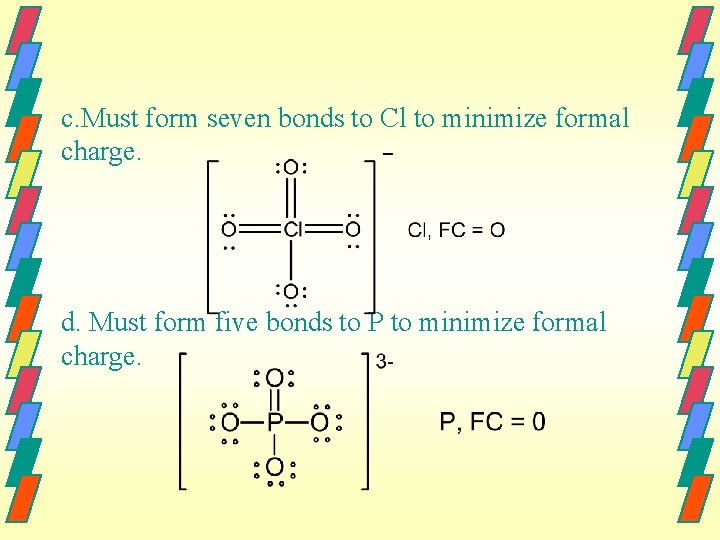
c. Must form seven bonds to Cl to minimize formal charge. d. Must form five bonds to P to minimize formal charge.
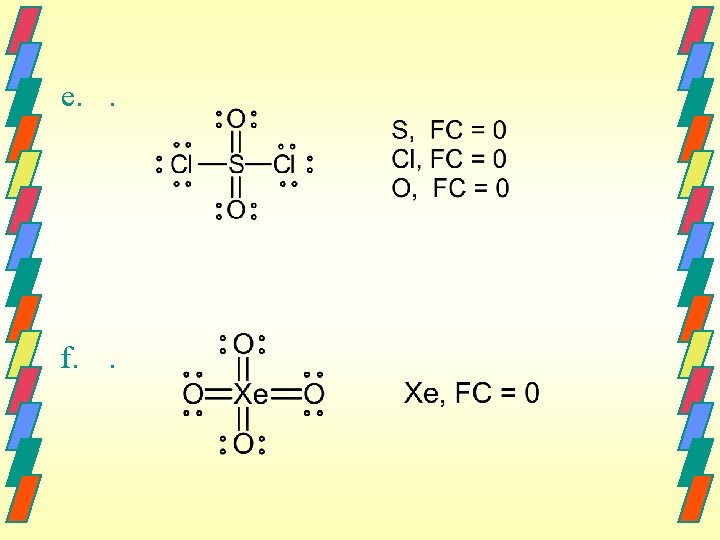

f. . h. We can’t. The following structure has a zero formal charge for N: But N does not expand its octet. We wouldn’t expect this resonance form to exist.

VSEPR Model Valence Shell Electron Pair Repulsion -- The structure around a given atom is determined principally by minimizing electron pair repulsions.

Molecular Geometry Parent Geometry is Actual Geometry is the electron pair arrangement of atoms arrangement about the central atom. • linear • bent • trigonal planar • trigonal pyramid • tetrahedral

Lone pair of electrons on the ammonia molecule.
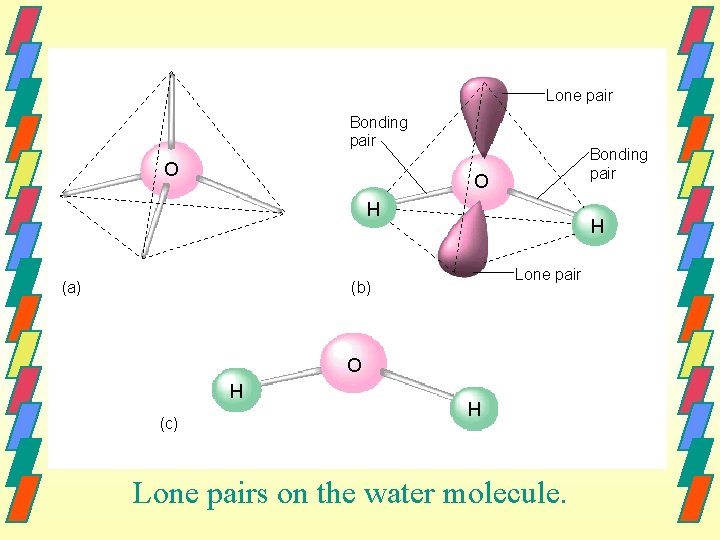
08_143 Lone pair Bonding pair O H (a) H Lone pair (b) O H (c) H Lone pairs on the water molecule.

VSEPR Two pairs of electrons are placed 180 o apart -linear arrangement. Three pairs of electrons are placed 120 o apart - trigonal planar arrangement. Four pairs of electrons are placed 109. 5 o apart -- tetrahedral arrangement. Double bonds and triple bonds count as one effective pair of electrons.
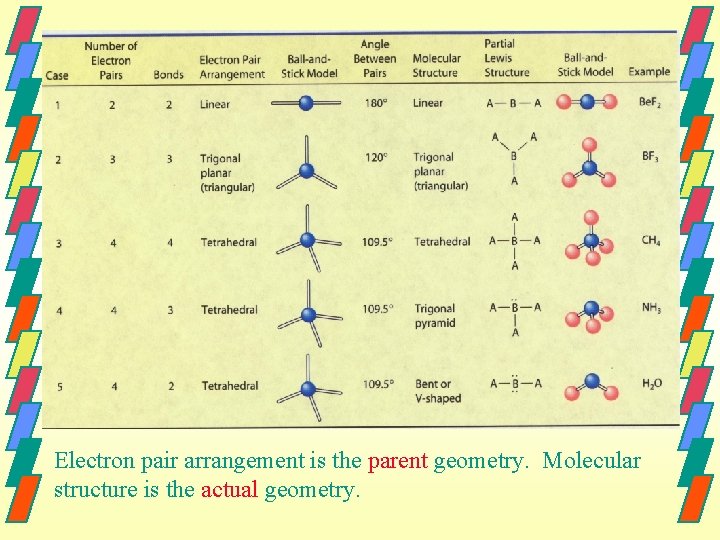
Electron pair arrangement is the parent geometry. Molecular structure is the actual geometry.

Parent & Actual Geometry When every pair of electrons on the central atom is shared with another atom, the parent and actual geometry are the same. When one or more pair of electron pairs around a central atom are unshared(lone pairs), the parent and actual geometry are different.

VSEPR Model Summary • Determine the Lewis structure(s) for the molecule. • For molecules with resonance structures, use any of the structures to predict the molecular structure. • Sum the electron pairs around the central atom to determine the parent geometry. • The arrangement of the pairs is determined by minimizing electron-pair repulsions. (Actual Geometry)

VSEPR Model Summary (Continued) Lone pairs require more space than bonding pairs since they are tightly attracted to only one nucleus. Lone pairs produce slight distortions of bond angles less than 120 o.
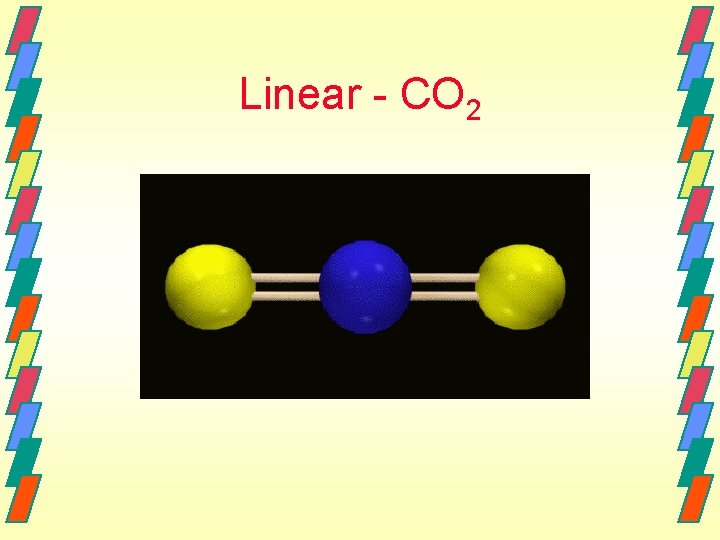
Linear - CO 2
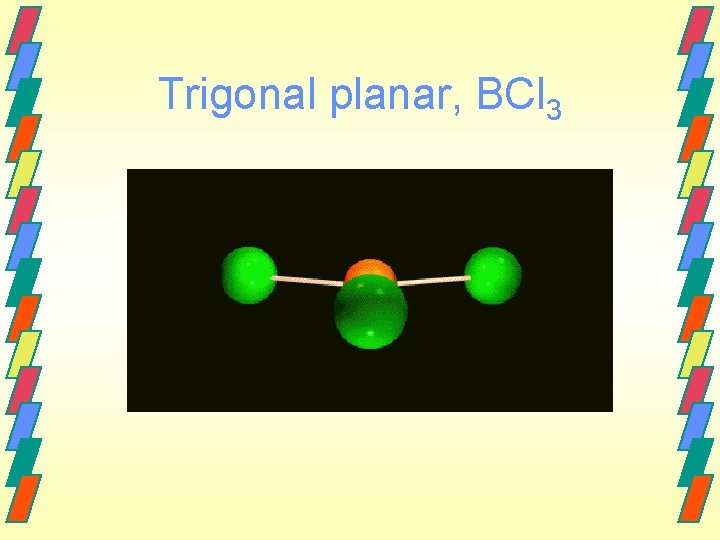
Trigonal planar, BCl 3
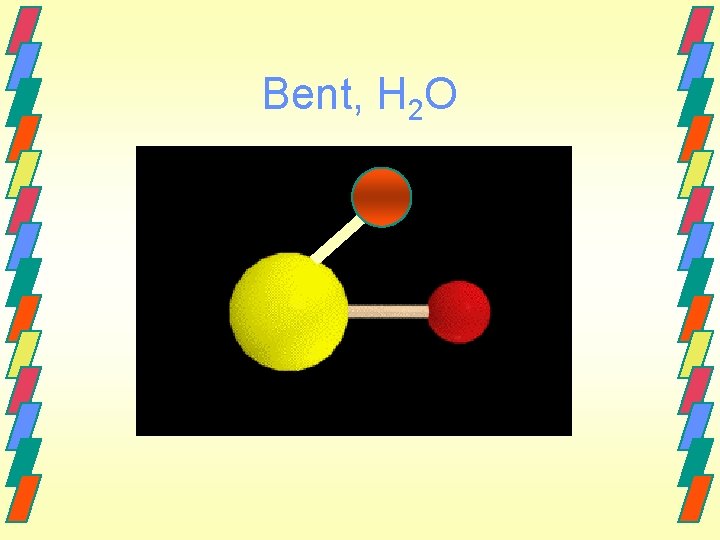
Bent, H 2 O
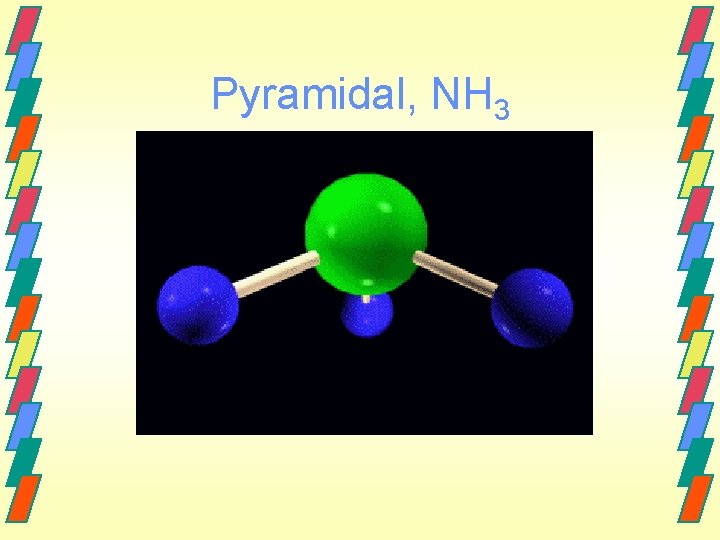
Pyramidal, NH 3
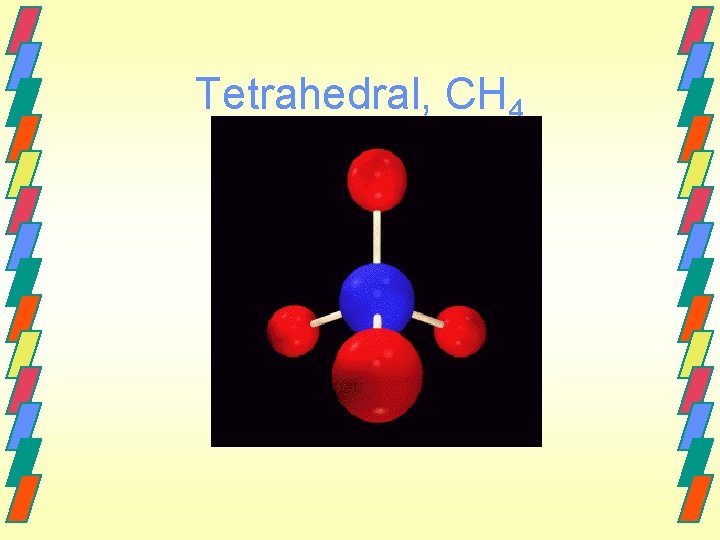
Tetrahedral, CH 4
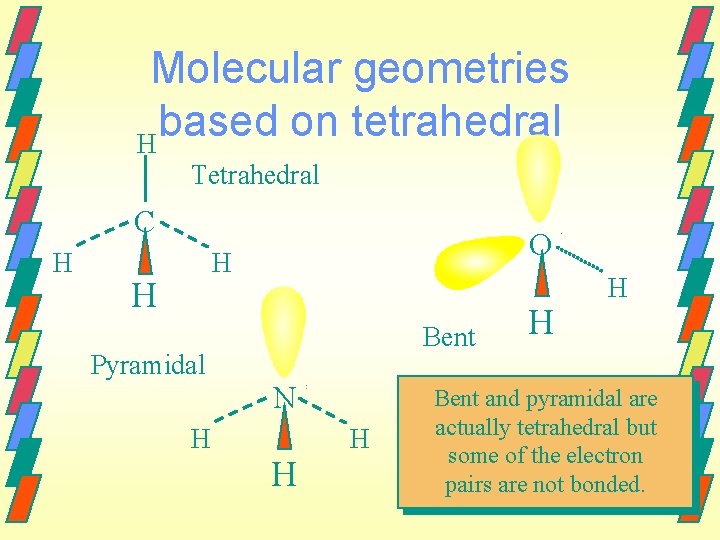
Molecular geometries based on tetrahedral H Tetrahedral C H O H H Pyramidal H Bent N H H Bent and pyramidal are actually tetrahedral but some of the electron pairs are not bonded.

Other geometries. Other shapes are also observed. Five bonds or lone electron pairs Trigonal bipyramidal Seesaw T-shaped Linear Six bonds or lone electron pairs Octahedral Square pyramidal Square planar
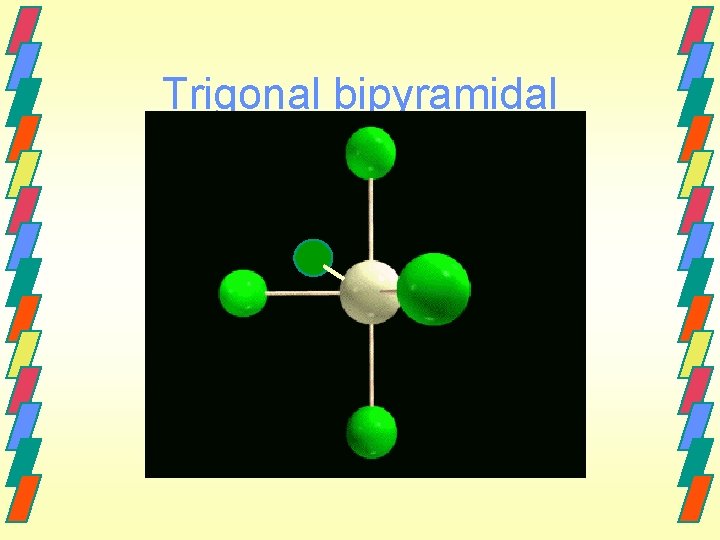
Trigonal bipyramidal
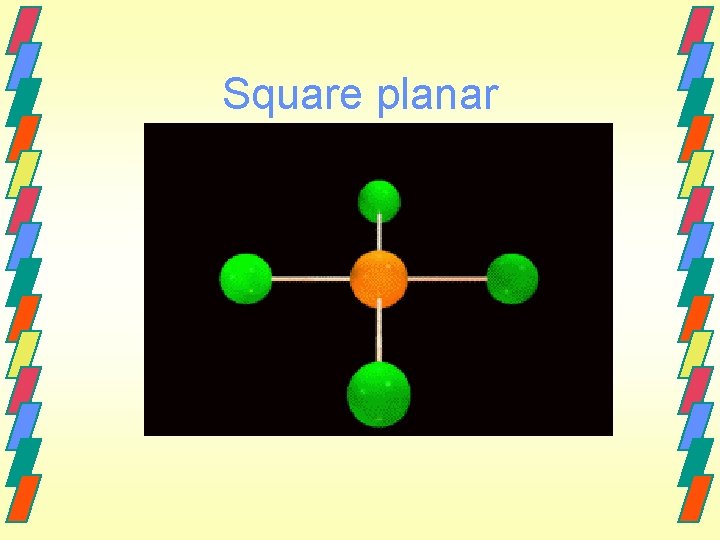
Square planar
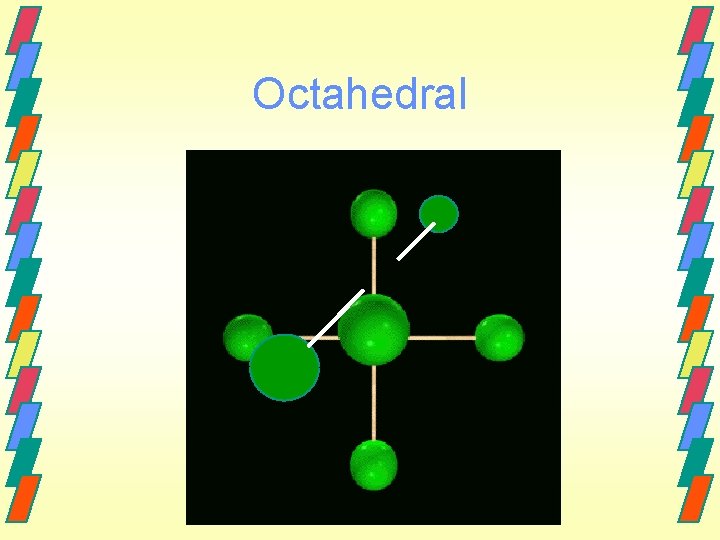
Octahedral
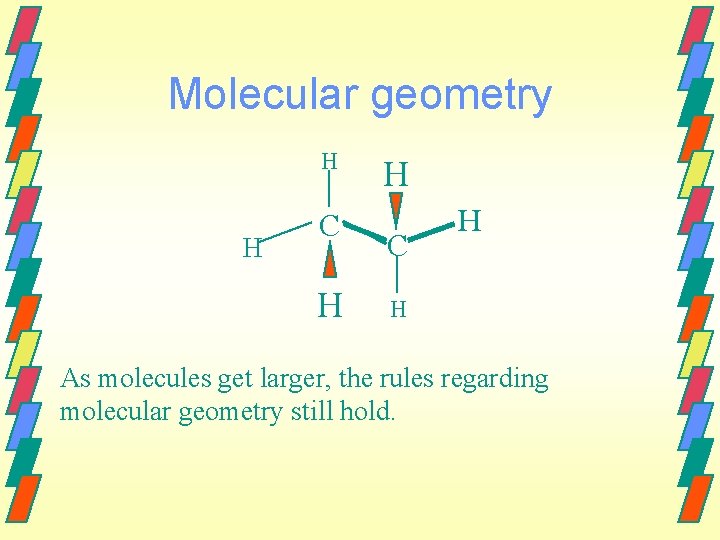
Molecular geometry H H C H H As molecules get larger, the rules regarding molecular geometry still hold.
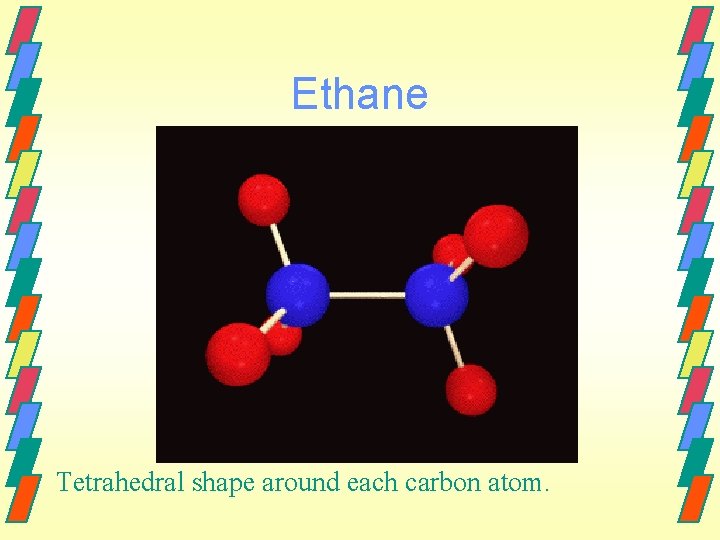
Ethane Tetrahedral shape around each carbon atom.

VSEPR shapes Coordination Number Electron pairs General Bonding Unshared Formula Shape Linear 2 2 0 AB 2 3 3 0 AB 3 Trigonal planar 2 1 AB 2 Bent 4 0 AB 4 Tetrahedral 3 1 AB 3 Trigonal 4 pyramidal 2 2 AB 2 Bent 1 3 AB Linear

VSEPR shapes Coordination Electron pairs General Number Bonding Unshared Formula 5 6 Shape 5 4 0 1 AB 5 AB 4 Trigonal Bipryramidal 3 2 AB 3 T-shaped 2 3 AB 2 Linear 6 0 AB 6 Octahedral 5 1 AB 5 Square pyramidal 4 2 AB 4 Square Planar Seesaw

Polar and nonpolar molecules Most bonds between atoms of different elements in a molecule are polar. That does not mean that the molecule will be polar. O=C=O Electronegativities: Oxygen = 3. 5 Carbon = 2. 5 Difference 1. 0 (polar bond) The electronegativity values Show that the C-O bond would be polar with electrons being pulled towards the oxygens. However, due to the geometry, the pull happens in equal and opposite directions.

Polar and nonpolar molecules For a molecule to be polar, the effects of bond polarity must not cancel out. One way is to have a geometry that is not. . symmetrical Olike in water. Electronegativity H H difference = 1. 3 Here, the effects of the polar bonds do not cancel, so the molecule is polar.

Polar and nonpolar molecules Polarity is an important property of molecules. It affects physical properties such as melting point, boiling point and solubility. Chemical properties also depend on polarity. Dipole moment, moment µ, is a quantitative measure of the polarity of a molecule.
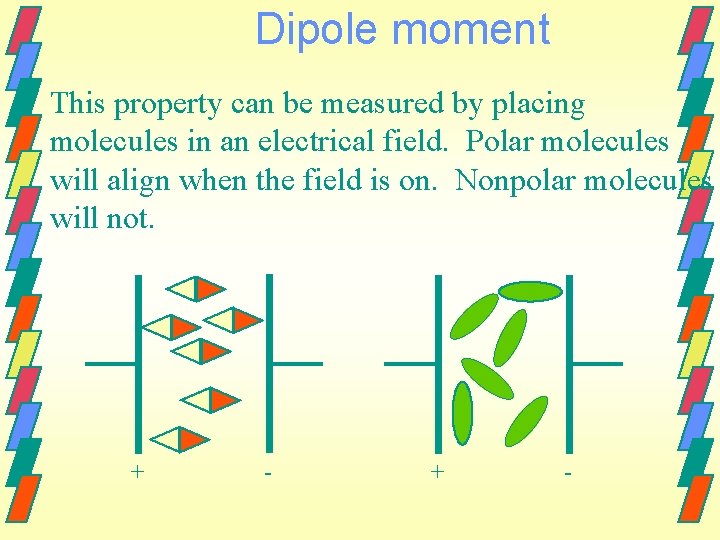
Dipole moment This property can be measured by placing molecules in an electrical field. Polar molecules will align when the field is on. Nonpolar molecules will not. + -

Polar and nonpolar molecules A molecule is nonpolar if the central atom is symmetrically substituted by identical atoms. CO 2, CH 4 , CCl 4 A molecule will be polar if the geometry is not symmetrical. H O, NH , CH Cl

Geometry and polar molecules For a molecule to be polar – - must have polar bonds – - must have the proper geometry • CH 4 • CH 3 Clpolar – • CHCl 3 polar • CCl 4 non-polar CH 2 Cl 2 non-polar


Bonding theory Two methods of approximation are used to describe bonding between atoms. Valence bond method Bonds are assumed to be formed by overlap of atomic orbitals Molecular orbital method
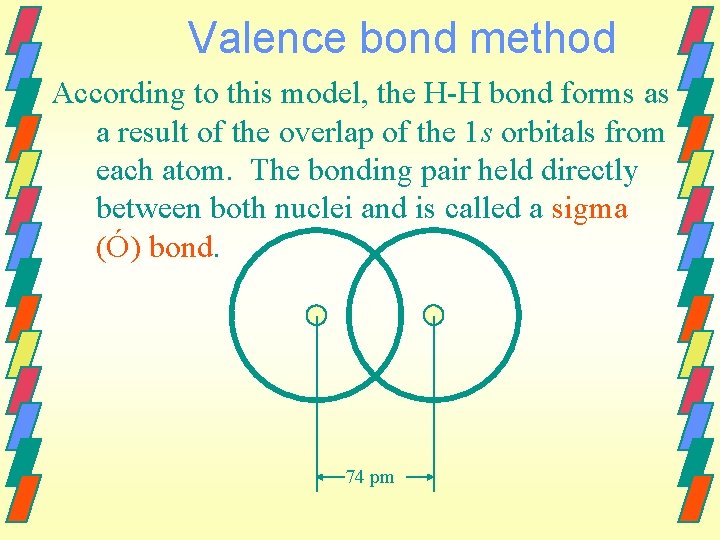
Valence bond method According to this model, the H-H bond forms as a result of the overlap of the 1 s orbitals from each atom. The bonding pair held directly between both nuclei and is called a sigma (Ó) bond. 74 pm
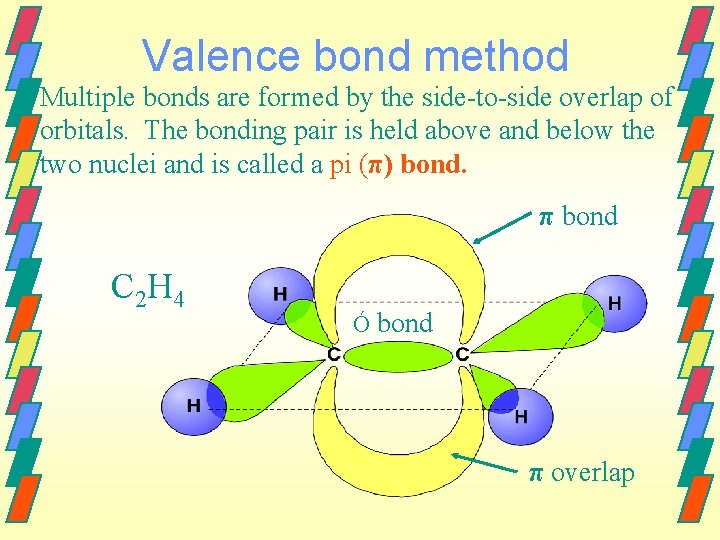
Valence bond method Multiple bonds are formed by the side-to-side overlap of orbitals. The bonding pair is held above and below the two nuclei and is called a pi (π) bond. π bond C 2 H 4 Ó bond π overlap

Valence bond method Hybrid orbitals are need to account for the geometry that we observe for many molecules. Example - Carbon Outer electron configuration of 2 s 2 2 px 1 2 py 1 We know that carbon will form four equivalent bonds - CH 4, CH 2 Cl 2 , CCl 4.
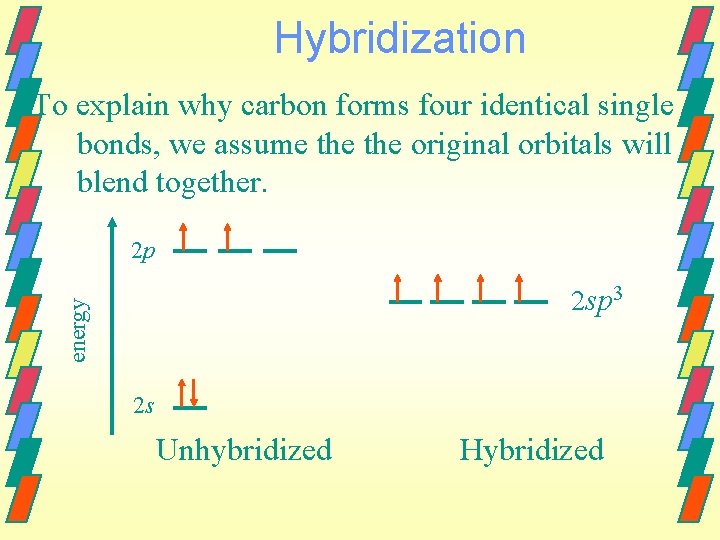
Hybridization To explain why carbon forms four identical single bonds, we assume the original orbitals will blend together. 2 p energy 2 sp 3 2 s Unhybridized Hybridized

Hybridization In the case of a carbon that has 4 single bonds, all of the orbitals are hybrids. sp 3 +3 1 4 25% s and 75% p character s p sp 3
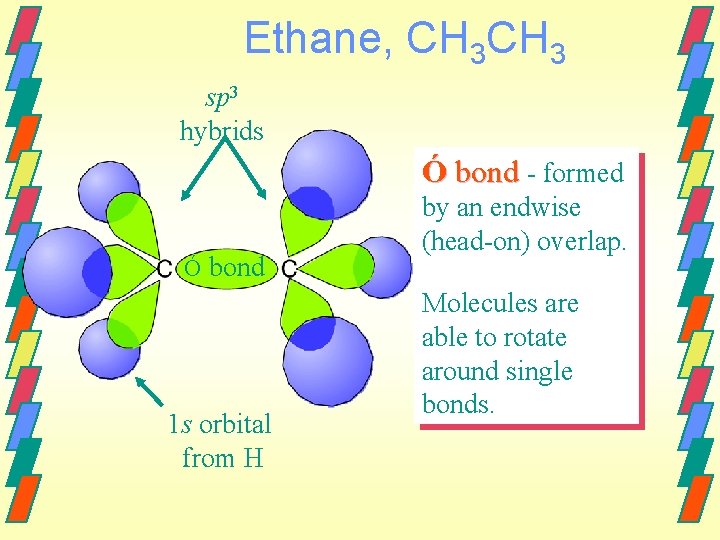
Ethane, CH 3 sp 3 hybrids Ó bond - formed Ó bond 1 s orbital from H by an endwise (head-on) overlap. Molecules are able to rotate around single bonds.
Ethane , CH 3 Rotation of single bond

sp 2 hybrid orbitals To account for double bonds, a second type of hybrid orbital must be pictured. An sp 2 hybrid is produced by combining one s and 2 p orbitals. One p orbital remains. 2 p energy 2 p 2 sp 2 2 s Unhybridized Hybridized
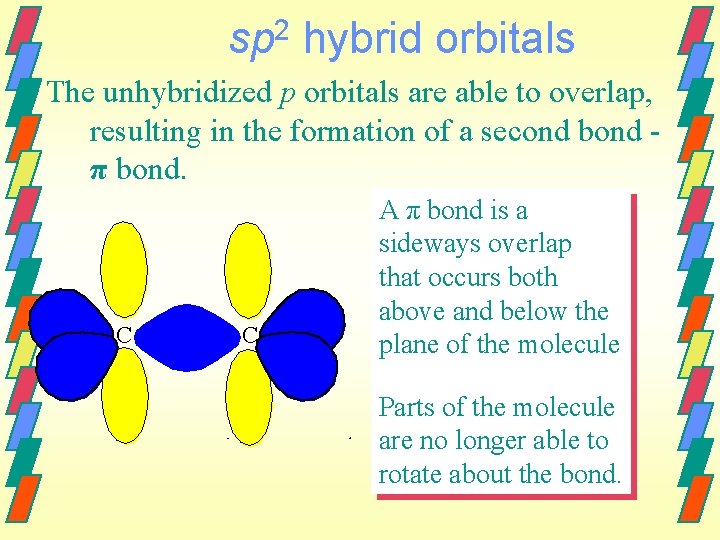
sp 2 hybrid orbitals The unhybridized p orbitals are able to overlap, resulting in the formation of a second bond π bond. C C A π bond is a sideways overlap that occurs both above and below the plane of the molecule Parts of the molecule are no longer able to rotate about the bond.
Ethene
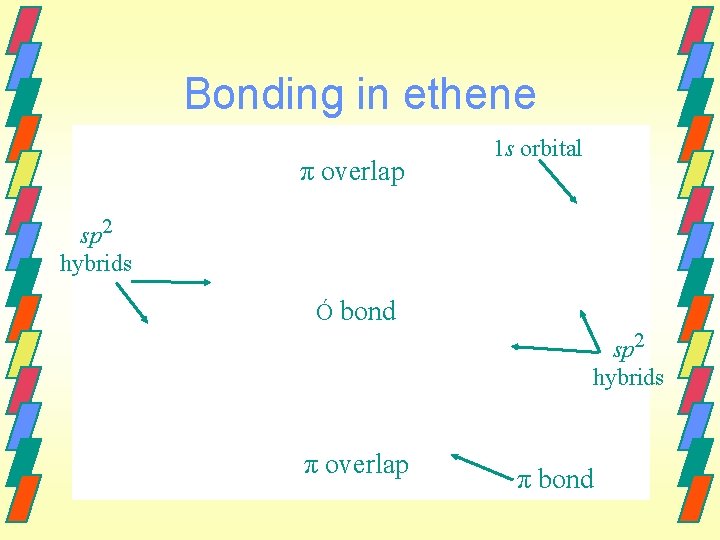
Bonding in ethene π overlap 1 s orbital sp 2 hybrids Ó bond sp 2 hybrids π overlap π bond

Bonding in ethene
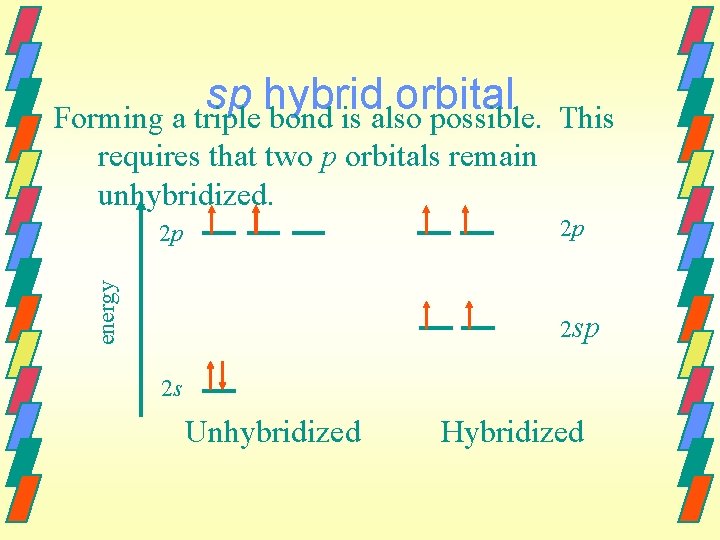
sp hybrid orbital Forming a triple bond is also possible. This requires that two p orbitals remain unhybridized. 2 p energy 2 p 2 s Unhybridized Hybridized

sp hybrid orbital Now two p orbitals are available to form π bonds.
Ethyne
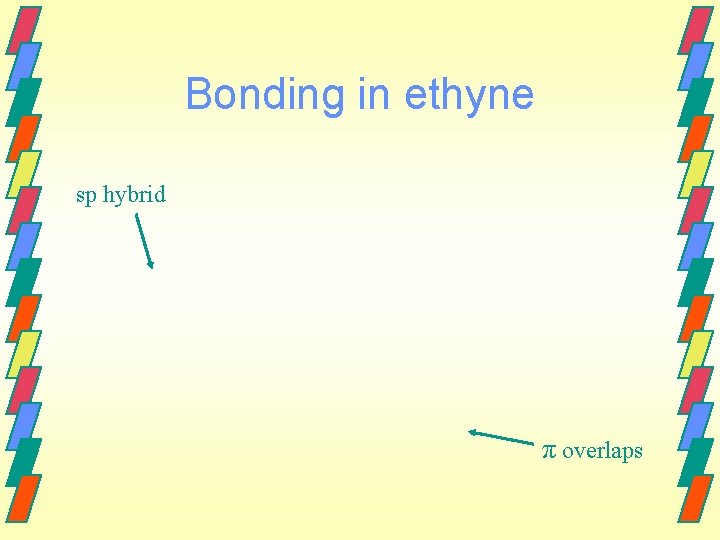
Bonding in ethyne sp hybrid π overlaps

Bonding in ethyne

Other hybrid orbitals can also be involved in the formation of hybrid orbitals. Hybrid Shape sp Linear sp 2 Trigonal planar sp 3 Tetrahedral sp 3 d Trigonal bipyramidal sp 3 d 2 Octahedral

Molecular Orbital Method When atomic orbitals combine to form molecular orbitals, the number of molecular orbitals formed must equal the number of atomic orbitals mathematically combined. Example - H 2 Two 1 s orbitals will combine forming two
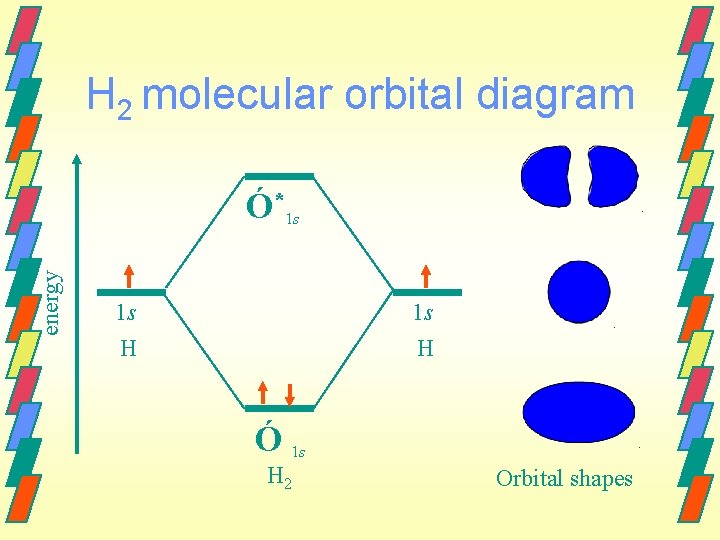
H 2 molecular orbital diagram energy Ó*1 s 1 s H Ó 1 s H 2 Orbital shapes

Molecular orbitals When two atomic orbitals combine, three types of molecular orbitals are produced. Bonding orbital - Ó or π The energy is lower than the atomic orbitals and the electron density overlaps. Antibonding orbital - Ó* or π* The energy is higher than the atomic orbitals
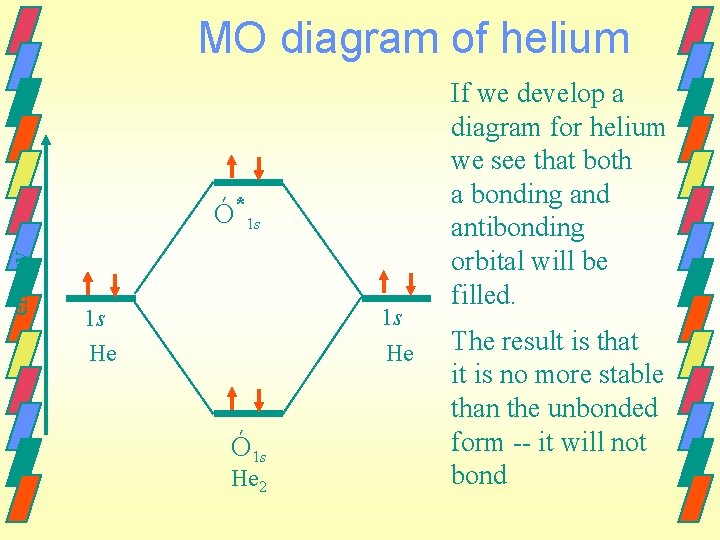
MO diagram of helium energy Ó*1 s 1 s He Ó 1 s He 2 If we develop a diagram for helium we see that both a bonding and antibonding orbital will be filled. The result is that it is no more stable than the unbonded form -- it will not bond

Molecular orbital bonding For a molecule to be stable, you must have more electrons in bonding orbitals than in antibonding orbitals. The bonded form will be at a lower energy so will be more stable. #bonds formed=bonding e- -antibonding e-
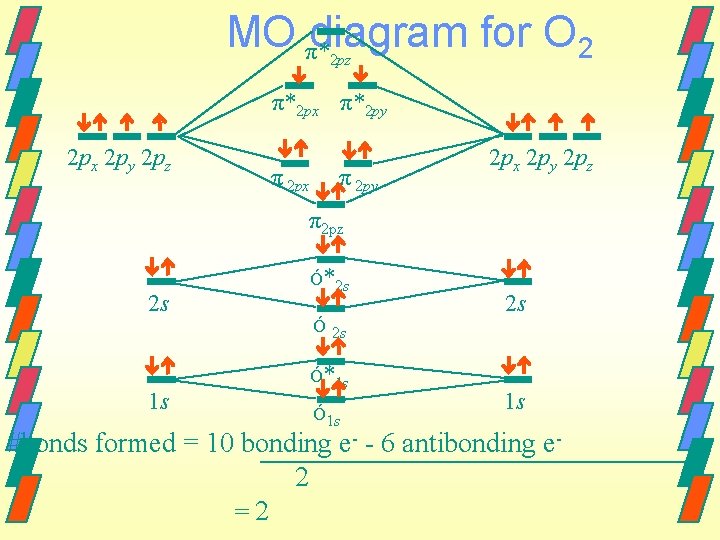
MO π*diagram for O 2 2 pz π*2 px π*2 py 2 px 2 py 2 pz π 2 px π 2 py 2 px 2 py 2 pz π2 pz 2 s Ó*2 s 2 s Ó 2 s 1 s Ó*1 s Ó 1 s 1 s #bonds formed = 10 bonding e- - 6 antibonding e 2 =2
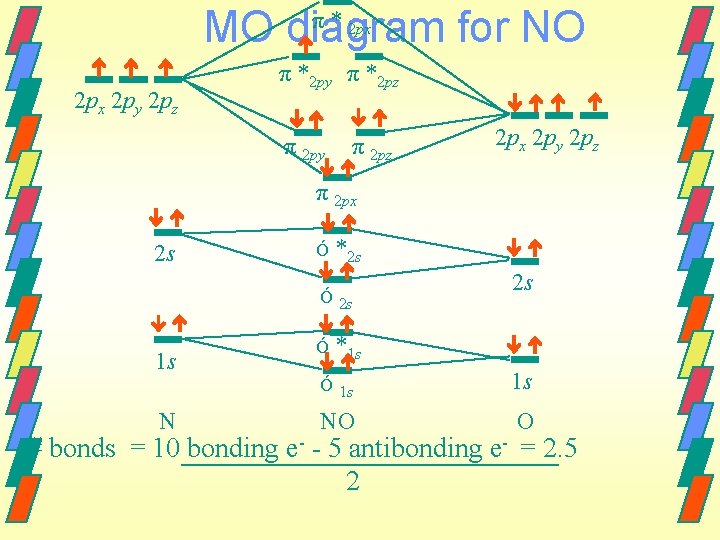
MO diagram for NO π * 2 px 2 py 2 pz π *2 py π *2 pz π 2 py π 2 pz 2 px 2 py 2 pz π 2 px 2 s Ó *2 s Ó 2 s 1 s N Ó 2 s *1 s Ó 1 s 1 s NO O # bonds = 10 bonding e- - 5 antibonding e- = 2. 5 2

Delocalized electrons MO diagrams for polyatomic species are often simplified by assuming that all Ó and some π orbitals are localized -- shared between two specific atoms. Resonance structures require that electrons in some π orbitals be pictured as delocalized. Delocalized - free to move around three or more atoms.
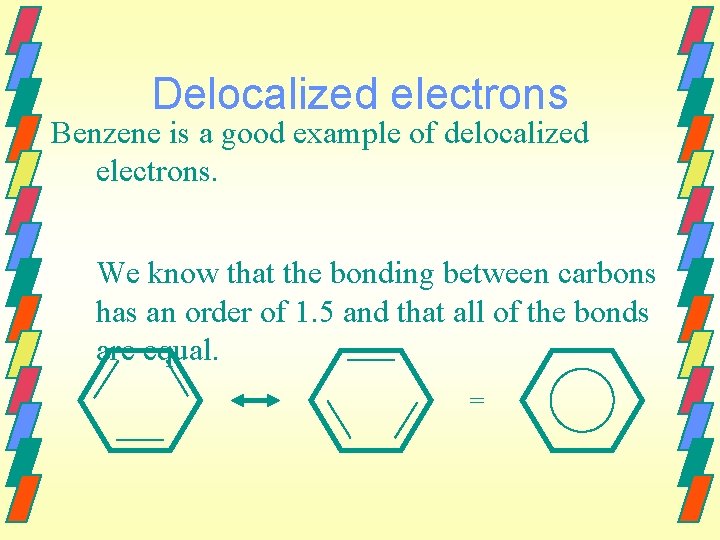
Delocalized electrons Benzene is a good example of delocalized electrons. We know that the bonding between carbons has an order of 1. 5 and that all of the bonds are equal. =

Aromatic hydrocarbons H H H p orbitals overlap sidewise all around the ring. No localized double bonds.
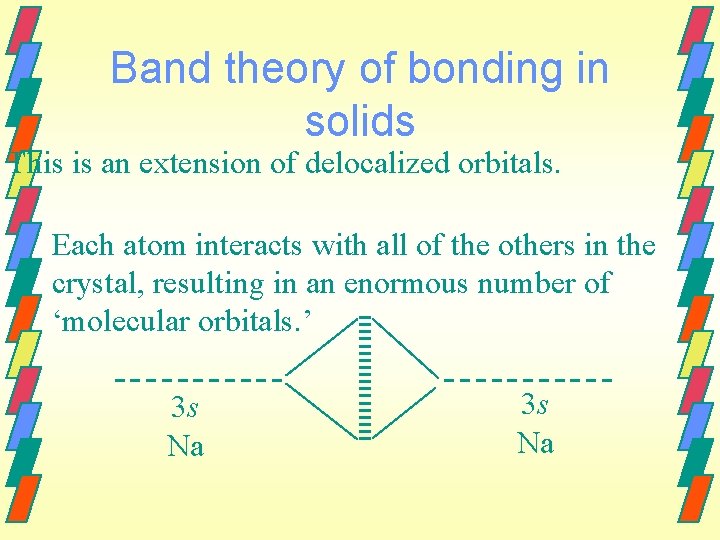
Band theory of bonding in solids This is an extension of delocalized orbitals. Each atom interacts with all of the others in the crystal, resulting in an enormous number of ‘molecular orbitals. ’ 3 s Na

Band theory of bonding in solids Band A group of very closely spaced energy levels. Energy gap The difference in energy between the bonding and antibonding orbitals. Forbidden bands

Band theory of bonding in solids Conductor - A material with a partially filled energy band. Insulator - The highest occupied band is filled or almost completely filled. The forbidden band just above the highest filled is wide. Semiconductor - The gap between the highest filled band the next higher permitted band is relatively narrow.
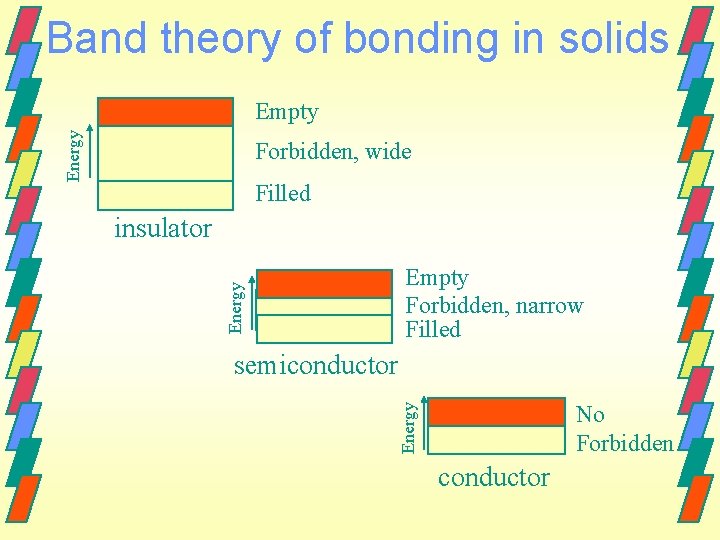
Band theory of bonding in solids Energy Empty Forbidden, wide Filled insulator Energy Empty Forbidden, narrow Filled semiconductor Energy No Forbidden conductor
Intermolecular Attractions What type of attractive forces hold molecules together in liquids and solids?

Intermolecular Forces Intermolecular forces - are forces between the molecules of molecular substance that tend to be weaker than normal covalent bonds There are several types of intermolecular bonds which we will discuss

London forces Temporary dipole attractions that exist between nonpolar molecules - also called the dispersion forces Results from an unbalanced distribution of valence electrons, resulting in a temporary dipole. A relatively weak force that increases with

Dipole-Dipole Interactions Dipole-dipole interactions are weak intermolecular forces that can occur between polar molecules. Polar molecules attract one another when a positive side (∂+) and a negative side (∂) of two polar molecules are near one another.
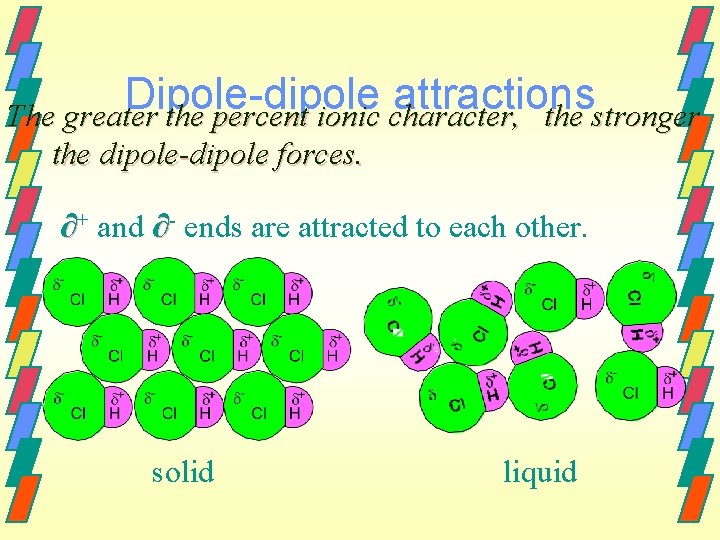
Dipole-dipole attractions The greater the percent ionic character, the stronger the dipole-dipole forces. ∂+ and ∂- ends are attracted to each other. solid liquid

Hydrogen Bonding Are intermolecular forces resulting from a specific interaction between a hydrogen atom in one molecule and a fluorine, oxygen, chlorine or nitrogen atom in another molecule The hydrogen attempts to form a coordinate covalent bond with an extra pair of electrons on a neighboring atom.

Hydrogen Bonding The small sizes of the elements involved and the large electronegativity differences result in large ∂+ and ∂- values. Hydrogen bonds are represented using a dashed line.
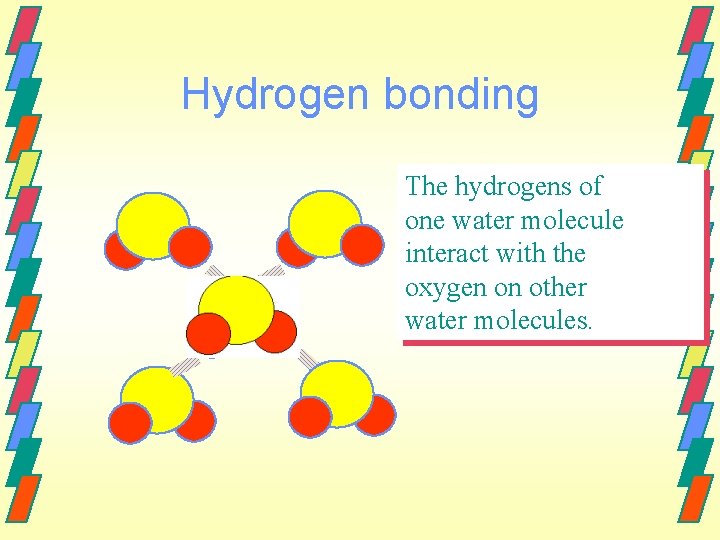
Hydrogen bonding The hydrogens of one water molecule interact with the oxygen on other water molecules.

Ion-Dipole Attractions When ionic compounds dissolve in water, the negative ions are attracted to the oxygen atoms, and the positive ions are attracted to the hydrogen atoms. The degree of dissociation depends on the strength of these ion-dipole attractions

Intermolecular forces For molecules to form liquids and solids, there must be attractions between them. Intermolecular attractive forces • ion-dipole attraction • dipole-dipole attraction including hydrogen bonding • London (dispersion) forces Relative strength

Dissolving covalent compounds Covalent compounds do not dissociate (separate into ions) when they dissolve.
- Slides: 167