Chemical bonding Bonding Chemical bond the transfer or

Chemical bonding Bonding Chemical bond – the transfer or sharing of electrons Ionic bond- the transfer of electrons Covalent bonding – the sharing of electrons (2) Nonpolar –nearly equal, or equal sharing Polar - unequal sharing
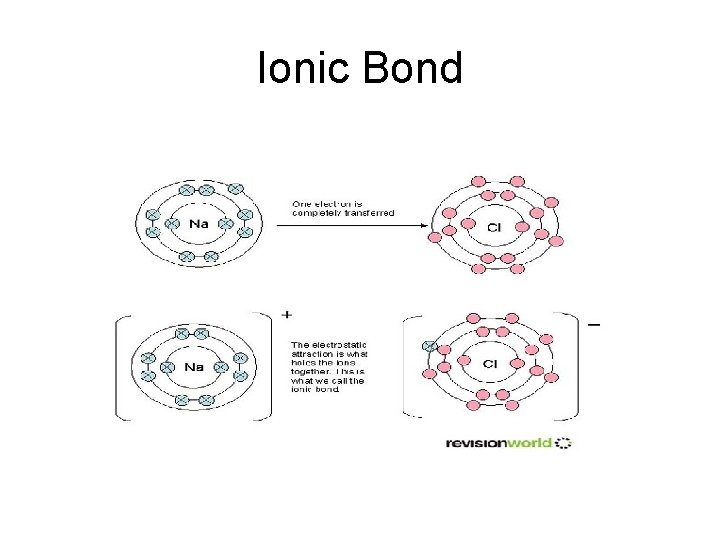
Ionic Bond
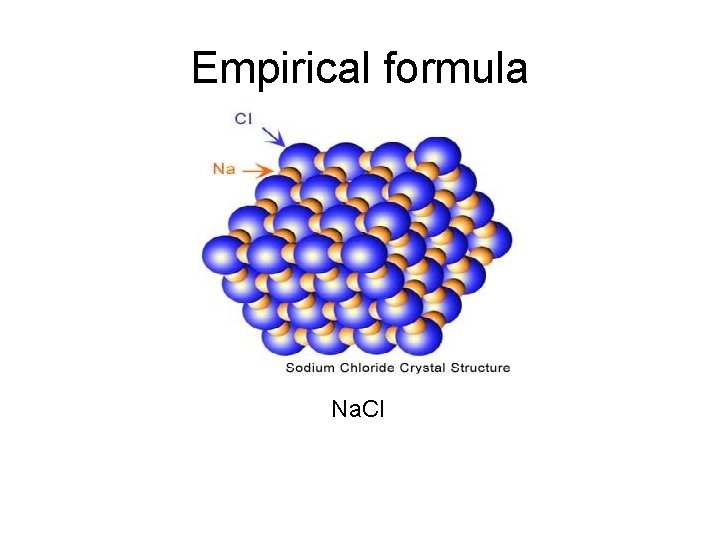
Empirical formula Na. Cl

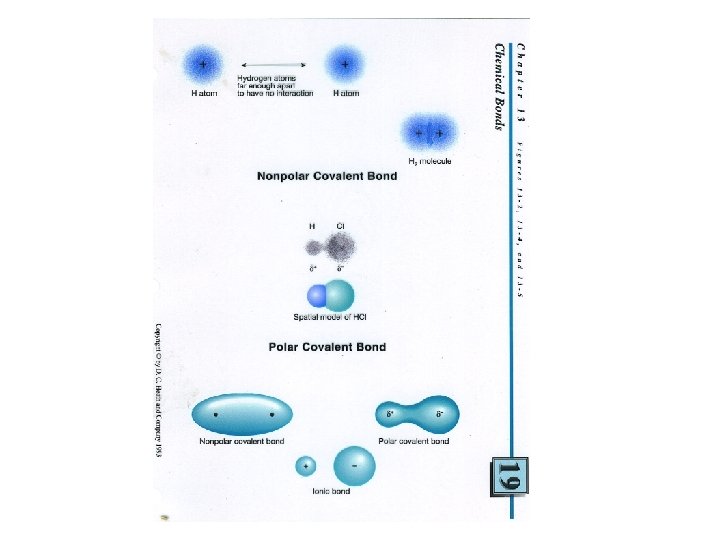
Covalent bond non polar • Two chlorine atoms They always share in pairs Can be single ( 2 electrons) Double ( 4 electron, 2 pairs) Or triple ( 6 electrons, 3 pairs)
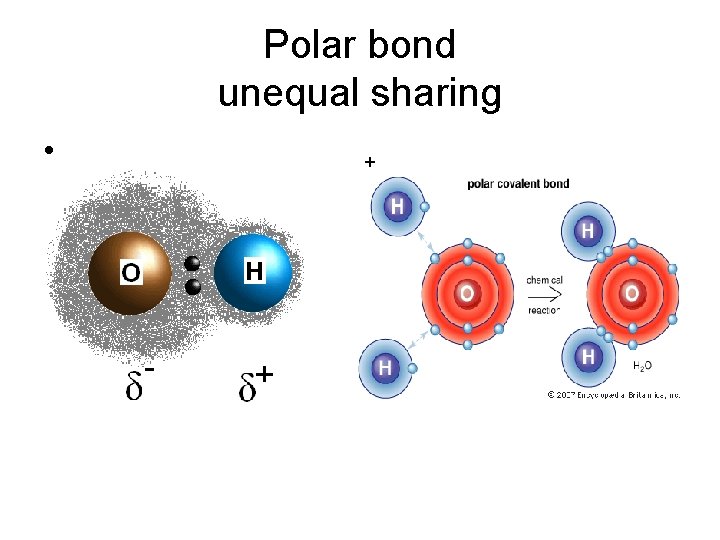
Polar bond unequal sharing • +

• One atom has a greater pull and there for a greater share of the electrons. Giving it a slight negative charge, and the other atom a slight positive charge
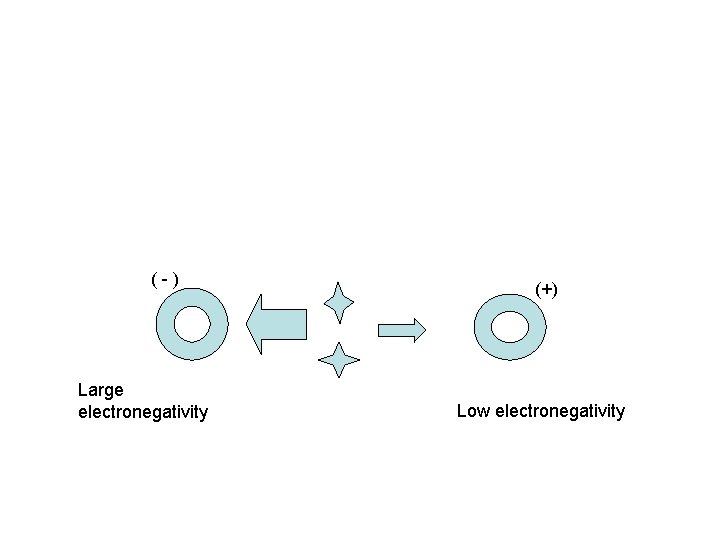
(-) Large electronegativity (+) Low electronegativity

• Electronegativity - measure of an atoms attraction for electrons used for bonding • The difference in electronegativity values determines the type of bond. • . 4 -------1. 7 • Non polar ionic
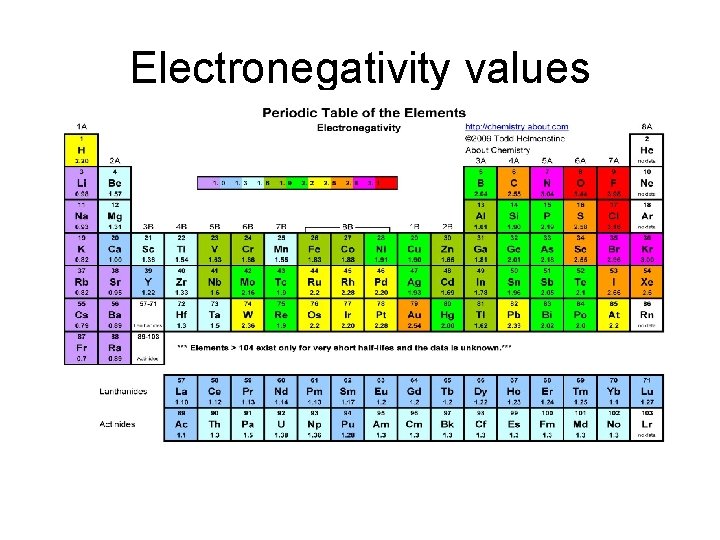
Electronegativity values

VSEPR Theory • • Families 1 2 3 4 5 6 7 8 X X X X • 1 linear diatomic, 2 linear triatomic, 3 planar triangle, 4 tetrahedron, 5 pryamidal, 6 bent, 7 linear diatomic • Demonstrate models
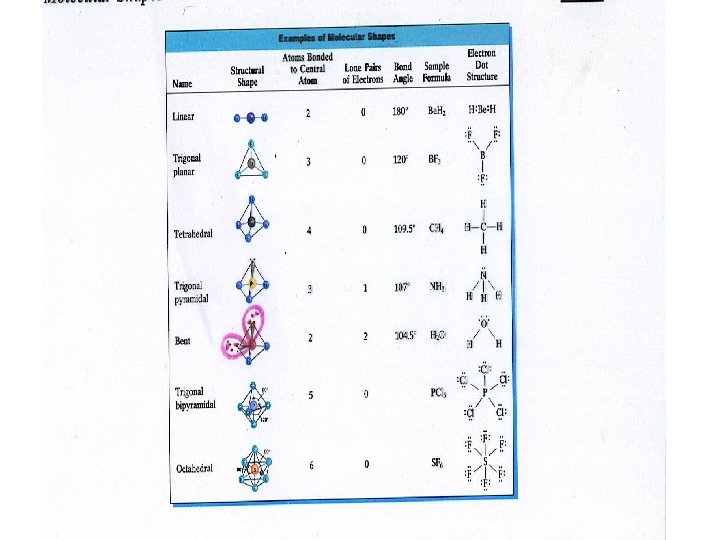
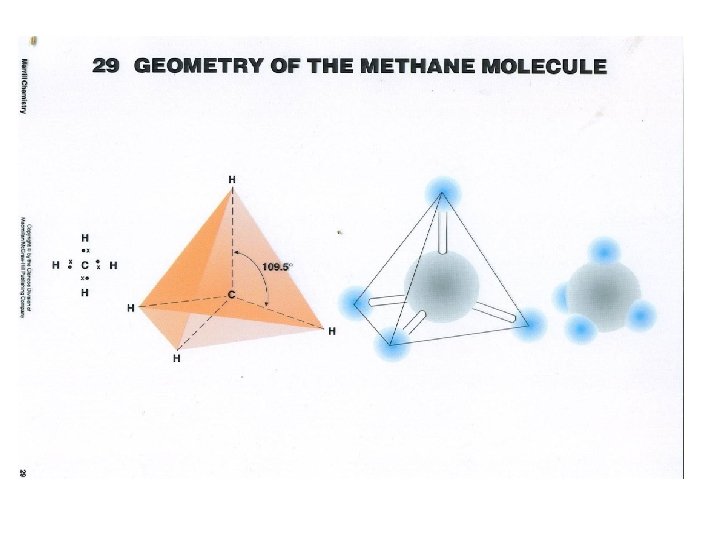

• • Polar and nonpolar Molecules To determine this two factors must be considered. • What are the bond types? ( electronegativity diff) • What is the shape of the molecule ? (VSEPR ) Is it symmetrical or not? • • If the bonds are nonpolar , the molecule is nonpolar If the bonds are polar and the shape is symmetrical the molecule is nonpolar If the bonds are polar and the shape is not symmetrical the molecule is polar
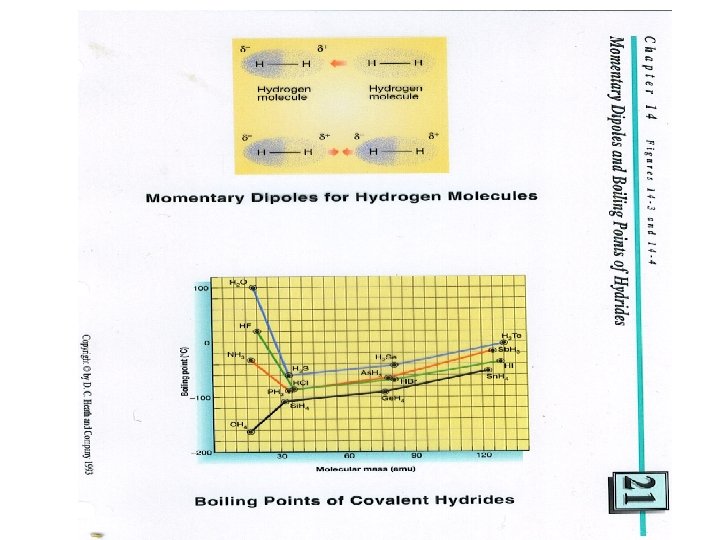
• Bonds between molecules (intermolecular) • Dipole – dipole • Hydrogen bonding • H- O, H – N, H – F, molecules with these combination have • Hydrogen bonding
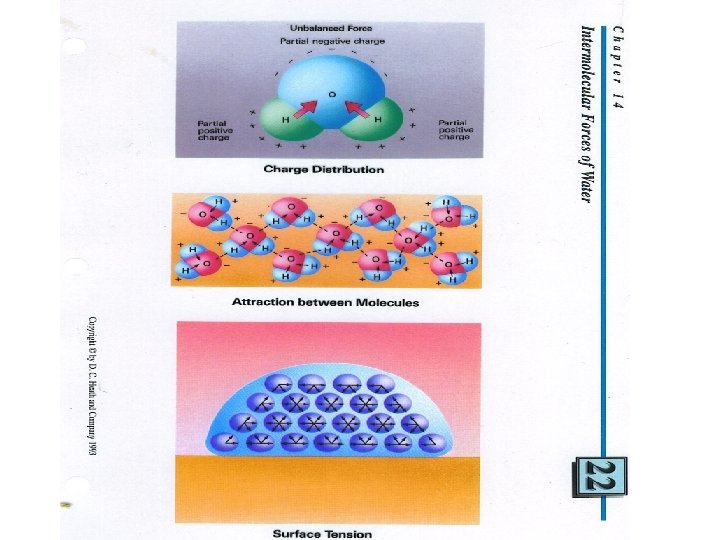

What would make things with no charge attract each other? • Nonpolar molecules use • Van Der Waal Forces to form • liquids • Also called London forces • Which are momentary • dipoles
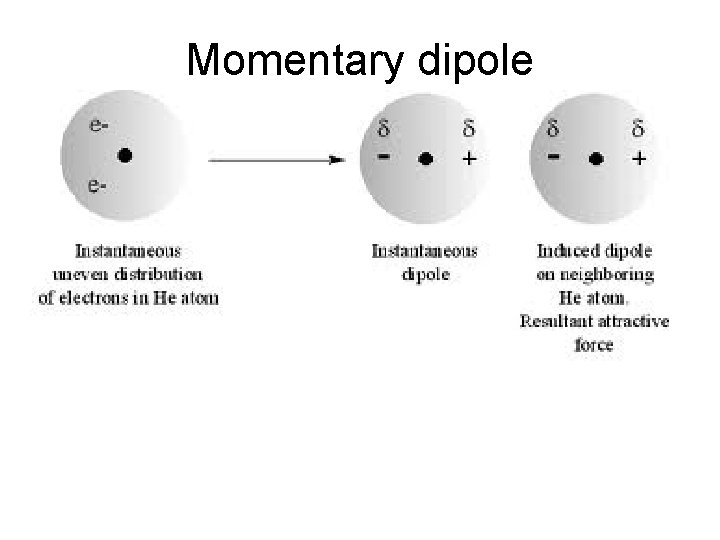
Momentary dipole
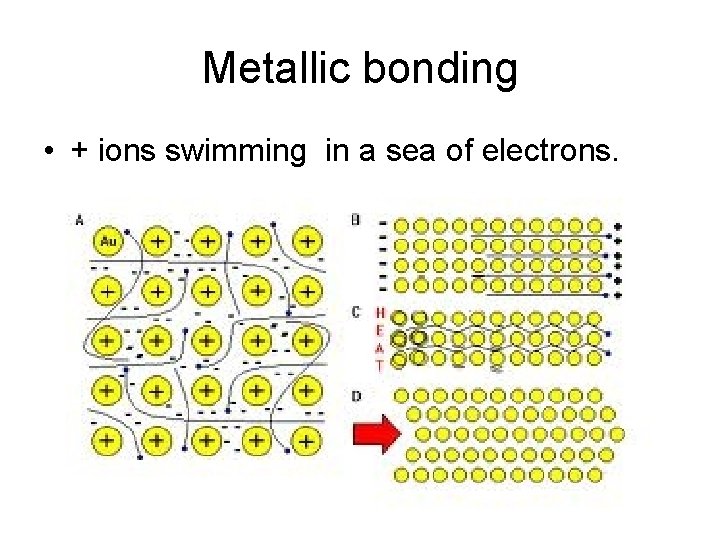
Metallic bonding • + ions swimming in a sea of electrons.
- Slides: 20