CHEMICAL BONDING THE NATURE OF CHEMICAL BONDING A
CHEMICAL BONDING `

THE NATURE OF CHEMICAL BONDING A _______ results from the simultaneous attraction of _____ to two ______. _____ energy is _____ in the chemical bond. This energy is equivalent to ______ due to position or condition of the bond. When a chemical bond is formed in a reaction, energy is _____. They are in a _____ energy position then when they are separated. When a chemical bond is broken energy is _____. The atoms separated have _______ energy.

BONDING AND STABILITY A compound represents a _____ energy condition than its individual atoms and is generally considered _____ stable. In general chemical changes will occur among atoms if the changes lead to a ____ energy condition and hence a ____ stable structure. Bond _____ is favored. In general ____ reactions in nature are favorable.

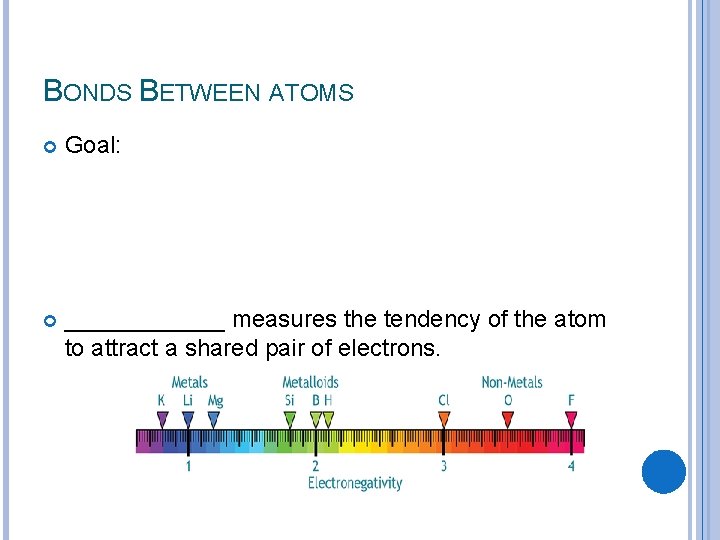
BONDS BETWEEN ATOMS Goal: ______ measures the tendency of the atom to attract a shared pair of electrons.

ELECTRONEGATIVITY DIFFERENCE AND BOND TYPE Ionic Bond Covalent Bond Transfer of electrons Metal and nonmetal E. N difference≥ 1. 7 Greater the E. N. difference the greater the ionic character Sharing of electrons Nonmetals E. N. difference between 0 and 1. 7 E. N. difference=0 is a nonpolar bond (equal share. (<. 3) E. N difference between. 3 and 1. 7 polar bond (unequal share)

METALLIC BOND Positive ion immersed in a sea of mobile electrons Metals only Few valence electrons Low ionization energy

DETERMINE THE ELECTRONEGATIVITY DIFFERENCE FOR EACH COMPOUND AND DETERMINE THE BOND TYPE Na. Cl HCl Ca. Br 2 H 2 O Ba. O Cl 2
TRY THESE TOO!! Na. H Cu Ca. H 2 Mg

WITHOUT FIGURING OUT THE ELECTRONEGATIVITY DIFFERENCE WHAT BOND TYPE DO THESE COMPOUNDS HAVE? Cu. SO 4 Na. NO 3 NH 4 Cl Why?
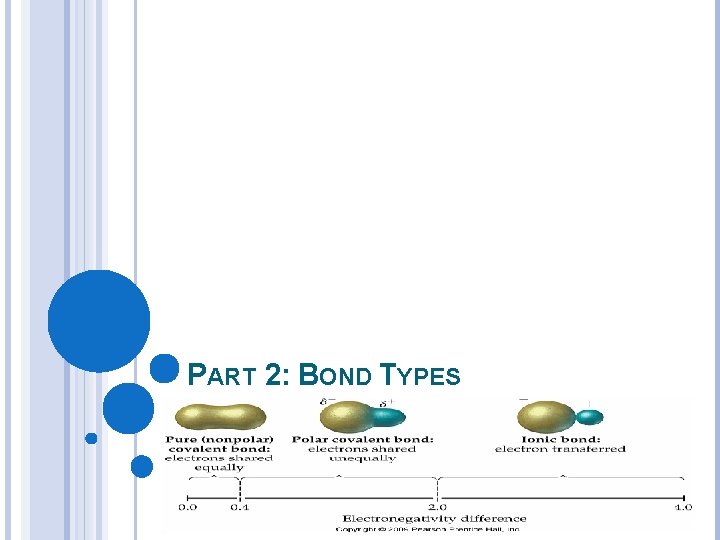
PART 2: BOND TYPES

IONIC BONDING (ELECTROVALENT) _______of _______ Between a ______ and a ______. A _____ loses an electron to form a _______ ion called a _____. A ______ gains an electron to form a ______ ion called an _______. The ions are drawn together by electrostatic forces and form an ionic compound known as a ____. The greater the elctronegativity difference the greater the ionic character and the stronger the attractive forces. (electronegativity difference≥ 1. 7)

SHOW THE BOND BETWEEN SODIUM AND CHLORINE.

SHOW THE BOND BETWEEN CALCIUM AND FLUORINE.

COVALENT BONDING ______ of ______ Between _______. If the electrons are shared equally they form a _________. Electronegativity difference =0 or <. 3. For example: _______ If the electrons are shared unequally they form a ________. Electronegativity difference between. 3 and 1. 7. Two _____nonmetals. The greater the electronegativity difference the greater the polarity or ionic character. Covalent compounds form _______. Molecules may be ______ or ______ Or they can be _______ or _______. The bond type and molecule type may be different.

NONPOLAR BOND: (EQUAL SHARING) H 2 F 2 Note: A _______ bond represents ______pair of electrons or _____electrons
O 2 Note: A _____ bond stands for _____pairs of electrons or _____ electrons. Note: A ____bond stands for ______pairs of electrons or ____electrons.

POLAR COVALENT BOND: (UNEQUAL SHARE) HCl Br. Cl

DIATOMIC MOLECULES (2 ATOMS) If the atoms are the same the bond is ____(equal share) and the molecule is _____(symmetric) If the atoms are different the bond is ____(unequal share) and the molecule is _______ (asymmetric) A polar molecule is also called a _____ because it has_______.

MORE THAN TWO ATOMS If a molecule is made up of more than two atoms the bond may be _____ (unequal share) but the molecule may be ______(symmetric) or ______(asymmetric) Molecules are a SNAP S(symmetric)N(nonpolar)A(asymmetric)P(polar)
TRY THESE!!!! CH 4 CO 2

AND THESE!!!! H 2 O NH 3

COORDINATE COVALENT BOND Water and ammonia both have________. They can form a coordinate covalent bond. A coordinate covalent bond is one formed when two shared electrons forming a covalent bond are both donated by one of the atoms and the other atoms donates none. Once the coordinate covalent bond is formed it behaves as an ordinary covalent bond. The difference ________________. The _____ ion can attach to the water and ammonia molecule forming the ______ ions called ______ and _______. (table
TRY THESE!!!! H 3 O + NH 4+
LET’S MIX IT UP!!!!! What kind of bond are these? Practice time!!!!!

PART 3: SHAPES AND POLARITY OF MOLECULES See sheet on shapes and polarity

PART 4: FORCES THAT HOLD COMPOUNDS TOGETHER

IONIC COMPOUNDS: Bond is ionic between a _______ and a _____. Form ionic compounds called_____. Each compound is attracted to another compound by an ________ between the + ion in one compound and the – ion in an adjacent compound. Very strong force making them solids at room temperature. Stronger the force of attraction the ____the boiling point.

INTERMOLECULAR FORCES OF ATTRACTION: Forces of attraction between molecules: _____bonded substances. Note: intramolecular forces are within a molecule between the atoms. (polar and nonpolar bonds) Depends if a molecule is a nonpolar molecule (symmetric) or a polar molecules (asymmetric) Three types 1. Dipole-Dipole 2. Hydrogen bonding 3. Van Der Waals forces
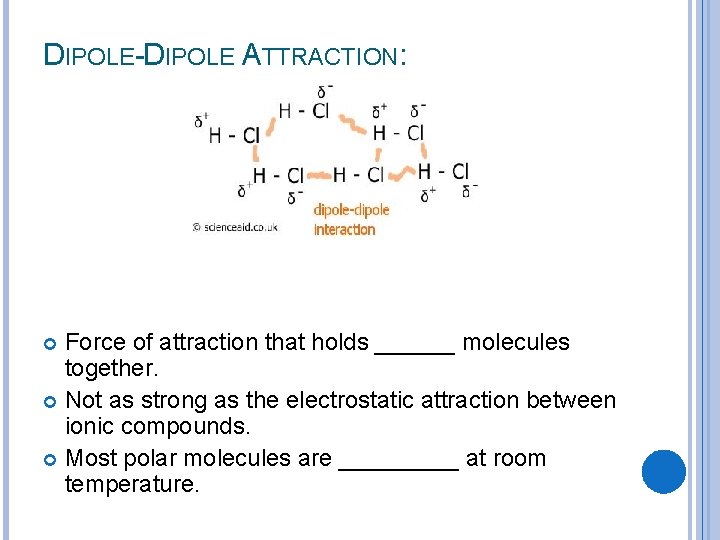
DIPOLE-DIPOLE ATTRACTION: Force of attraction that holds ______ molecules together. Not as strong as the electrostatic attraction between ionic compounds. Most polar molecules are _____ at room temperature.
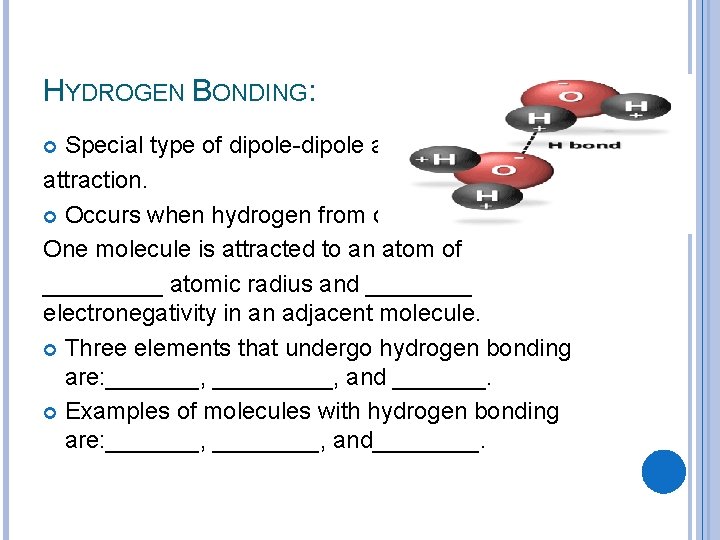
HYDROGEN BONDING: Special type of dipole-dipole aat attraction. Occurs when hydrogen from one One molecule is attracted to an atom of _____ atomic radius and ____ electronegativity in an adjacent molecule. Three elements that undergo hydrogen bonding are: _______, and _______. Examples of molecules with hydrogen bonding are: _______, and____.

SPECIAL CHARACTERISTICS OF HYDROGEN BONDING The covalent bond between the atoms is stronger than the hydrogen bond between the molecules. Stronger than a regular dipole-dipole. Accounts for an abnormally high boiling point and freezing point of a similar compound. For example: Which has the highest boiling point? HCl HF HBr HI The _____ the intermolecular force of attraction the ____ the boiling point.
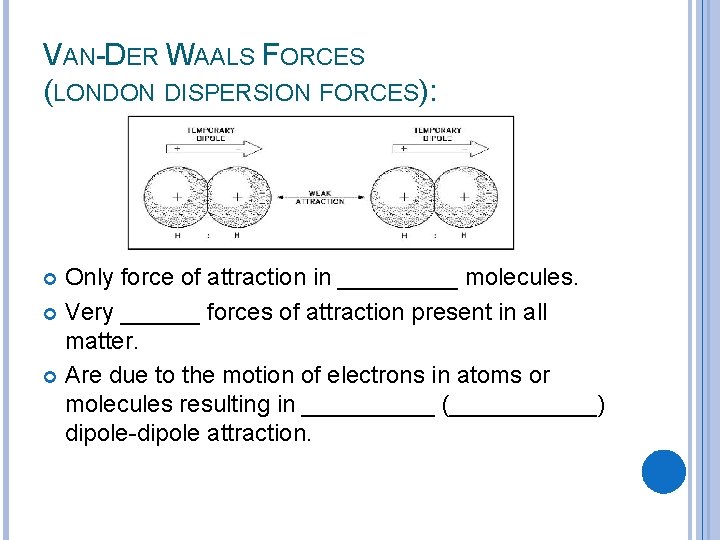
VAN-DER WAALS FORCES (LONDON DISPERSION FORCES): Only force of attraction in _____ molecules. Very ______ forces of attraction present in all matter. Are due to the motion of electrons in atoms or molecules resulting in _____ (______) dipole-dipole attraction.

SPECIAL CHARACTERISTICS OF VAN-DER WAALS FORCES Nonpolar molecules are usually ____ at room temperature. Allows small nonpolar molecules (ex: Hydrogen, helium , carbon dioxide, etc. ) to exist in the liquid and solid phase under conditions of _______ temperature and _____ pressure. Why? The molecules become _____ together _____ the strength of the van der waals forces. An increase in molecular mass will ____ the van der waals forces.
FOR THOUGHT: Explain the phases of the halogens in terms of van der waals forces? Which has the highest boiling point? CH 4 C 2 H 6 C 3 H 8 C 4 H 10

COMPARISON OF THE FORCES HOLDING COMPOUNDS/MOLECULES TOGETHER Strongest: _____________________ Weakest: _______________ Which has the highest boiling point? Carbon dioxide Ammonia Hydrochloric acid Sodium chloride
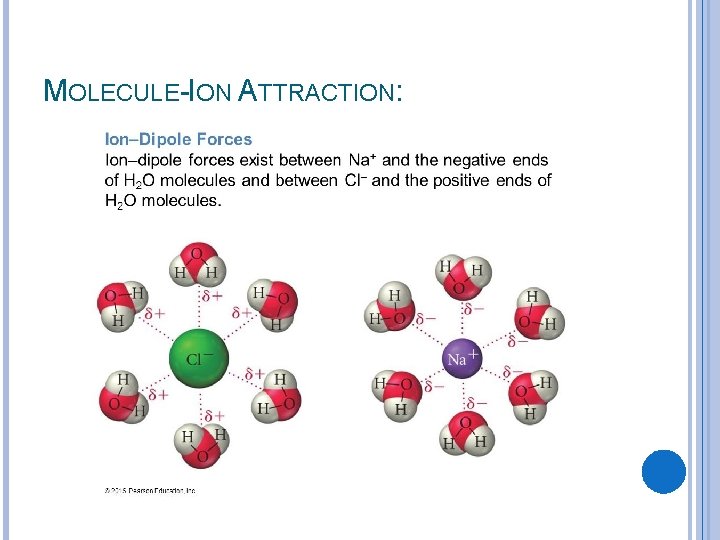
MOLECULE-ION ATTRACTION:

RULES OF SOLUBILITY Ionic and polar covalent solutes ex: _______ and _____ are generally soluble in polar solvents ex: _______ and are relatively insoluble in nonpolar liquids. Nonpolar solutes such as CCl 4 are soluble in nonpolar solvents such as C 6 H 6 and are relatively insoluble in polar solvents such as water. Key phrase: _______________
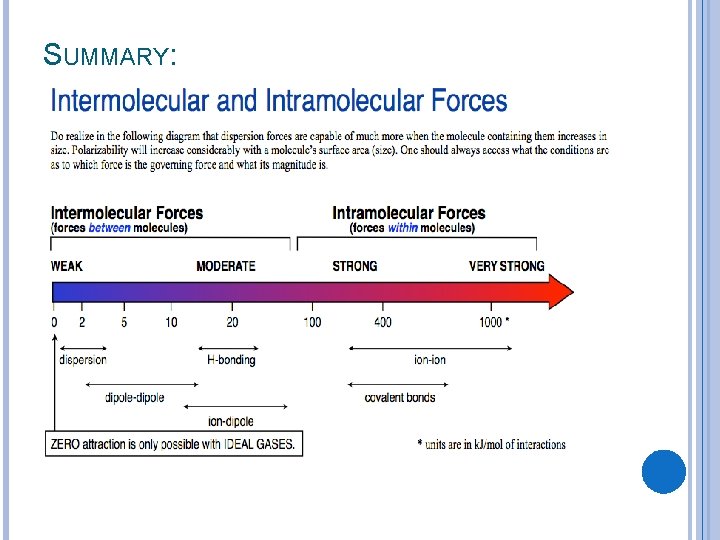
SUMMARY:

PART 5: TYPES OF SOLIDS

DIRECTIONS: In groups of four use your knowledge of chemistry along with websites to complete the table on Comparing types of Solids. Some key concepts to help you: 1. The stronger the force of attraction holding the compound together the ______ boiling point. 2. To conduct an electric current you need ________ or in the case of a metallic bond _________. 3. Rule of solubility: Like dissolves like

- Slides: 42