Chemical Bonding Bonding n A chemical bond is

Chemical Bonding

Bonding n A chemical bond is an electrical attraction between nuclei of one atom and the valence (or outer shell) electrons of a different atom.

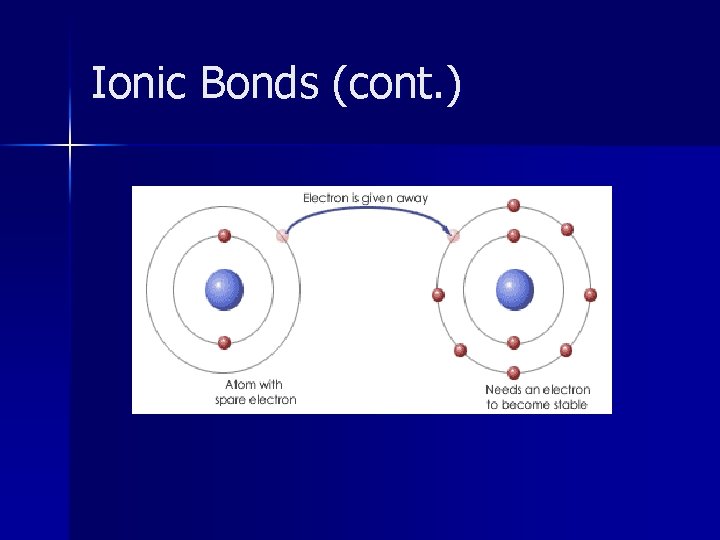
Ionic Bonds n This is an INTRAMOLECULAR bond. n Transfer of electrons from the metal to the nonmetal n Electrical attraction between large numbers of cations and anions “clusters”

Ionic Bonds n Cation = Positively charged particle – Typically are metals – Loves to give electrons away – Low Ionization Energy & Electronegativity – Most metallic element = Francium

Ionic Bonds (cont. ) n Anion = Negatively charged particle – Typically are nonmetals. – Loves to accept electrons – Higher Ionization Energy – Higher Electronegativity – Most nonmetallic element= Fluorine
Ionic Bonds (cont. )

Ionic Bonds (cont. ) n Ionic compounds have strong bonds. n Ionic bonds dissociate in water – Fall apart into cations and anions. – Will conduct electricity when in water. n These compounds are solids at room temp

Covalent Bonds n These are INTRAMOLECULAR bonds. n Formed when 2 nonmetal atoms share electrons
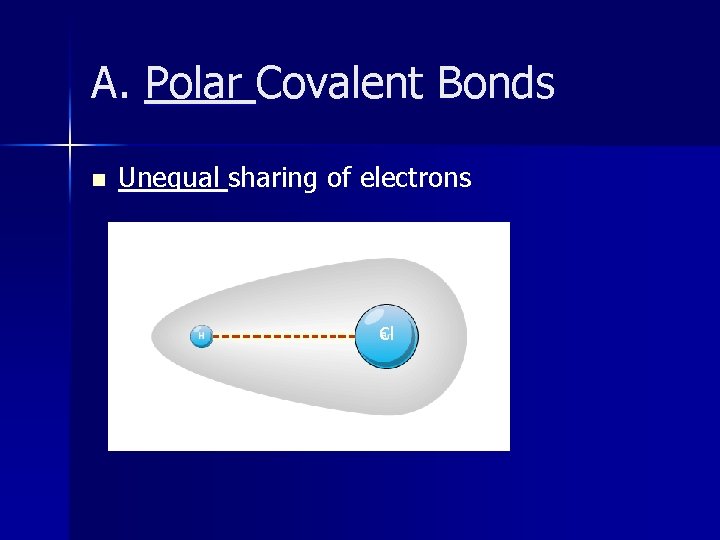
A. Polar Covalent Bonds n Unequal sharing of electrons Cl

Polar Covalent Bonds n Electrons attracted MORE to atom with higher electronegativity. n Polar Covalent Bonds – – – Dissolve in water Don’t fall apart into ions (don’t dissociate) No conductivity! Made of two different non-metals Can be a gas, liquid, or solid

B. Non-polar Covalent Bonds n Equal sharing of electrons Br Br

Non-polar Covalent n n n Electrons are shared equally between two atoms. Usually nonpolar covalent bonds are between diatomic molecules – two of the Same nonmetal Nonpolar covalent compounds – Do not dissolve in water – No conductivity – Can be a gas, liquid, or solid

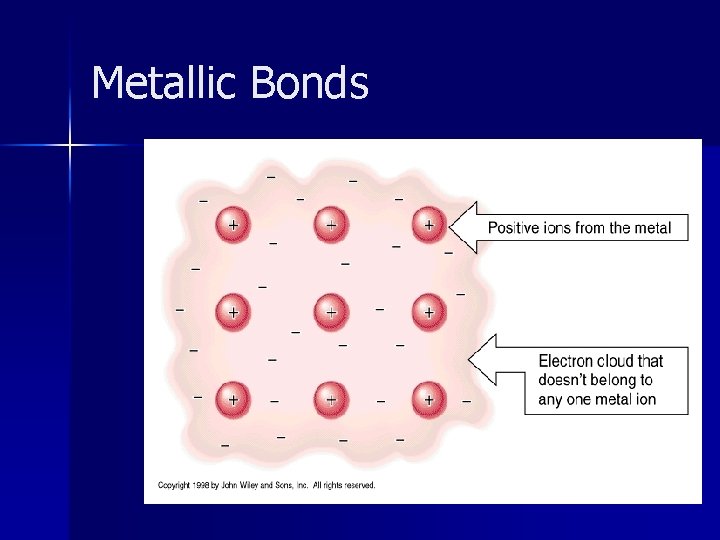
Metallic Bonds n These are INTRAMOLECULAR bonds. n Bond formed between 2 or more metal atoms. n Larger outer electron shells - which overlap

Metallic Bonds Electrons are free to move within electron clouds of all metal ions = Sea of Electrons n Electrons are delocalized – they don’t belong to any one atom anymore.
Metallic Bonds

p. 12 Intermolecular Bonds -occurs between 2 different molecules

Hydrogen Bonds n The intermolecular force in which a hydrogen atom that is bonded to a highly electronegative atom is attracted to an unshared pair of electrons of an electronegative atom in a nearby molecule.

Hydrogen Bonds n One molecule is polar and has Hydrogen in a H-F, H-O or H-N bond n Other molecule has an unshared pair of electrons, usually F, O, N n Hydrogen forms a weak bond with the unshared electron pair of the other molecule
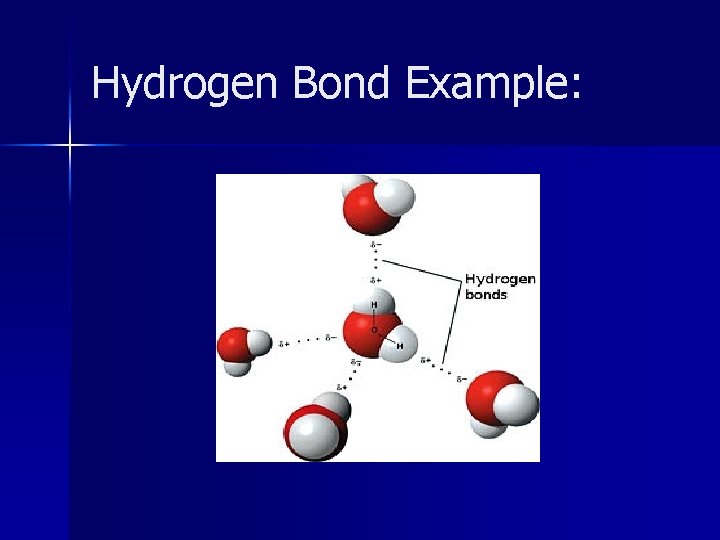
Hydrogen Bond Example:

Van der Waals Bonds These bonds are important for Noble Gases and nonpolar molecules. n Very weak bonds n Electrons of Noble Gases get shifted to one side causing it to attract a slightly positive atom that is nearby. n

Van der Waals Bond Example:
- Slides: 21