IONIC COVALENT AND METALLIC BONDING DAY 8 METALLIC

IONIC, COVALENT, AND METALLIC BONDING DAY 8 – METALLIC BONDING, PROPERTIES OF SUBSTANCES READING, AND GRAPHIC ORGANIZER

WARM UP FIND: Warm Up Page on your table GLUE: In Warm Up into Warm Up Section and answer questions TIME: 4 minutes WHEN DONE: Reread answers to the Metallic

AGENDA REVIEW VENN DIAGRAM REVIEW METALLIC BONDING PROPERTIES OF SUBSTANCES READING DEMONSTRATION: PROPERTIES GRAPHIC ORGANIZER

LEARNING TARGETS 3 - I CAN DESCRIBE HOW IONIC, COVALENT AND METALLIC BONDS ARE FORMED BETWEEN ATOMS 4 - I CAN EXPLAIN HOW INTERACTIONS AT THE LEVEL OF ATOMS, IONS AND MOLECULES RESULT IN DIFFERENT PROPERTIES IN IONIC, COVALENT AND METALLIC SUBSTANCES

REVIEW VENN DIAGRAM FIND: YOUR VENN DIAGRAM IN BLUE BIN TIME: 1 MINUTE WHEN DONE: BE READY TO ADD MORE INFORMATION
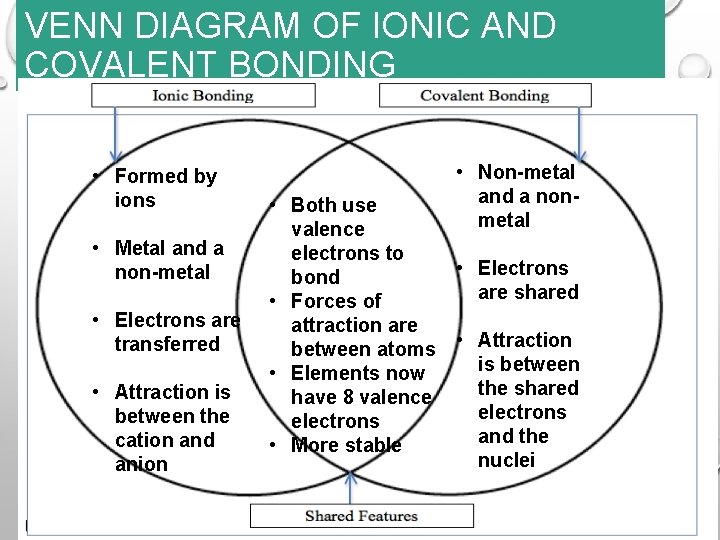
VENN DIAGRAM OF IONIC AND COVALENT BONDING • Formed by ions • Metal and a non-metal • Electrons are transferred • Attraction is between the cation and anion • Non-metal and a nonmetal • Both use valence electrons to • Electrons bond are shared • Forces of attraction are between atoms • Attraction is between • Elements now the shared have 8 valence electrons and the • More stable nuclei

METALLIC BONDING REVIEW 1. How are ions in an ionic crystal different from ions in a metallic crystal? 2. What happens to valence electrons in a metal? 3. What are the free valence electrons called? 4. What holds atoms together in a metallic crystal? 5. Why do metals bend, but not break, when a force is applied to them?

LEARNING TARGET CHECK IN TURN: To the Learning Target for this Unit PICK: Two colors of highlighter First color Highlight: Supporting Targets 3. 1, 3. 2 and 3. 3 Write: Page # Column for “Covalent Tutorial” Second Color Highlight: Supporting Targets 4. 1, 4. 2 and 4. 3 Write in: Page # Column for “Metallic Bonding”

READING: "PROPERTIES OF IONIC, COVALENT AND METALLIC SUBSTANCES” TAKE OUT: PROPERTIES OF SUBSTANCES READING SHOW ME: HOMEWORK • HIGHLIGHT MAIN IDEA IN EACH PARAGRAPH I WILL: STAMP IT FOR FULL CREDIT TIME: 90 SECONDS WHEN DONE: GRAB A GREEN CARD FROM BLUE BIN

READING: "PROPERTIES OF IONIC, COVALENT AND METALLIC SUBSTANCES” REREAD ANNOTATE: PROPERTIES OF SUBSTANCES FOCUS ANNOTATION ON: WHAT SURPRISES YOU? WHAT DOES THE AUTHOR ASSUMER I KNOW? SENTENCE STARTERS FOR MARGIN NOTES: “THIS IS IMPORTANT BECAUSE. . . ” “THIS IS CONNECTED TO. . . ” “I AM CONFUSED ABOUT. . . ” Minimum ONE NOTE PER PARAGRAPH! TIME: 14 MINUTES WHEN DONE: TAPE/GLUE READING INTO NOTEBOOK, ALONG WITH WORK THAT HAS BEEN PASSED BACK (LAB

DEMONSTRATIONS MELTING POINT • WAX NON-POLAR COVALENT SUGAR POLAR COVALENT SALT IONIC SHOT METALLI C DISSOLVE IN WATER • WAX NON-POLAR COVALENT SUGAR POLAR COVALENT SALT IONIC FOIL METALLI C ELECTRICITY • SUGAR POLAR COVALENT SALT IONIC LIGHT BULB METALLI C
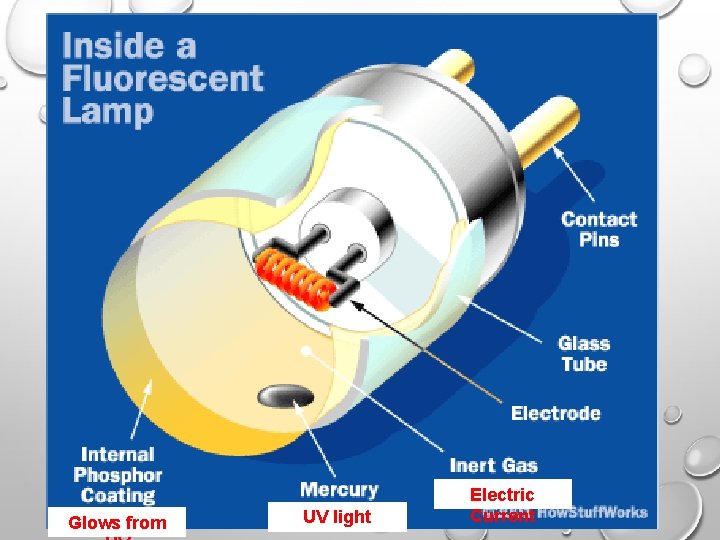
Glows from UV light Electric Current

GRAPHIC ORGANIZER COMPLETE: ALL THREE GRAPHIC ORGANIZERS IDENTIFY: • THE TYPE OF BONDS THAT HOLD TOGETHER ATOMS OF SUBSTANCES WITH SPECIFIC PROPERTIES • WHY THIS TYPE OF BONDING RESULTS IN THESE PROPERTIES WORK: WITH YOUR TABLE GROUP! HELP: EACH OTHER OUT! TIME: UNTIL END OF CLASS WHEN DONE: TURN INTO CLASS BOX AND FIND READING HOMEWORK (TO BE READY FOR CHECK FOR UNDERSTANDING)
- Slides: 13