Ionic Covalent Metallic Bonding Unit 4 Octet Rule

Ionic, Covalent, & Metallic Bonding Unit 4

Octet Rule • Gilbert Lewis used the fact that noble gases are stable, therefore, unreactive to explain why atoms form certain kinds of ions and molecules. • Lewis explained: in forming compounds, atoms tend to achieve the electron configuration of a noble gas. – Octet: a set of 8 – ns 2 np 6 – With Helium being an exception 1 s 2 {stable}

Formation of Cations • Loss of valence electrons • Result: – Produces a positively charged ion – Ion has a pseudo-noble gas configuration • Properties promoting formation of cations: – Large radii – Low ionization energy – Low electronegativity
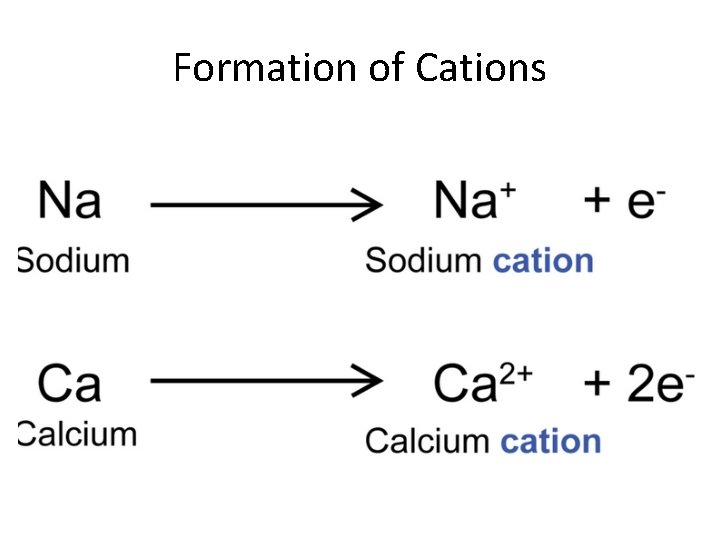
Formation of Cations

Formation of Cations

Formation of Cations • Practice: 1)Mg 2)Al 3)Sr 4)Be

Formation of Anions • Gain of valence electrons • Result: – Produces a negatively charged ion – Ion has a pseudo-noble gas configuration • Properties promoting formation of anions: – Small radii – High ionization energy – High electronegativity

Formation of Anions
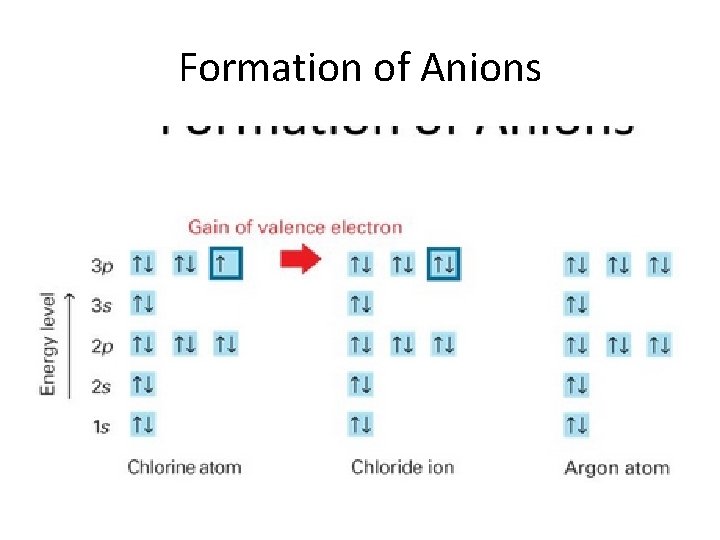
Formation of Anions

Formation of Anions • Practice: 1) N 2) O 3) S 4) F
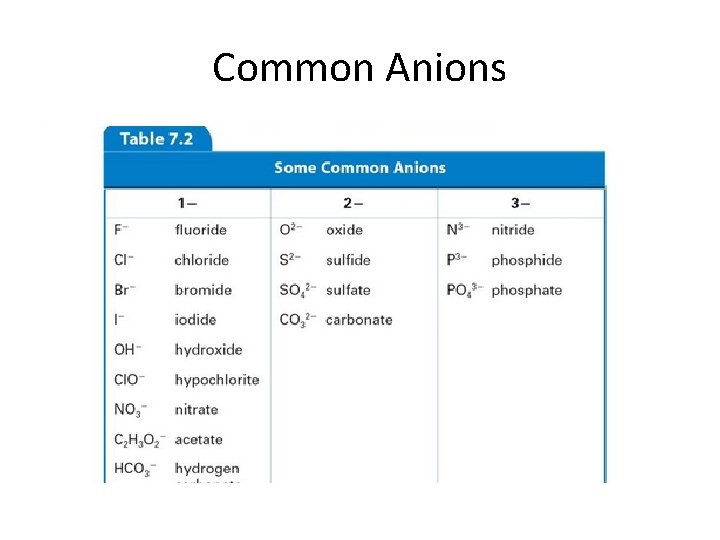
Common Anions
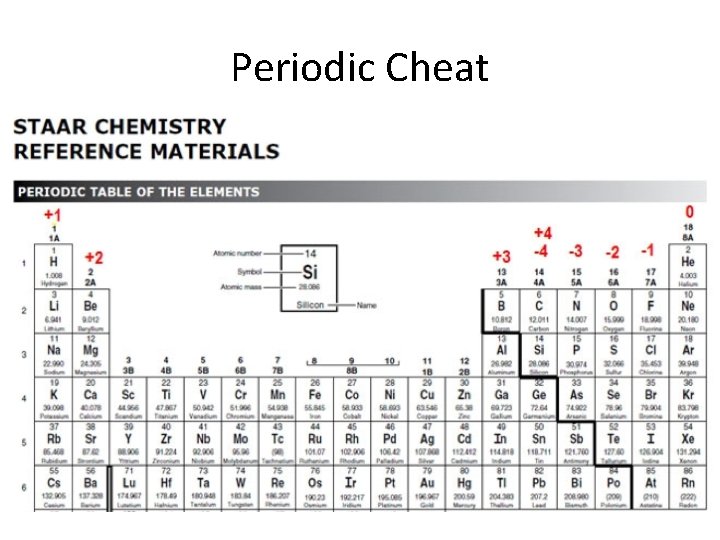
Periodic Cheat

Nomenclature • Cation – Element Name + Cation ØExample: potassium (K) potassium cation (K 1+) • Anion – Element Name – Drop ending and add “-ide” ØExample: chlorine (Cl) chloride (Cl 1 -)

Monatomic Ions Worksheet 1 • See Handout

Formation of Ionic Bonds • Ionic compound: Compound made up of cations(metals) and anions(nonmetals) – Electrically NEUTRAL compounds – Exist as a collection of positively and negatively charged ions arranged in repeating patterns. • Ionic bond: electrostatic forces that hold ions together in an ionic compound. – Cations and anions are oppositely charged/ attracted to one another

Formation of Ionic Compunds

Formation of Ionic Compounds • Chemical Formula: shows the kinds and numbers of atoms in the smallest representative unit of a substance. – Na. Cl ~ 1: 1 ratio of sodium to chlorine atoms • Formula Units: the lowest whole number ratio of ions in an ionic compound.
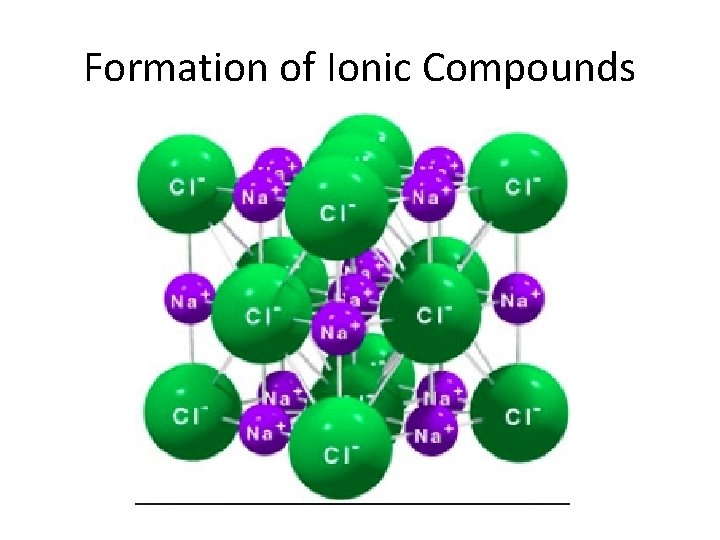
Formation of Ionic Compounds

Ionic Compounds - Formation • • K + Cl Na + F Mg + O Al + F Cs + P Mg + N Fr + S

Ionic Compound Practice • K + Cl 1+ 1 • K + Cl • KCl • Potassium chloride

Ionic Compound Practice • Na + F 1+ 1 • Na + F • Na. F • Sodium Fluoride

Ionic Compound Practice • Mg + O 2+ 2 • Mg + O • Mg. O • Magnesium oxide

Ionic Compound Practice • Al+ F 3+ 1 • Al + F • Al. F 3 • Aluminum fluoride
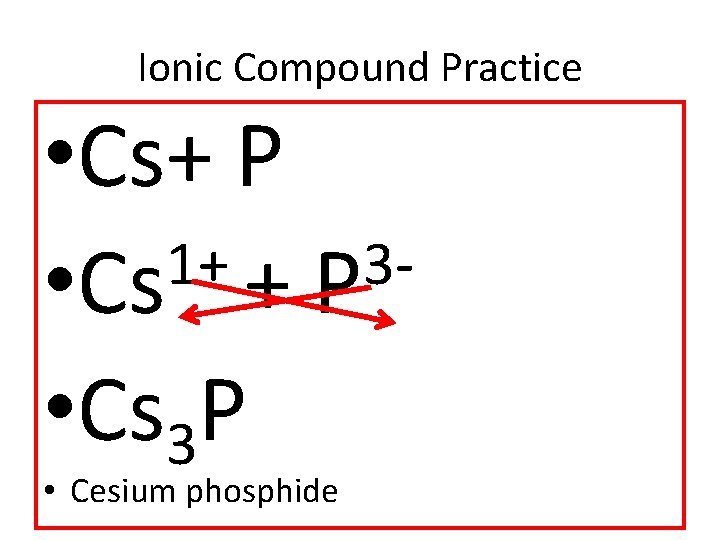
Ionic Compound Practice • Cs+ P 1+ 3 • Cs + P • Cs 3 P • Cesium phosphide

Ionic Compound Practice • Mg+ N 2+ 3 • Mg + N • Mg 3 N 2 • Magnesium nitride
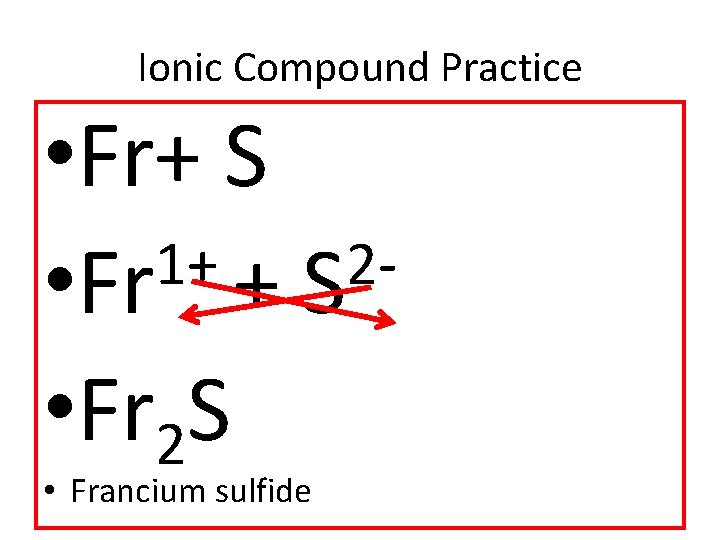
Ionic Compound Practice • Fr+ S 1+ 2 • Fr + S • Fr 2 S • Francium sulfide

Ionic Assessment • Which pairs of elements are likely to form ionic compounds? – Cl, Br – K, He – Li, Cl – I, Na – Al, Br – Cs, K

Ionic Assessment • Write the formula for the following compounds: – Barium chloride – Lithium oxide – Calcium fluoride – Magnesium sulfide

Properties of Ionic Compounds

Simple Binary Ionic Compounds Worksheet 2 • See Handout

3 Atomic Bond Types • Ionic bond • Molecular bond • Metallic bond

Molecular Compounds • Covalent bond: atoms held together by sharing electrons – Made of Molecules (nonmetal + nonmetal) – Diatomic molecule: H, N, O, Halogens – Low melting points/ boiling points – Polar, Nonpolar • Nonpolar: Do NOT conduct electricity – Gas and Liquid at room temp

Metallic Bonding • Metals are made up of closely packed cations • Metallic Bond: Attraction of the free-floating valence electrons for the positively charged metal ions – Forces of attractions that hold metals together. • Valence electrons of metal atoms can be modeled as a sea of electrons

Metallic Bonding • Sea of electrons – Good conductors – Ductile – Malleable • Metal atoms are arranged in very compact and orderly patterns
Alloys of Metals • Mixtures composed of two or more metals • Properties are superior to those of their component elements

Alloys of Metals • Most important alloys are steel. – Iron and Carbon = main components – Corrosion resistance – Ductility – Hardness – Toughness

Ionic Compounds Summary • Ionic bonds: electrostatic forces holding formula units together – Made of Formula Units (cation[metal] + anion[nm]) – Crystalline solids – High melting points – Neutral – Very stable – Dissolved in water will conduct electricity

Key Terms • Formula Units: lowest whole number ratio of ions in an ionic compound • Molecules: Neutral group of atoms joined by covalent bonds • Diatomic molecule: molecule consisting of 2 atoms • Molecular compounds: compounds composed of molecules (2 or more different nonmetals) • Molecular formula: show # atoms in each molecule. • Polar Covalent: electrons are shared unequally (slight charges) • Non. Polar Covalent: atoms in bond pull equally (no charge)
- Slides: 38