Chemical Bonding 3 Types of Bonding Ionic Bonding

Chemical Bonding
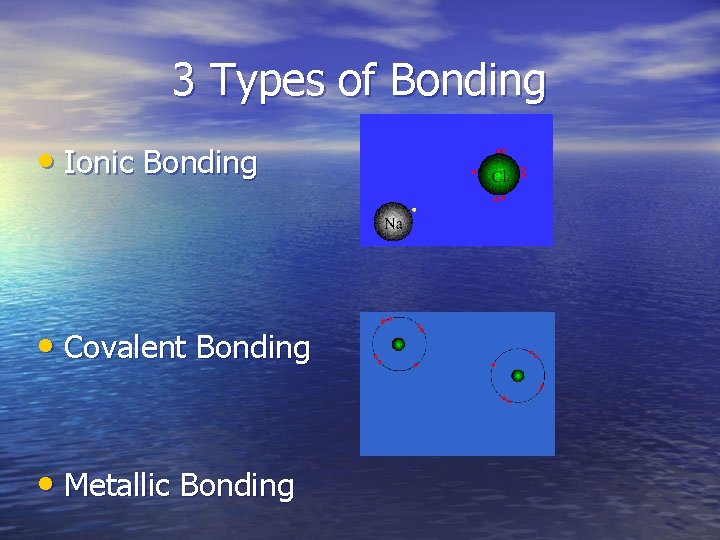
3 Types of Bonding • Ionic Bonding • Covalent Bonding • Metallic Bonding

Ionic Bonding • Ionic bonding is an electrical attraction between two oppositely charged atoms.

• Lose an electron Atom is Positive • Gain an electron Atom is Negative • Positive Ion is called Cation • Negative Ion is called Anion

Characteristics of Ionic Compounds • 1) Crystalline solid at room temperature • 2) Have higher melting and boiling points compared to covalent bonds. • 3)Conduct electrical current in solution state. • 4)Extremely polar bonds. • 5)Most are soluble in water.

Magic # • The magic number is 8 • Every atom wants to gain electrons up to 8, or lose electrons to equal zero, In order to be stable just like the noble gases • Exception= Hydrogen= only needs two electrons

Helpful Hint • An easy way to check if a bond is Ionic: • One atom must be from group 1, 2, or 13 on the periodic table and the other atom from groups 14 -17.

Oxidation Number (Charge) Group 1 2 13 14 15 16 17 18 Charge +1 +2 +3 -4 -3 -2 -1 0

Covalent Bonds • Covalent Bonds are formed as a result of the sharing of one or more pairs of bonding electrons. Each atom donates half of the electrons to be shared.

2 forms of covalent bonds • 1) Polar Covalent - unequal sharing of electrons • 2) Non-Polar Covalent – equal sharing of electrons between atoms.

Single Covalent Bond • A single covalent bond would be the sharing of two electrons between the 2 bonded atoms. • H-H H-Cl F-F • The dash line is symbolic of the bonding pair.

Double Covalent Bond • A double covalent bond is 2 pairs of electrons being shared. • O=O C=O
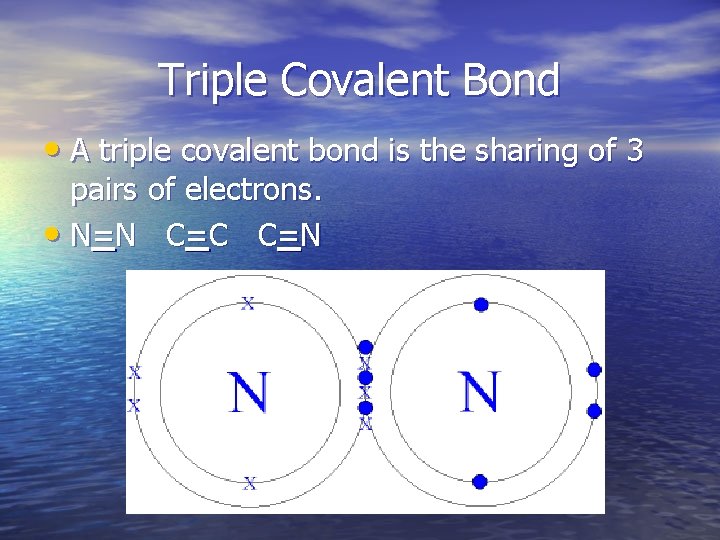
Triple Covalent Bond • A triple covalent bond is the sharing of 3 pairs of electrons. • N=N C =C C =N

Metallic Bond • The metallic bond occurs only between 2 metal atoms. In this type of bond the valence electrons are free to move about the electron clouds of all metals participating in the bond.
- Slides: 14