New concepts in chemical bonding Chargeshift bonding and

New concepts in chemical bonding. Charge-shift bonding and its manifestations in chemistry. Just a study of the single bond… P. C. Hiberty, D. Lauvergnat (Orsay) B. Silvi (Paris 6) S. Shaik (Jerusalem) W. Wu, L. Song, P. Su, F. Ying (Xiamen)

The classical paradigm of the two-electron bond: 1) Covalent bond A • – • B H-H, H 3 C-CH 3, H 2 N-NH 2, HO-OH, H 3 C-H, F-F, Cl-Cl, … 2) Polar bond A +–B H-Cl, HF, Li-H, , H 3 C-OH, H 3 Si-Cl … varying degree of polarity B Na-Cl, … 3) Ionic bond A+

Some commonly accepted views: 1) Reaction barriers A–B potential well B–C potential well A–B + C –> [A--B--C] –> A + B–C “The stronger the bonds that form or break, the larger the barrier”

Some commonly accepted views: 1) Reaction barriers A–B potential well B–C potential well “The stronger the bonds that form or break, the larger the barrier” A–B + C –> [A--B--C] –> A + B–C 2) Electronic density accumulates between the bonded atoms However, a few anomalous facts. . . A B
![Barriers tendencies in radical exchange reactions X • + H–X [X…H…X] • X–H + Barriers tendencies in radical exchange reactions X • + H–X [X…H…X] • X–H +](http://slidetodoc.com/presentation_image_h/a6de8b1747d696f1b24f2fb5b302a0ad/image-5.jpg)
Barriers tendencies in radical exchange reactions X • + H–X [X…H…X] • X–H + X • ∆E≠ (kcal/mol) FHF 20. 9 De(F-H) = 136 kcal/mol Cl. HCl 11. 0 De(Cl-H) = 103 kcal/mol Br. HBr 8. 0 De(Br-H) = 88 kcal/mol
![Barriers tendencies in radical exchange reactions H • + X–H [H…X…H] • H–X + Barriers tendencies in radical exchange reactions H • + X–H [H…X…H] • H–X +](http://slidetodoc.com/presentation_image_h/a6de8b1747d696f1b24f2fb5b302a0ad/image-6.jpg)
Barriers tendencies in radical exchange reactions H • + X–H [H…X…H] • H–X + H • X • + H–X [X…H…X] • X–H + X • ∆E≠ (kcal/mol) HFH FHF 42. 5 20. 9 De(F-H) = 136 kcal/mol HCl 18. 5 11. 0 De(Cl-H) = 103 kcal/mol HBr 8. 0 12. 9 De(Br-H) = 88 kcal/mol
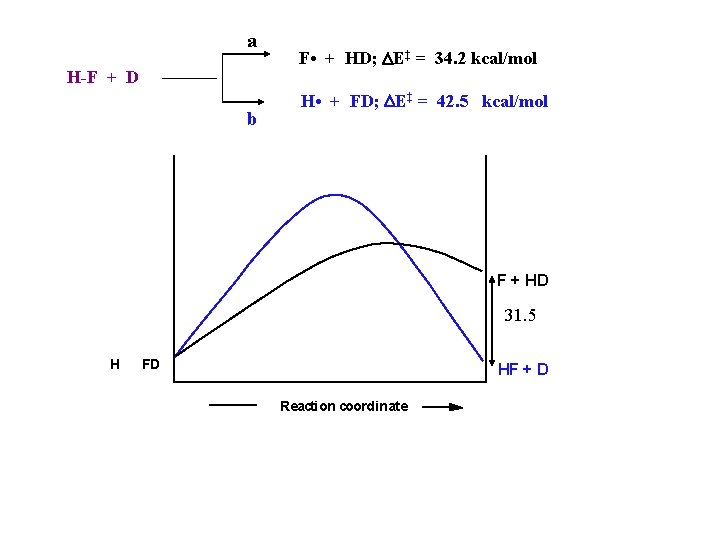
a H-F + D b F • + HD; DE‡ = 34. 2 kcal/mol H • + FD; DE‡ = 42. 5 kcal/mol F + HD 31. 5 H FD HF + D Reaction coordinate
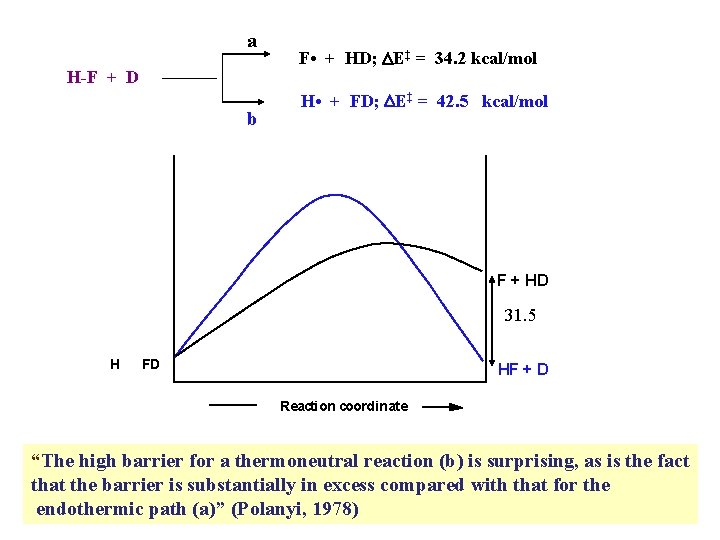
a H-F + D b F • + HD; DE‡ = 34. 2 kcal/mol H • + FD; DE‡ = 42. 5 kcal/mol F + HD 31. 5 H FD HF + D Reaction coordinate “The high barrier for a thermoneutral reaction (b) is surprising, as is the fact that the barrier is substantially in excess compared with that for the endothermic path (a)” (Polanyi, 1978)

20 Other intriguing facts… X X Separate atoms H–H, H 3 C–CH 3 … Density build-up in the bonding region F-F, Cl–Cl, … Deficit of density in the bonding region • Bonding density: positive for H 2, negative for F 2 • “F 2 and Cl 2 have a dominant covalent character, but not a shared-type interaction behavior “(Rincon & Almeida) Question: are the F-F or Cl-Cl bonds of similar natures as H-H or C-C bonds?

10 Quantifying the classical paradigm of the A—X bond: A • – • X covalent A+ : X– ionic A: – X+ ionic YA-X = c 1(A • – • X) + c 2(A+ : X–) + c 3(A: – X+)

10 Quantifying the classical paradigm of the A—X bond: A • – • X covalent A+ : X– ionic A: – X+ ionic YA-X = c 1(A • – • X) + c 2(A+ : X–) + c 3(A: – X+) “Modern” Valence Bond (VBSCF, SCVB) : • Optimizes ci coefficients and orbitals simultaneously (MCSCF) • Unique set of orbitals for all VB structures Not so accurate… De(F 2) = 14 kcal/mol (6 -31+G*) , vs 38 kcal/mol exp.

10 Quantifying the classical paradigm of the A—X bond: A • – • X covalent A+ : X– ionic A: – X+ ionic YA-X = c 1(A • – • X) + c 2(A+ : X–) + c 3(A: – X+) Breathing-Orbital Valence Bond (BOVB) • Optimizes ci coefficients and orbitals simultaneously (MCSCF) • Different orbitals for different VB structures Better… De(F 2) = 34 kcal/mol (6 -31+G*) , vs 38 kcal/mol exp.
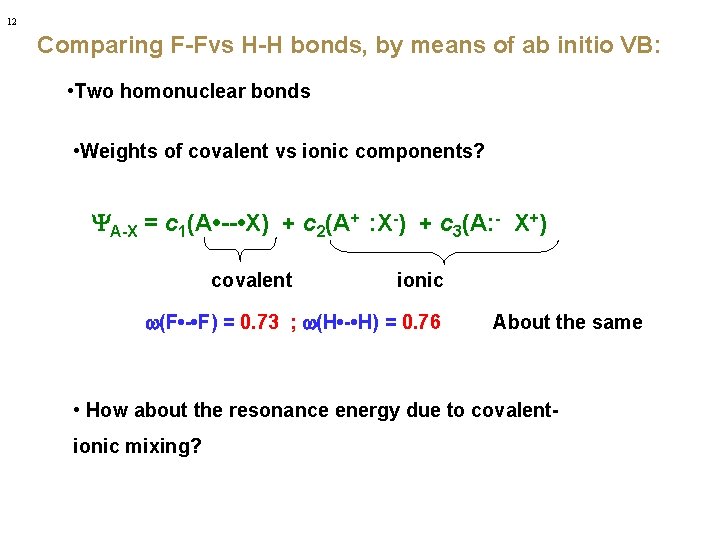
12 Comparing F-Fvs H-H bonds, by means of ab initio VB: • Two homonuclear bonds • Weights of covalent vs ionic components? YA-X = c 1(A • -- • X) + c 2(A+ : X-) + c 3(A: - X+) covalent ionic w(F • - • F) = 0. 73 ; w(H • - • H) = 0. 76 About the same • How about the resonance energy due to covalentionic mixing?

3 The classical paradigm of Covalent-Ionic Superposition (L. Pauling) • The homopolar or weakly polar bond • The covalent form is stabilized by singlet spin-coupling • RE is WEAK. Pauling takes RE= 0 for A = X (e. g. H-H or F-F) • The polar bond • The larger the electronegativity difference, the larger the RE
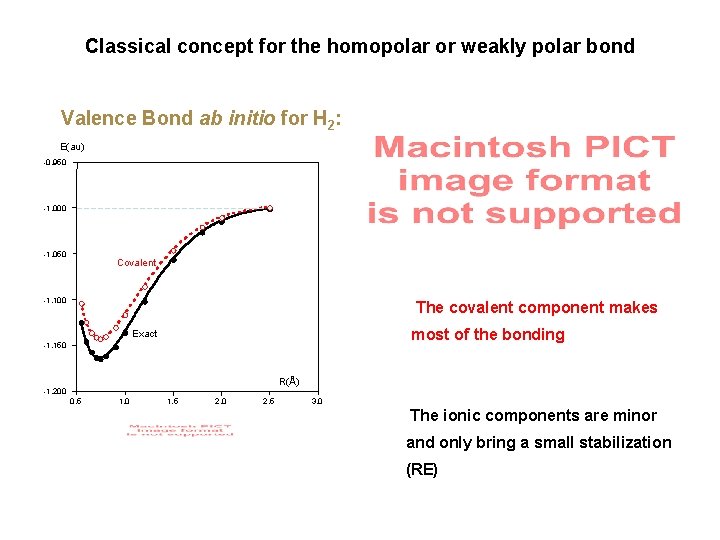
Classical concept for the homopolar or weakly polar bond Valence Bond ab initio for H 2: E(au) -0. 950 -1. 000 -1. 050 Covalent -1. 100 The covalent component makes most of the bonding Exact -1. 150 R(Å) -1. 200 0. 5 1. 0 1. 5 2. 0 2. 5 3. 0 The ionic components are minor and only bring a small stabilization (RE)
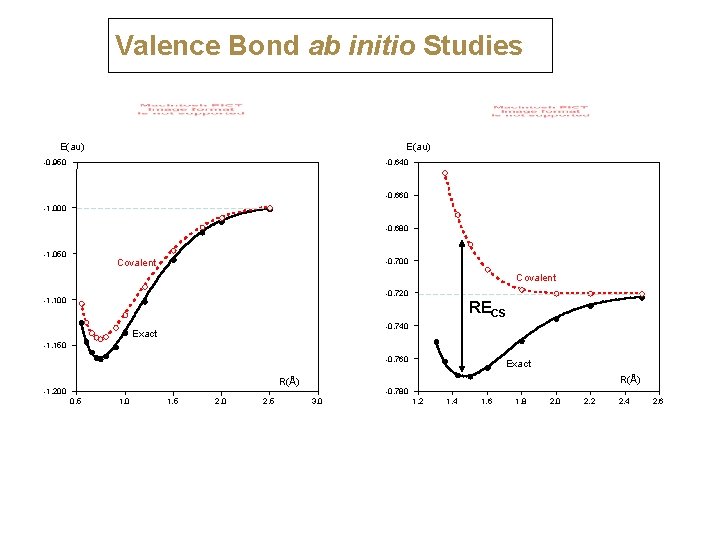
Valence Bond ab initio Studies E(au) -0. 950 -0. 640 -0. 660 -1. 000 -0. 680 -1. 050 -0. 700 Covalent -0. 720 -1. 100 RECS -0. 740 Exact -1. 150 -0. 760 R(Å) -1. 200 0. 5 1. 0 1. 5 2. 0 2. 5 Exact -0. 780 3. 0 1. 2 1. 4 1. 6 1. 8 2. 0 2. 2 2. 4 2. 6
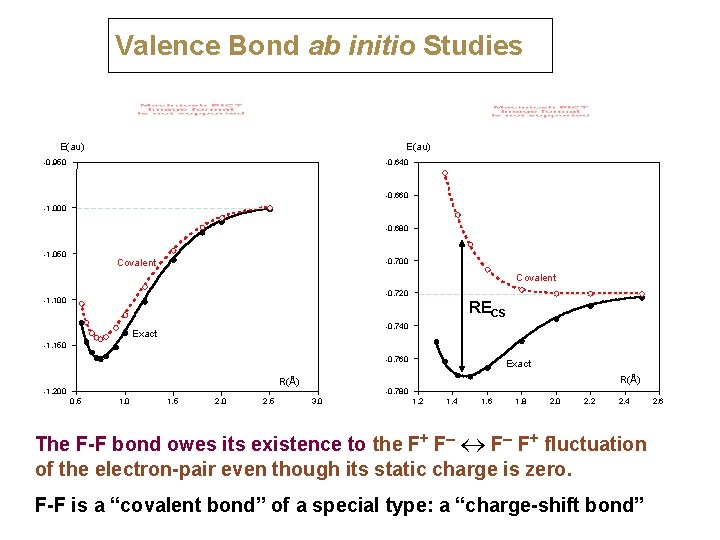
Valence Bond ab initio Studies E(au) -0. 950 -0. 640 -0. 660 -1. 000 -0. 680 -1. 050 -0. 700 Covalent -0. 720 -1. 100 RECS -0. 740 Exact -1. 150 -0. 760 R(Å) -1. 200 0. 5 1. 0 1. 5 2. 0 2. 5 Exact -0. 780 3. 0 1. 2 1. 4 1. 6 1. 8 2. 0 2. 2 2. 4 The F-F bond owes its existence to the F+ F– F+ fluctuation of the electron-pair even though its static charge is zero. F-F is a “covalent bond” of a special type: a “charge-shift bond” 2. 6
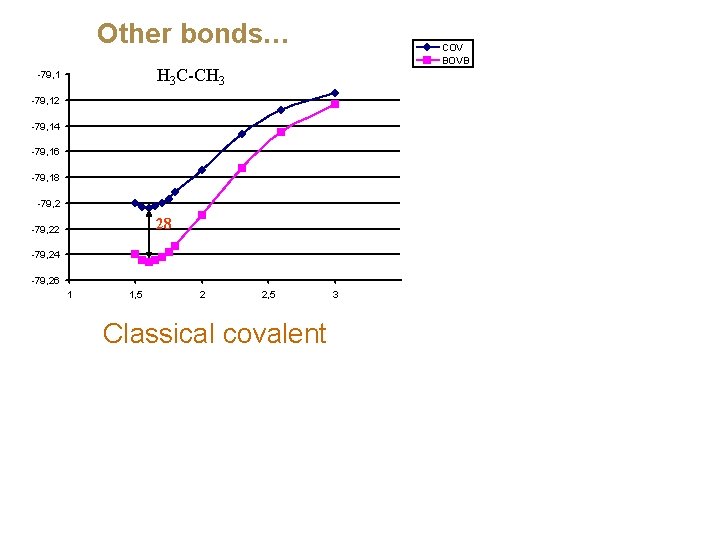
Other bonds… COV BOVB H 3 C-CH 3 -79, 12 -79, 14 -79, 16 -79, 18 -79, 2 28 -79, 22 -79, 24 -79, 26 1 1, 5 2 2, 5 Classical covalent 3
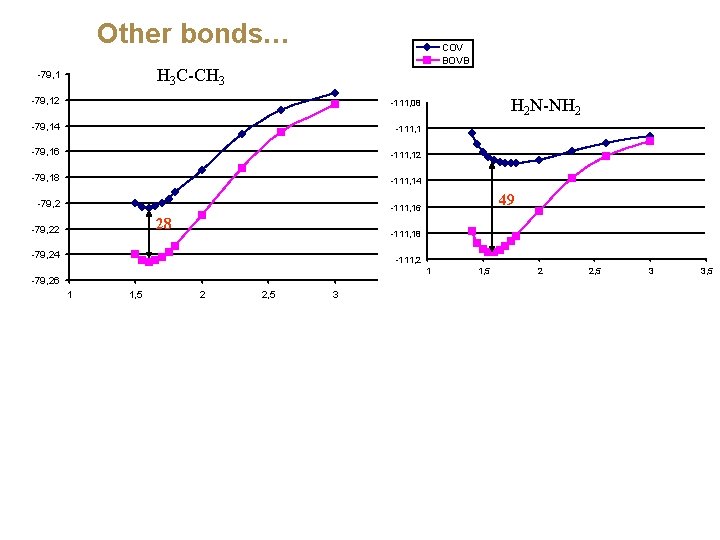
Other bonds… COV BOVB H 3 C-CH 3 -79, 12 -111, 08 -79, 14 -111, 1 -79, 16 -111, 12 -79, 18 -111, 14 -79, 2 49 -111, 16 -198, 55 28 -79, 22 H 2 N-NH 2 -111, 18 -79, 24 -111, 2 -198, 6 -79, 26 1 1, 5 2 2, 5 3 3, 5
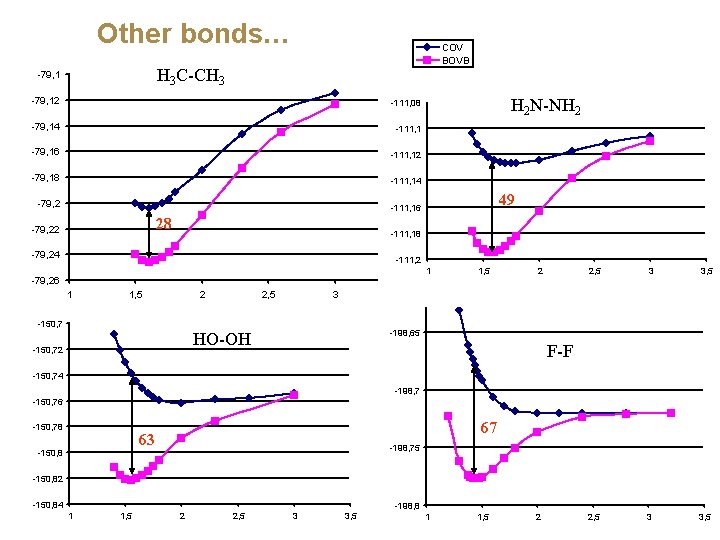
Other bonds… COV BOVB H 3 C-CH 3 -79, 12 -111, 08 -79, 14 -111, 1 -79, 16 -111, 12 -79, 18 -111, 14 -79, 2 49 -111, 16 -198, 55 28 -79, 22 H 2 N-NH 2 -111, 18 -79, 24 -111, 2 -198, 6 -79, 26 1 1, 5 2 -150, 7 2, 5 3 1, 5 2 2, 5 3 3, 5 -198, 65 HO-OH -150, 72 1 F-F -150, 74 -198, 7 -150, 76 -150, 78 67 63 -150, 8 -198, 75 -150, 82 -150, 84 1 1, 5 2 2, 5 3 3, 5 -198, 8 1 1, 5 2

Why is the covalent singlet-coupling less and less bonding from C-C all the way to F-F? The lone-pair bond weakening effect (LPBWE) (Sanderson) N-N LPBWE 53 (kcal/mol) O-O F-F 70 75 CS-bonding appears when there is much Pauli repulsion around the bond
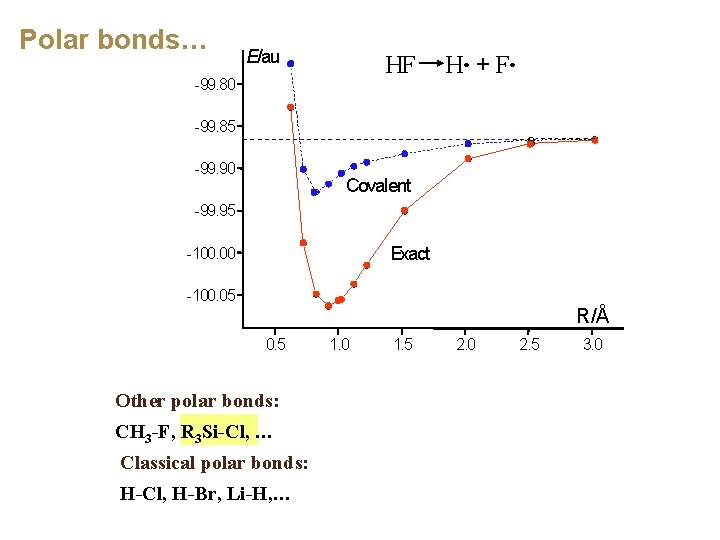
Polar bonds… E/au HF H・ + F・ -99. 80 -99. 85 -99. 90 Covalent -99. 95 Exact -100. 00 -100. 05 R/Å 0. 5 Other polar bonds: CH 3 -F, R 3 Si-Cl, … Classical polar bonds: H-Cl, H-Br, Li-H, … 1. 0 1. 5 2. 0 2. 5 3. 0
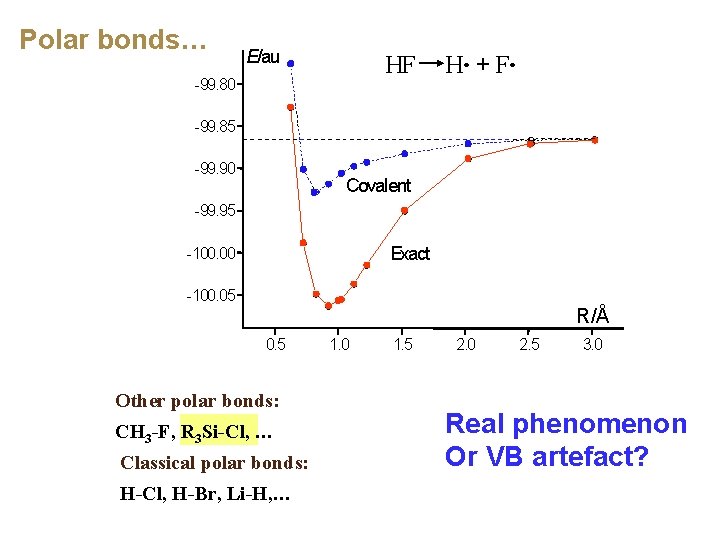
Polar bonds… E/au HF H・ + F・ -99. 80 -99. 85 -99. 90 Covalent -99. 95 Exact -100. 00 -100. 05 R/Å 0. 5 Other polar bonds: CH 3 -F, R 3 Si-Cl, … Classical polar bonds: H-Cl, H-Br, Li-H, … 1. 0 1. 5 2. 0 2. 5 3. 0 Real phenomenon Or VB artefact?
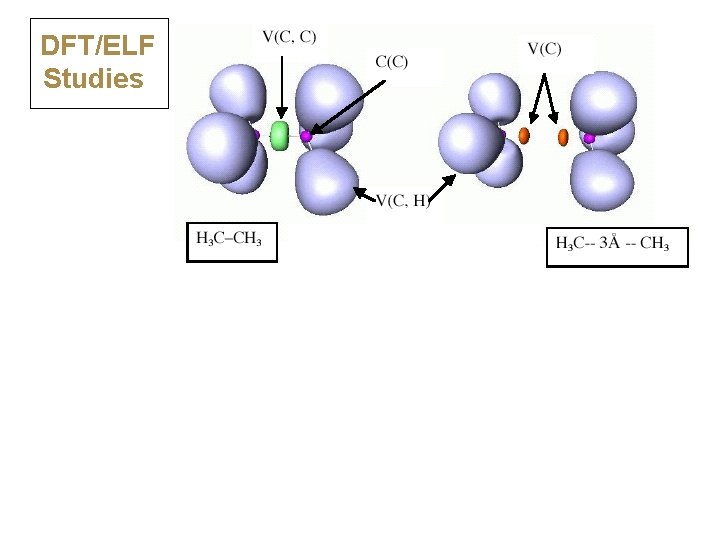
DFT/ELF Studies
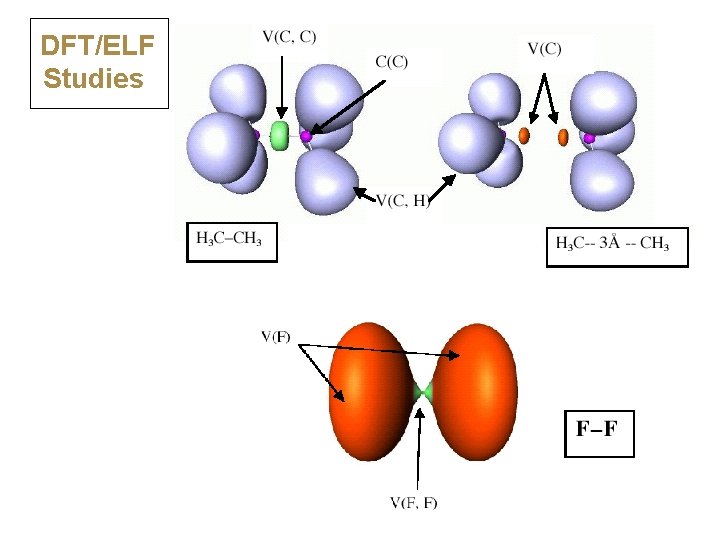
DFT/ELF Studies
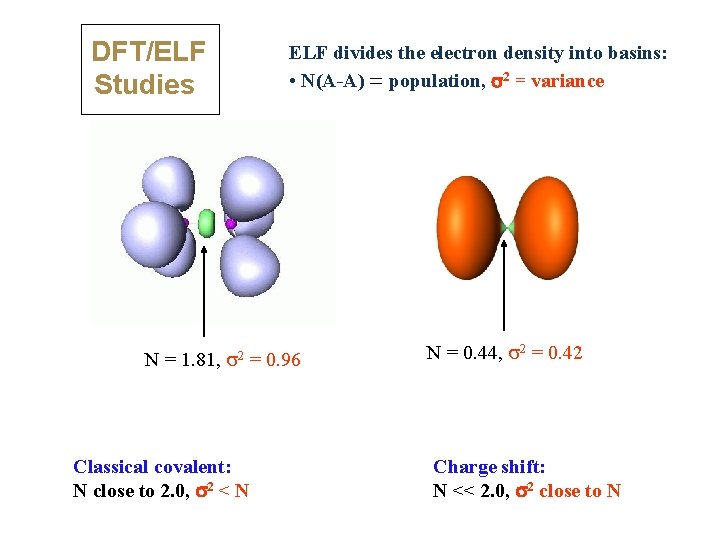
DFT/ELF Studies ELF divides the electron density into basins: • N(A-A) = population, s 2 = variance N = 1. 81, 2 = 0. 96 Classical covalent: N close to 2. 0, s 2 < N N = 0. 44, 2 = 0. 42 Charge shift: N << 2. 0, s 2 close to N
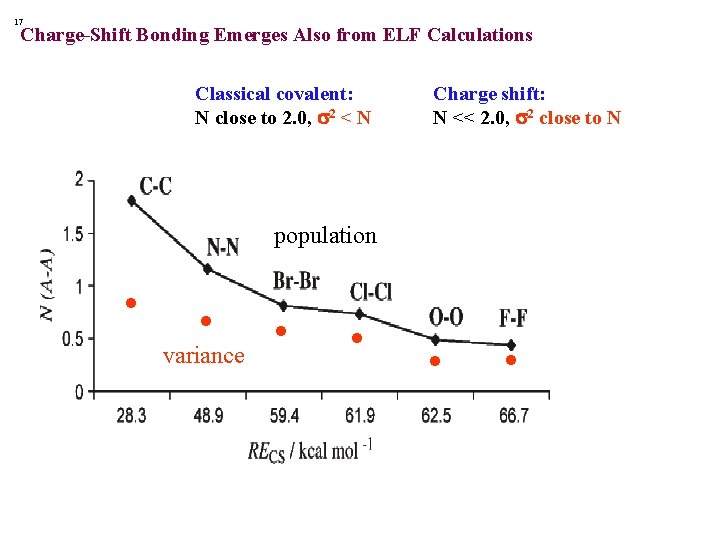
17 Charge-Shift Bonding Emerges Also from ELF Calculations Classical covalent: N close to 2. 0, s 2 < N Charge shift: N << 2. 0, s 2 close to N population • • variance • •

20 AIM theory (Bader) and at the bond critical point at BCP Covalent H-H 0. 27 -1. 39 bonds H 3 C-CH 3 0. 25 -0. 62 Na-Cl 0. 03 +0. 18 (large , < 0) (small , >0) Ionic bond

20 AIM theory (Bader) and at the bond critical point at BCP Covalent H-H 0. 27 -1. 39 bonds H 3 C-CH 3 0. 25 -0. 62 (large , (small , < 0) How about large , F-F 0. 25 +0. 58 Cl-Cl 0. 14 +0. 01 Na-Cl 0. 03 +0. 18 >0? >0) Ionic bond F 2 and Cl 2 are regarded as unexplained exceptions…
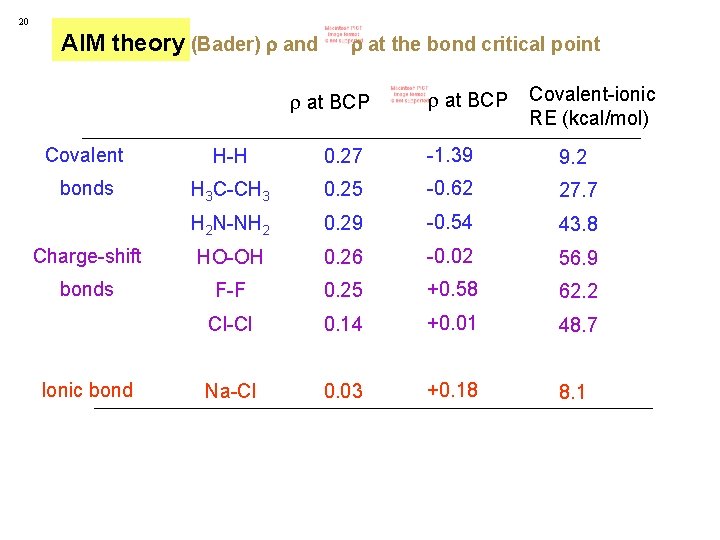
20 AIM theory (Bader) and at the bond critical point at BCP Covalent-ionic RE (kcal/mol) Covalent H-H 0. 27 -1. 39 9. 2 bonds H 3 C-CH 3 0. 25 -0. 62 27. 7 H 2 N-NH 2 0. 29 -0. 54 43. 8 Charge-shift HO-OH 0. 26 -0. 02 56. 9 bonds F-F 0. 25 +0. 58 62. 2 Cl-Cl 0. 14 +0. 01 48. 7 Na-Cl 0. 03 +0. 18 8. 1 Ionic bond
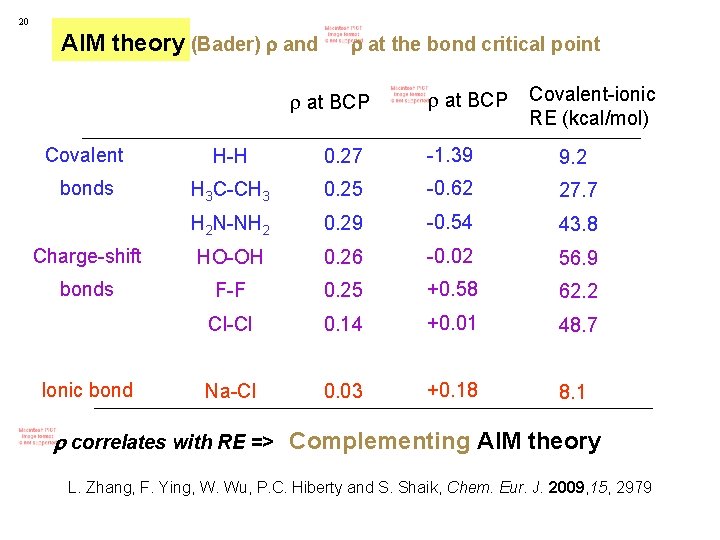
20 AIM theory (Bader) and at the bond critical point at BCP Covalent-ionic RE (kcal/mol) Covalent H-H 0. 27 -1. 39 9. 2 bonds H 3 C-CH 3 0. 25 -0. 62 27. 7 H 2 N-NH 2 0. 29 -0. 54 43. 8 Charge-shift HO-OH 0. 26 -0. 02 56. 9 bonds F-F 0. 25 +0. 58 62. 2 Cl-Cl 0. 14 +0. 01 48. 7 Na-Cl 0. 03 +0. 18 8. 1 Ionic bond correlates with RE => Complementing AIM theory L. Zhang, F. Ying, W. Wu, P. C. Hiberty and S. Shaik, Chem. Eur. J. 2009, 15, 2979
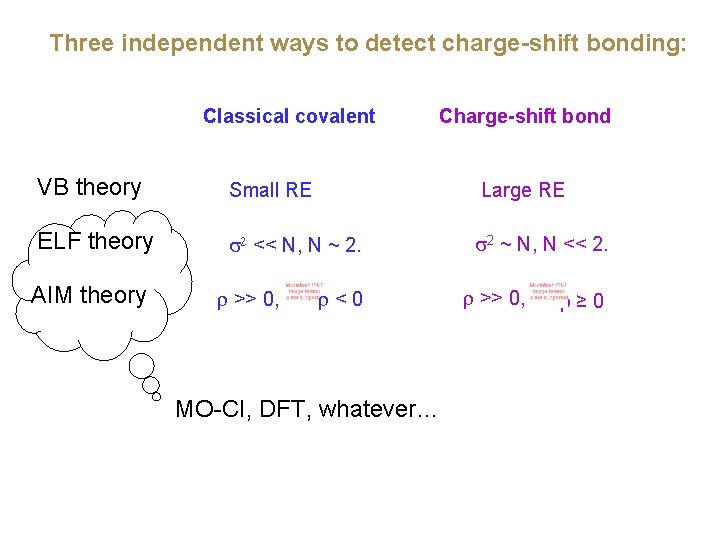
Three independent ways to detect charge-shift bonding: Classical covalent Charge-shift bond VB theory Small RE Large RE ELF theory << N, N ~ 2. 2 ~ N, N << 2. AIM theory >> 0, <0 MO-CI, DFT, whatever… >> 0, ≥ 0

Charge-shift bonding mechanism typifies Electronegative compact atoms (F, O, N) Lone-pair rich atoms (S, Cl, Br, etc) Some polar bonds: F–H , Si–Cl … but NOT all (e. g. Li-H) Some transition metals or bonds

Any experimental manifestations? Charge-shift bonding mechanism typifies Electronegative compact atoms (F, O, N) Lone-pair rich atoms (S, Cl, Br, etc) Some polar bonds: F–H , Si–Cl … but NOT all (e. g. Li-H) Some transition metals or bonds
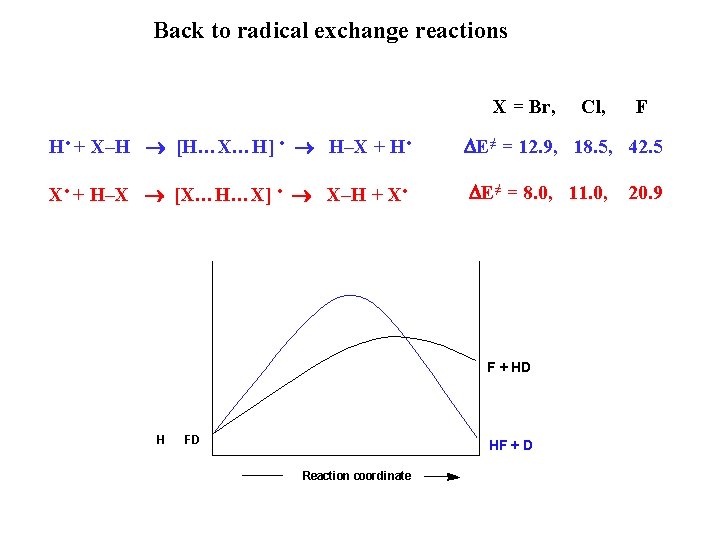
Back to radical exchange reactions X = Br, Cl, F H • + X–H [H…X…H] • H–X + H • DE≠ = 12. 9, 18. 5, 42. 5 X • + H–X [X…H…X] • X–H + X • DE≠ = 8. 0, 11. 0, F + HD H FD HF + D Reaction coordinate 20. 9

VB statecrossing diagrams

The covalent curves H • … • FH F • … • HF H …F • – • H + mirror image F …H • – • F + mirror image On the basis of covalent structures alone, both reactions should have the same barriers

The covalent curves H • … • FH F • … • HF H …F • – • H + mirror image F …H • – • F + mirror image Calculated covalent barriers (kcal/mol): 52. 0 kcal/mol 56. 6 kcal/mol

The ionic curves H. . F–. . H+ + mirror image F. . H+. . F– + mirror image
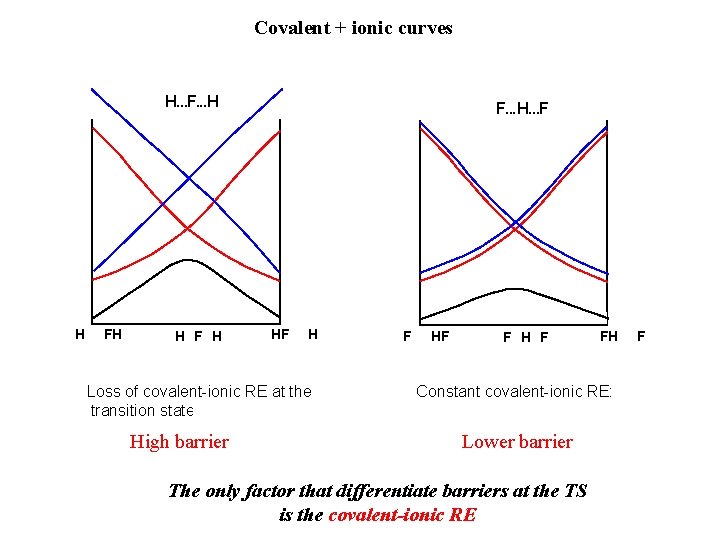
Covalent + ionic curves H. . . F. . . H H F H F. . . H. . . F HF H Loss of covalent-ionic RE at the transition state => high barrier High barrier F HF F H F FH Constant covalent-ionic RE: Lower barrier The only factor that differentiate barriers at the TS is the covalent-ionic RE F
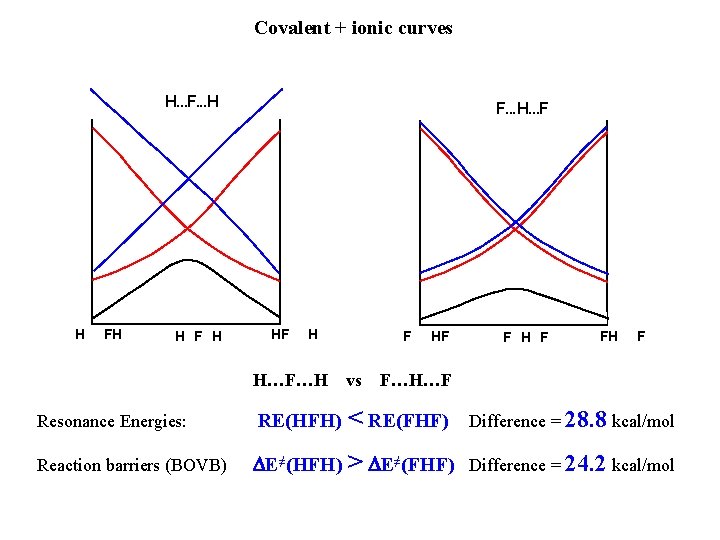
Covalent + ionic curves H. . . F. . . H H F H F. . . H. . . F HF H H…F…H F vs HF F H F F…H…F Resonance Energies: RE(HFH) < RE(FHF) Reaction barriers (BOVB) DE≠(HFH) > DE≠(FHF) Difference = 24. 2 kcal/mol Difference = 28. 8 kcal/mol

87. 5 30. 3 18. 3 RE(H-X) There is a correlation between: - the difference in barriers between HXH and XHX reactions - and the covalent-ionic resonance energy in HX

87. 5 30. 3 18. 3 DDE≠ = 0. 25 RE(H-X) DDE≠( Eq. 1) DDE≠( CCSD(T)) RE(H-X) (1) HFH-FHF HCl. H-Cl. HCl HBr. H-Br. HBr 21. 9 21. 6 7. 5 4. 6 4. 9 DDE≠ is an experimental probe of the covalent-ionic resonance energy in the H-X bond

Another experimental manifestation of charge-shift bonding… Free R 3 C+ cations exist in water… R 3 Si+ Free R 3 Si+ cations are extremely rare P. Su, L. Song, W. Wu, S. Shaik and P. C. Hiberty, J. Phys. Chem. A 2008, 112, 2988

Easy heterolysis of (CH 3)3 C-Cl in water (SN 1)… ∆G≠ = 19. 5 kcal/mol Easy because the C-Cl bond is polar… (CH 3)3 C+ is a free cation In solution

(CH 3)3 Si-Cl does not heterolyse in solution Bond polarities : (CH 3)3 C–Cl < (CH 3)3 Si–Cl Whatever R, the free R 3 Si+ ion does not exist in solution
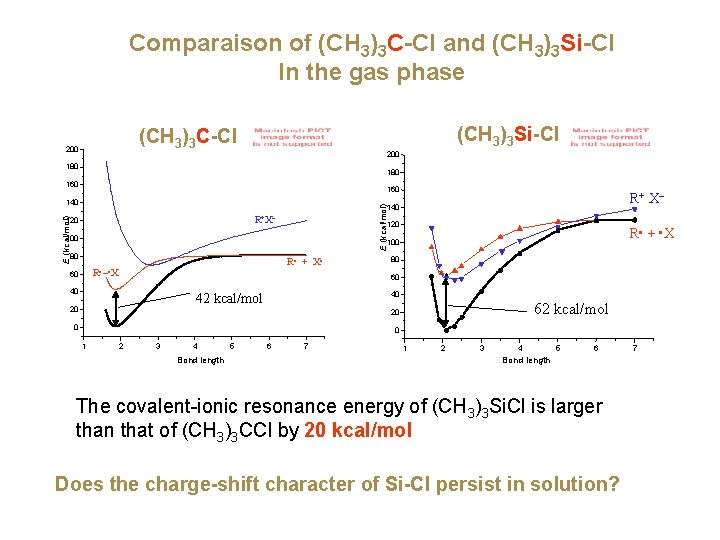
Comparaison of (CH 3)3 C-Cl and (CH 3)3 Si-Cl In the gas phase (CH 3)3 Si-Cl (CH 3)3 C-Cl 200 180 160 140 R+ X– E (kcal/mol) 140 R+X– E (kcal/mol) 120 100 R • + • X 100 80 R • + X • R • – • X 60 80 60 40 40 42 kcal/mol 20 62 kcal/mol 20 0 0 1 2 3 4 Bond length 5 6 7 1 2 3 4 5 6 Bond length The covalent-ionic resonance energy of (CH 3)3 Si. Cl is larger than that of (CH 3)3 CCl by 20 kcal/mol Does the charge-shift character of Si-Cl persist in solution? 7
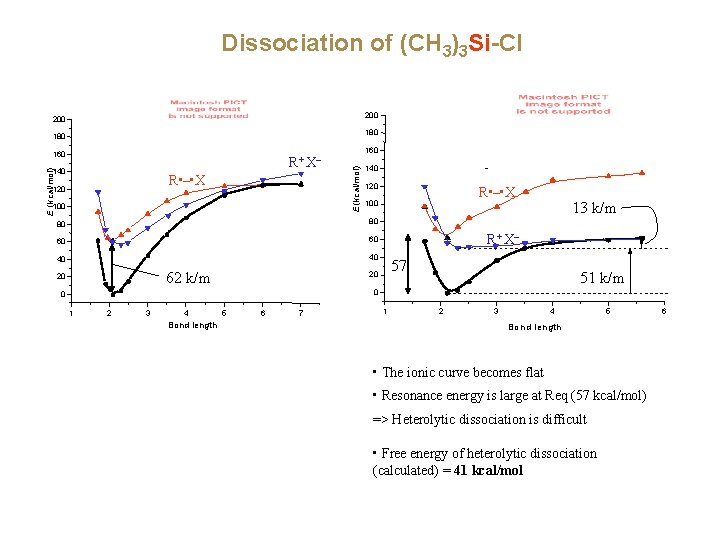
Dissociation of (CH 3)3 Si-Cl 200 180 R+ X– E (kcal/mol) 140 R • – • X 120 100 160 E (kcal/mol) 160 140 120 80 80 60 60 40 40 62 k/m 20 R • – • X 100 13 k/m R+ X– 57 20 51 k/m 0 0 1 2 3 4 Bond length 5 6 7 1 2 3 4 5 Bond length • The ionic curve becomes flat • Resonance energy is large at Req (57 kcal/mol) => Heterolytic dissociation is difficult • Free energy of heterolytic dissociation (calculated) = 41 kcal/mol 6
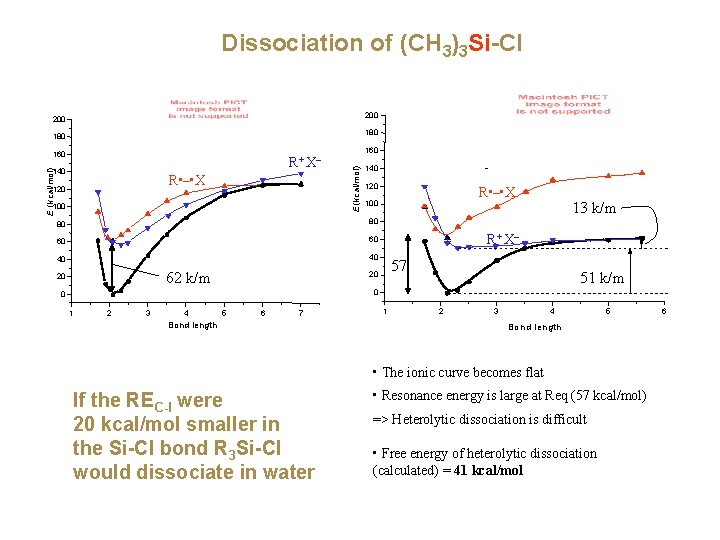
Dissociation of (CH 3)3 Si-Cl 200 180 R+ X– E (kcal/mol) 140 R • – • X 120 100 160 E (kcal/mol) 160 140 120 80 80 60 60 40 40 62 k/m 20 R • – • X 100 13 k/m R+ X– 57 20 51 k/m 0 0 1 2 3 4 5 6 7 Bond length 1 2 3 4 5 Bond length • The ionic curve becomes flat If the REC-I were 20 kcal/mol smaller in the Si-Cl bond R 3 Si-Cl would dissociate in water • Resonance energy is large at Req (57 kcal/mol) => Heterolytic dissociation is difficult • Free energy of heterolytic dissociation (calculated) = 41 kcal/mol 6
![The problem of “inverted bonds” in propellanes -2 H • [1. 1. 1] propellane The problem of “inverted bonds” in propellanes -2 H • [1. 1. 1] propellane](http://slidetodoc.com/presentation_image_h/a6de8b1747d696f1b24f2fb5b302a0ad/image-50.jpg)
The problem of “inverted bonds” in propellanes -2 H • [1. 1. 1] propellane
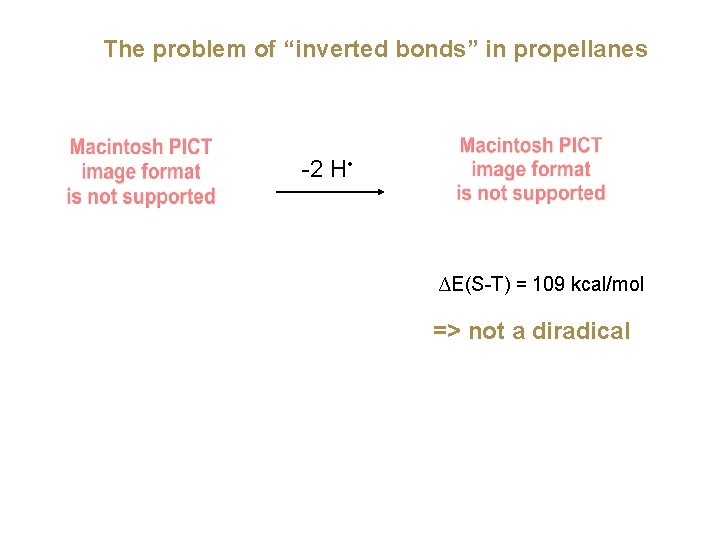
The problem of “inverted bonds” in propellanes -2 H • ∆E(S-T) = 109 kcal/mol => not a diradical
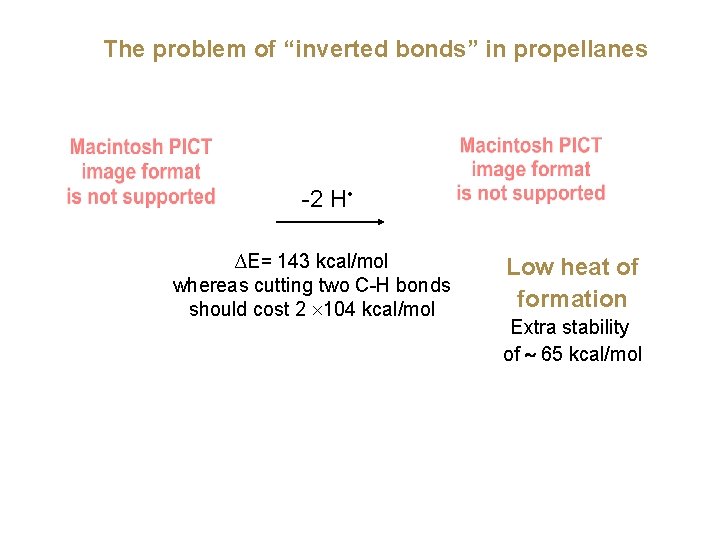
The problem of “inverted bonds” in propellanes -2 H • ∆E= 143 kcal/mol whereas cutting two C-H bonds should cost 2 ´ 104 kcal/mol Low heat of formation Extra stability of ~ 65 kcal/mol
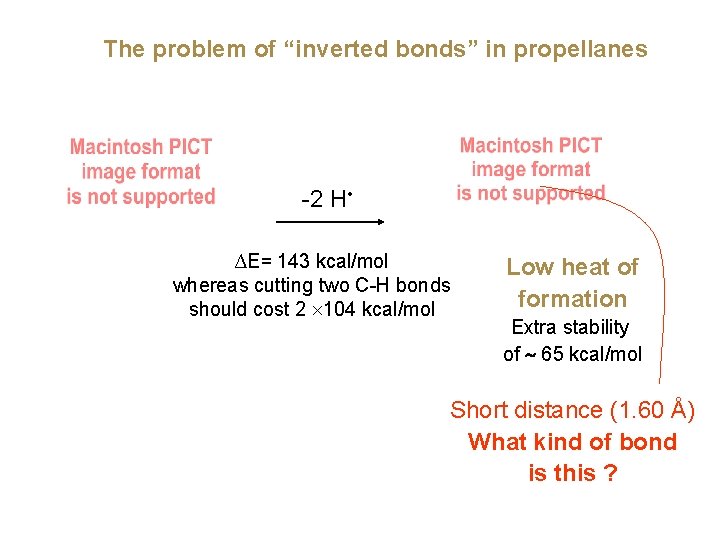
The problem of “inverted bonds” in propellanes -2 H • ∆E= 143 kcal/mol whereas cutting two C-H bonds should cost 2 ´ 104 kcal/mol Low heat of formation Extra stability of ~ 65 kcal/mol Short distance (1. 60 Å) What kind of bond is this ?

What kind of bond is it? Controversies in the literature • There is no bond ! - Hybrid AOs (sp 1. 5) are outward directed

What kind of bond is it? Controversies in the literature • There is no bond ! - Hybrid AOs (sp 1. 5) are outward directed - Depleted electron density between the carbons (from density differences with spherical carbons)

What kind of bond is it? Controversies in the literature • There is no bond ! - Hybrid AOs (sp 1. 5) are outward directed - Depleted electron density between the carbons - Positive = -13. 0 at bond critical point = +10. 3

What kind of bond is it? Controversies in the literature • There is no bond ! - Hybrid AOs (sp 1. 5) are outward directed - Depleted electron density between the carbons - Positive = -13. 0 at bond critical point • There is a bond ! - low heat of formation E. R. Davidson: = +10. 3

Classical covalent : ~ 2. 0, << , < 0 Charge-shift : << 2. 0, ~ > 0 small

Valence bond calculations (BOVB) The covalent curve is repulsive

Valence bond calculations (BOVB) The covalent curve is repulsive The resonance energy is huge A typical charge-shift bond

Valence bond calculations (BOVB) A typical charge-shift bond The three CH 2 bridges create a lot of Pauli repulsion around the C--C bond => This is reminiscent of … Overlap repulsion around the bond in both cases > 0
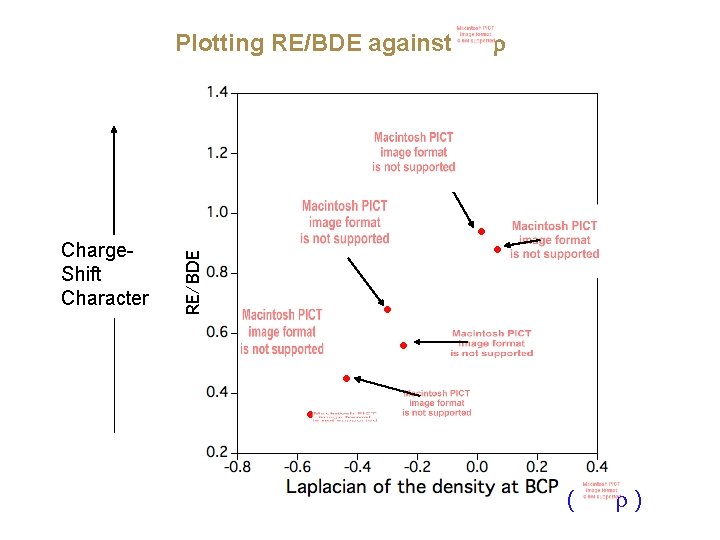
Plotting RE/BDE against Charge. Shift Character ( )

Charge-shift bonds • A category of its own - Hidden in MO/CI calculations - Revealed by VB Theory - Confirmed by ELF and /or AIM analysis of DFT • Have some experimental manifestations - densities - some reaction barriers - Charge-shift bonds resist heterolysis - highly stressed « inverted » bonds - other funny phenomena ?
A three-center interaction… Pr. B. Silvi, Dr. B. Braida, Dr. D. Lauvergnat Pr. S. Shaik Pr. W. Wu & collaborators
- Slides: 64