Change of Phase Chapter 17 Evaporation Evaporation A































- Slides: 33

Change of Phase Chapter 17

Evaporation

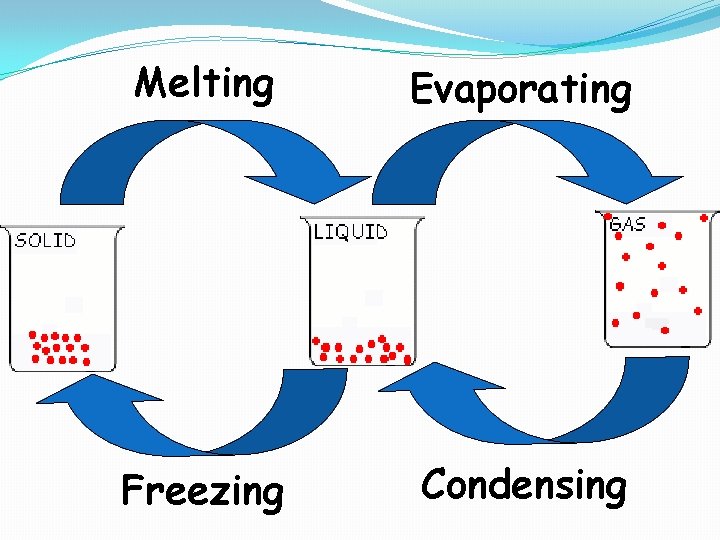
Evaporation �A change of phase from liquid to gas that occurs at the surface of a liquid.

�The temperature of any substance is related to the average kinetic energy of its particles. �Molecules within a liquid have a variety of speeds �Molecules within a liquid also move in all directions

�Molecules at the surface gain kinetic energy while others lose kinetic energy �Molecules gain KE at the surface by bumping into each other

�Increased KE of molecules bumped hard enough to break free from the liquid comes from the molecules remaining in the liquid. �When molecules bumped into one another �Some gain KE �Others lose KE � The average KE of the molecules remaining in the liquid is lowered � Evaporation is a cooling process

�The fast molecules that break free from the surface are slowed as they fly away due to their attraction to the surface. �So although water is cooled by evaporation, the air above is not correspondingly warmed by the process.

Condensation

Condensation �The changing of a gas to a liquid �This is the opposite of evaporation

�When gas molecules near the surface of a liquid are attracted to the liquid, they strike the surface with increased KE and become apart of the liquid �Excess KE is shared with the liquid �This increases the liquids temperature �Condensation is a warming process

Condensation in the Atmosphere �There is always some water vapor in the air �A measure of the amount of this water vaopr is called humidity

Fog and Clouds �Warm air rises. �As it rises it expands. �As it expands it chills �As the air chills water-vapor molecules are slowed. �Lower-speed molecular collisions result in water molecules sticking together. �If there are larger and slower-moving particles or ions present, water vapor condenses upon these particles, and with sufficient buildup we have a cloud

Boiling

�Evaporation can take place beneath the surface of a liquid, forming bubbles of vapor that are buoyed to the surface, where they escape. �This change of phase throughout a liquid rather than only at the surface is called boiling.

�Bubbles in the liquid can form only when the pressure of the vapor within the bubble is great enough to resist the pressure of the surrounding liquid �So bubbles do not form until the boiling point is reached

�If the pressure is increased, the molecules in the vapor must move fast to exert enough pressure to keep the bubble form collapsing. �Extra pressure can be provided either by going deeper below the surface of the liquid �Like in geysers �Or by increasing the air pressure above the liquids surface �Which is how a pressure cooker works.

Melting Evaporating Freezing Condensing

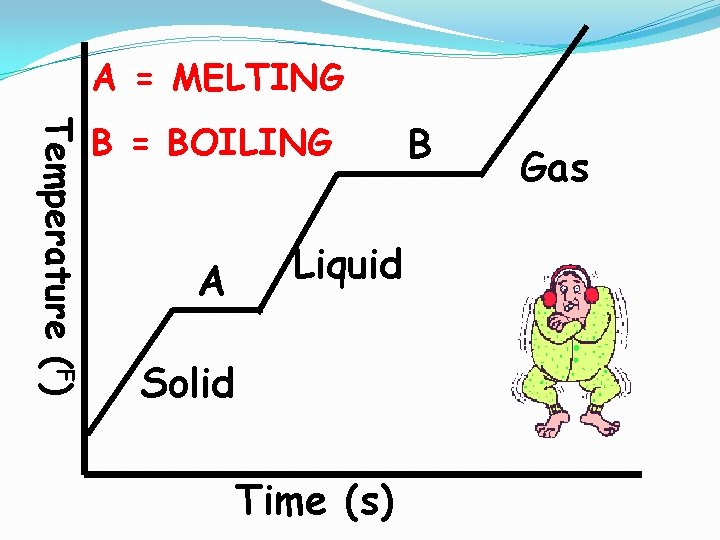
A = MELTING Temperature (F) B = BOILING A Liquid Solid Time (s) B Gas

Melting & Freezing

�Suppose that you are holding hands with someone and both of you start jumping. �What happens as you start to jump around randomly?

�Something like this happens to the molecules of a solid when it is heated. �As heat is absorbed, the molecules vibrate more and more violently. �If enough heat is absorbed, the attractive forces between the molecules will no longer be able to hold them together �The solid melts

�Freezing is the opposite of this process �Energy is withdrawn from a liquid, molecular motion diminishes until finally the molecules, on the average are moving slowly enough so that the attractive forces between them are able to cause cohesion. �The molecules then vibrate about a fixed position and form a solid

�What would happen if you were to add salt or sugar to water? �Would it freeze faster, slower, the same? �Why?

Regelation

�Melting under pressure and freezing again when the pressure is reduced �The ability to do this is one of the properties of water that distinguishes if from other materials �Can you think of any examples?

Energy and Changes of Phase

�If we continually add heat to a sold or liquid, the solid or liquid will eventually change phase �Energy input is required for both liquefaction of the solid and vaporization of a liquid �Energy must be extracted to reverse the process

�A refrigerator is a heat pump that “pumps” heat from a cold environment to a warm one. �This is accomplished by a liquid of low boiling point, the refrigerant, which is pumped into the cooling unit, where it turns into a gas

�Heat pumps of various designs are increasingly being utilized to heat and cool homes �What these heat pumps have in common with one another is that they function like a standard refrigerator.

�An air conditioner is a heat pump in reverse. �Employing the same principles, it simply pumps heat energy from inside the home to the outside

Quick Check �In order to become a liquid a solid must _______ energy. �In order to become a liquid a gas must ____ energy.

Heat of Fusion �Either the energy needed to separate molecules from the solid phase or the energy released when bonds form in a liquid that change it to the solid phase

Heat of Vaporization �Either the energy required to separate molecules from the liquid phase or the energy released when gas condenses to the liquid phase