Phases of Matter Energy and Phase Changes Phase


























- Slides: 37

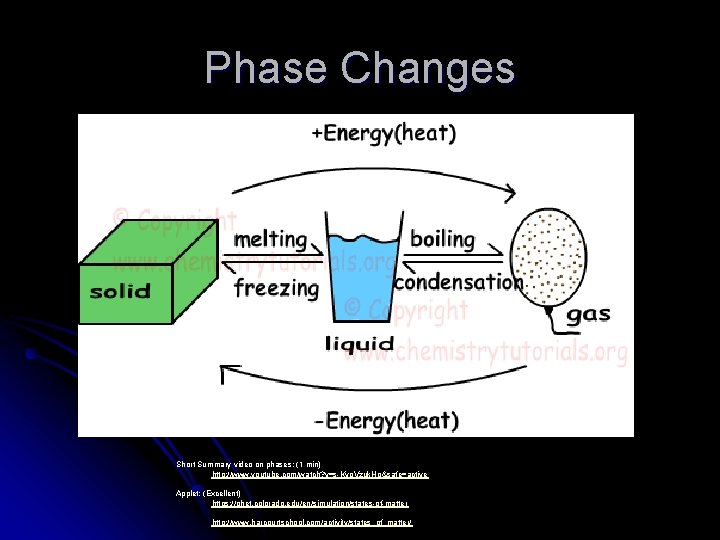
Phases of Matter, Energy and Phase Changes

Phase of Matter l . Depends on strength of forces of attraction between particles.

Solids l Definite shape and volume. Most dense phase (Exception is water!) Difficult to compress. Particles vibrate in fixed positions Crystalline lattice structure. Most attraction between particles. l Note: l l l Amorphous solids include glass, plastic, wax, and silly putty

Liquids Definite volume l No definite shape l Hard to compress l Particles slide past each other l Forces of attraction between particles still high l

Gases l l l No definite shape or volume Expands to fill container Lowest density Little attraction between particles “Vapor” = a gaseous state of something that is normally liquid l (Ex: water vapor)

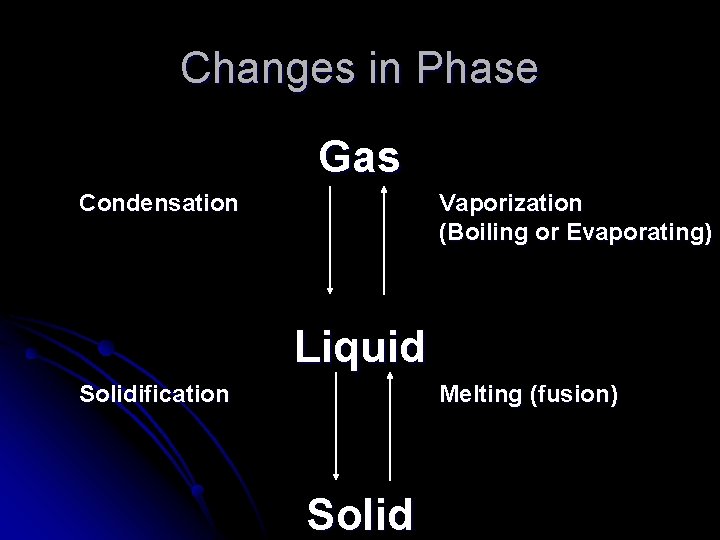
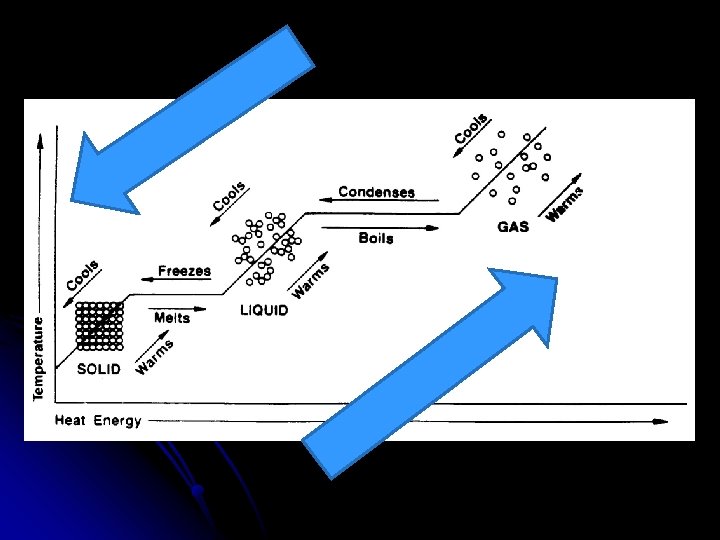
Changes in Phase Gas Condensation Vaporization (Boiling or Evaporating) Liquid Solidification Melting (fusion) Solid

Phase Changes Short Summary video on phases: (1 min) http: //www. youtube. com/watch? v=s-Kvo. Vzuk. Ho&safe=active Applet: (Excellent) https: //phet. colorado. edu/en/simulation/states-of-matter http: //www. harcourtschool. com/activity/states_of_matter/

Let’s Skip a Phase l Sublimation l Directly from the solid phase to the gas phase. l Happens with substances with weak intermolecular forces of attraction l They separate easily! l Ex: CO 2(s) dry ice, Iodine CO 2(s) → CO 2 (g) http: //www. youtube. com/watch? v=8 t. HOVVg. Gkpk

Energy l Energy = capacity to do work or produce heat. It can be anything that causes matter to move or change direction. l l l Many different types of energy Ex: electrical, thermal, atomic, mechanical “Chemical” energy is the potential energy stored in the bonds between atoms

Law of Conservation of Energy l Energy can’t be created or destroyed, just transferred from one form to another

PE vs. KE l Potential Energy stored energy l l Energy can be stored in bonds between atoms Kinetic Energy energy of motion l All atoms are moving and vibrating unless at absolute zero

Energy and Changes to Matter l Exothermic Change: l l A + B → C + D + energy Energy is released or “ex”its Endothermic Change: A + B + energy → C + D l Energy is absorbed or “en”ters

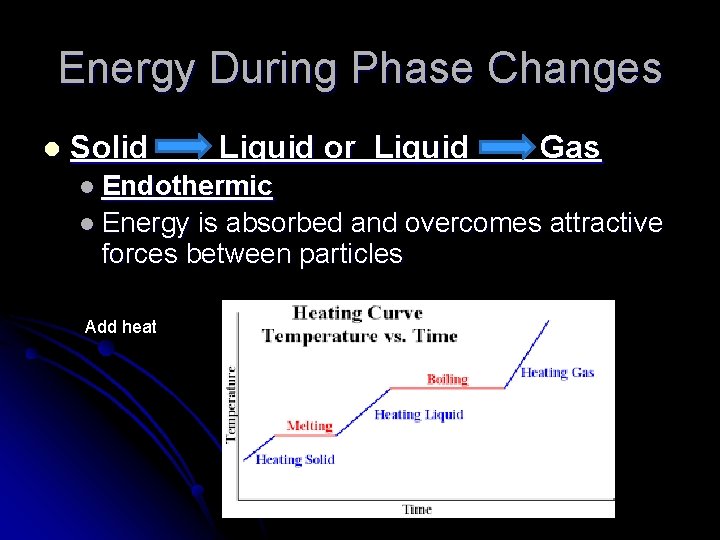
Energy During Phase Changes l Solid Liquid or Liquid Gas l Endothermic l Energy is absorbed and overcomes attractive forces between particles Add heat

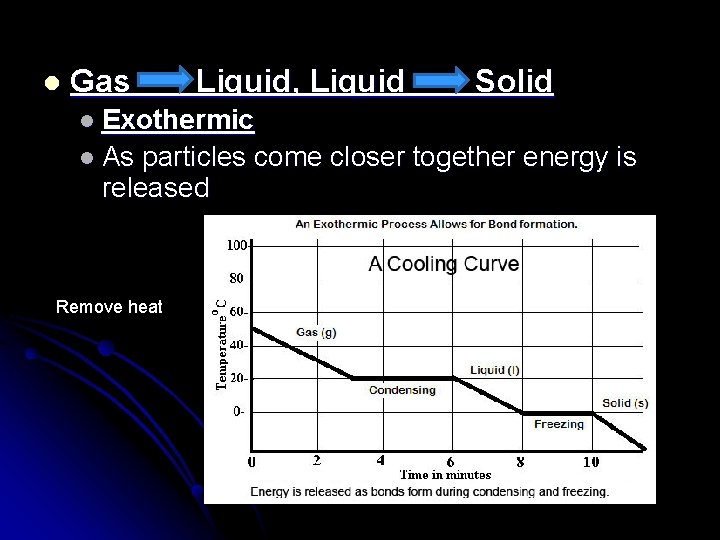
l Gas Liquid, Liquid Solid l Exothermic l As particles come closer together energy is released Remove heat


Heat Energy l Also called Thermal energy, it makes particles move more as it is added l Measured in Joules or calories. http: //www. youtube. com/watch? v=f 1 e. AOyg. DP 5 s&safe=active

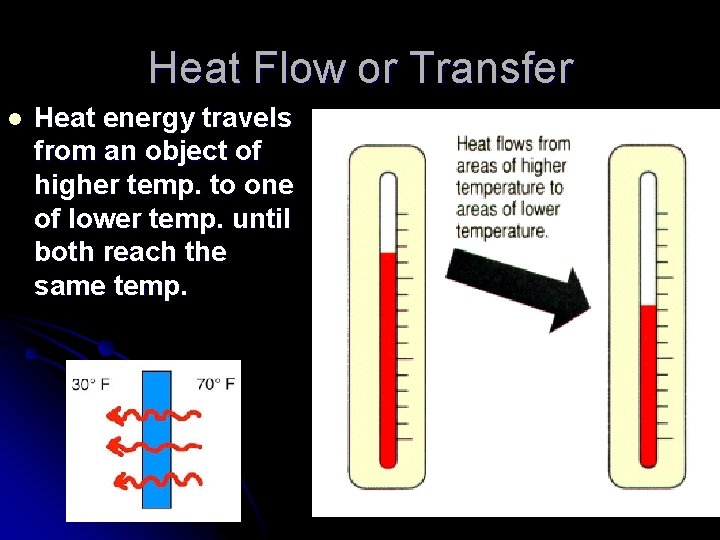
Heat Flow or Transfer l Heat energy travels from an object of higher temp. to one of lower temp. until both reach the same temp.


Temperature l Measure of the average kinetic energy (motion) of all the particles in a sample. l Not a form of energy!!! l But if you add heat energy or take it away, it causes particles to move faster or slower and thus changes the temp. Heat vs Temp. https: //www. youtube. com/watch? v=yx. BTE Mnr. ZZk

Heat vs. Temperature l Teacup vs. Bathtub l Both at 25˚C l Which one contains more heat energy? l Which one has the greater average KE?

Temperature Scales Used in Chemistry Celsius l Fixed points of scale based on the freezing point and boiling point of water l 0 °C = water freezes, 100 °C = water boils Kelvin l Scale based on lowest temperature possible l 0 K = absolute zero https: //www. youtube. com/watch? v=-G 9 Fd. Nq. UVBQ

Temperature Scales and Conversions K = ˚C + 273

Absolute Zero l Temperature at which particles have slowed down so much they no longer possess any kinetic energy. 0 Kelvin -273° Celsius

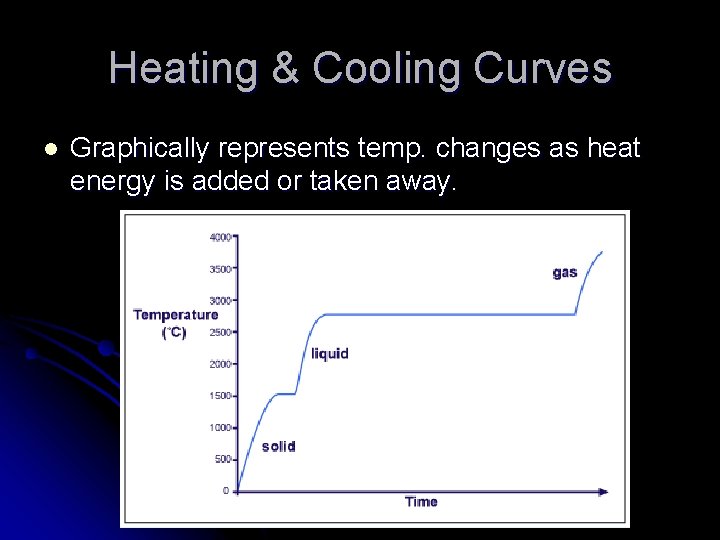
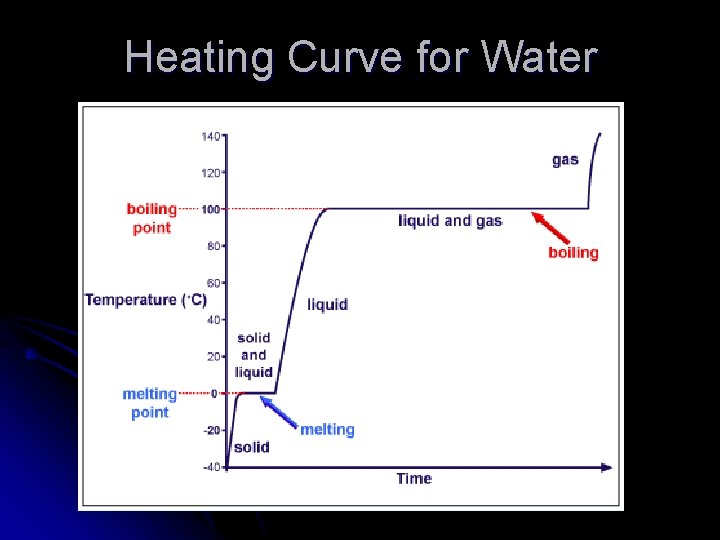
Heating & Cooling Curves l Graphically represents temp. changes as heat energy is added or taken away.

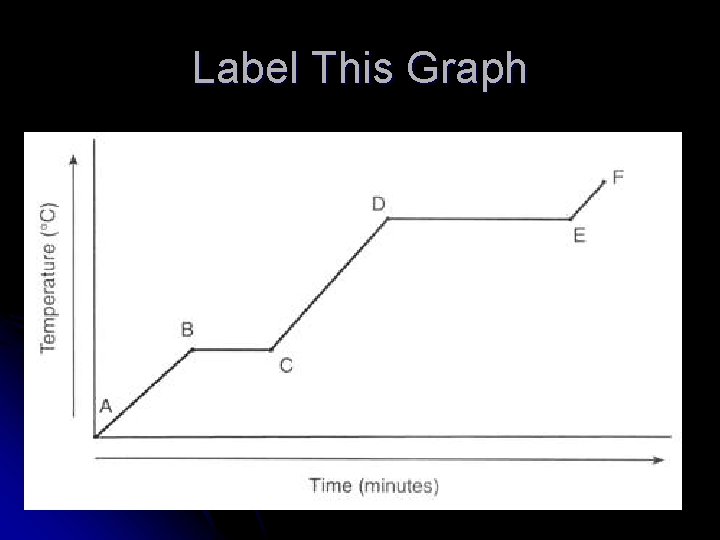
Label This Graph

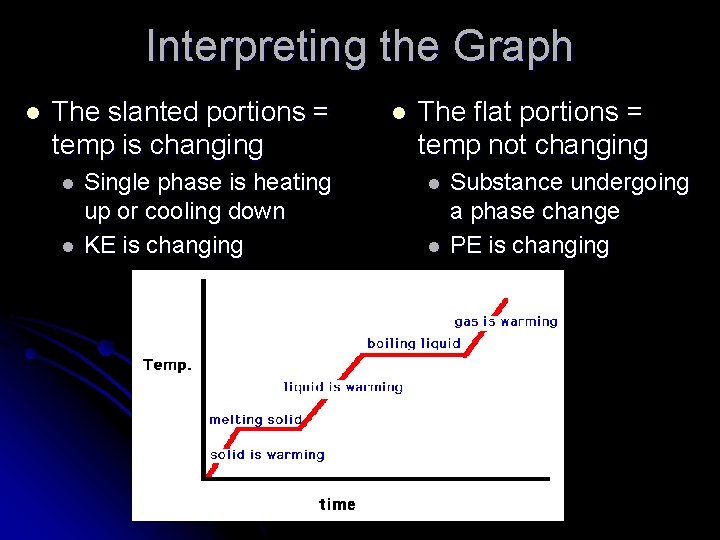
Interpreting the Graph l The slanted portions = temp is changing l l Single phase is heating up or cooling down KE is changing l The flat portions = temp not changing l l Substance undergoing a phase change PE is changing

Heating Curve for Water

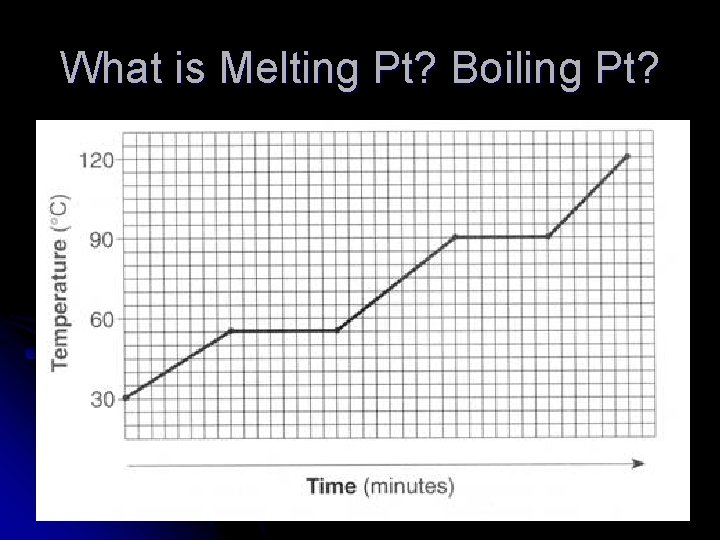
What is Melting Pt? Boiling Pt?

Heat Equations l Calculates the energy involved when a substance changes in temperature or undergoes a phase change. l Use this when temperature of substance changes use this formula:

l When Undergoing Phase Change use one of these formulas: TEMPERATURE CONSTANT l. Q = m. Hf Use when changing from solid to liquid (melting) or liquid to solid (freezing) l. Q = m. Hv Use when changing from liquid to gas (vaporization) or gas to liquid (condensing)

Physical Constants for Water Table B Use these constants in Heat Equations Hf = heat of fusion = 334 J/g Hv = heat of vaporization = 2260 J/g Specific Heat Capacity (“c”) = 4. 18 J/g x K

What is Specific Heat Capacity? Specific Heat: “c” Joules of heat needed to raise 1 gram of a substance 1°C. l Substances have different abilities to absorb heat when energy is applied depending on their composition. Ex: Piece of Iron vs. Water.

Calorimeter l Instrument to determine amount of heat lost or gained in a reaction by measuring changes in the temp. of water surrounding the system. Virtual Calorimetry http: //group. chem. iastate. edu/G reenbowe/sections/projectfolder /flashfiles/thermochem/heat_me tal. html Q = mcΔT

Multi-step Heat Problems (Honors) l Need to use more than one of the heat equations and add up the total heat. l Note: Specific Heat of different phases of water! l H 2 O(s) = 2. 10 J/gx°C l H 2 O (l) = 4. 18 J/gx°C l H 2 O(g) = 1. 84 J/gx°C l Ex: Calculate the heat energy to raise 10 grams of water at -25°C to 80°C. l Draw a heating curve. Figure out # of steps. l 1. ) Heat ice from -25° to 0° q = mcΔT l 2. ) Melt ice to liquid at 0° q = m. Hf l 3. ) Heat liquid water from 0° to 80° q = mcΔT


Heat Lost = Heat Gained (Honors) l When two objects of different temperatures are placed together in a closed system, heat flows from hotter to colder object until they reach same temperature. mcΔT = mcΔT l Total heat lost = total heat gained

Try This!! l Online App Demonstrates Specific Heat and Calorimetry l http: //elearning. classof 1. com/demo/2 D_Lab/Chemistry/specific. Heat/ specific. Heat. html