Evaporation Evaporation is a process of vaporizing large









- Slides: 13

Evaporation • Evaporation is a process of vaporizing large quantities of liquids (volatile) to get a concentrated product. . • Evaporation is the removal of solvent from the liquids (solution/mixture/aq. extract) by boiling the liquor in a suitable vessel and withdrawing the vapours, leaving the concentrated liquid residue in the vessel. • Equipment used called Evaporators

• Application • Manufacture of bulk drug • Manufacture of biological products

• Factors Influencing Evaporation 1. Temperature 2. Vapor Pressure 3. Surface Area 4. Moisture content in the feed 5. Type of product required 6. Time of evaporation 7. Film and Deposits

• Classification I. Evaporators with heating Medium in Jacket eg. , Steam jacketed kettle (evaporating Pan) II. Evaporators with tubular heating surface A. Evaporators with tubes placed horizontally eg. , horizontal tube evaporator B. Evaporators with tubes placed vertically i) Evaporators with short tubes a) single effect Evaporators eg. , short tube vertical Evaporators b) multiple effect evaporators. , triple effect Evaporators ii) Evaporators with long tubes a) Evaporators with natural circulation. , climbing film Evaporators b) Evaporators with forced circulation. , forced circulation


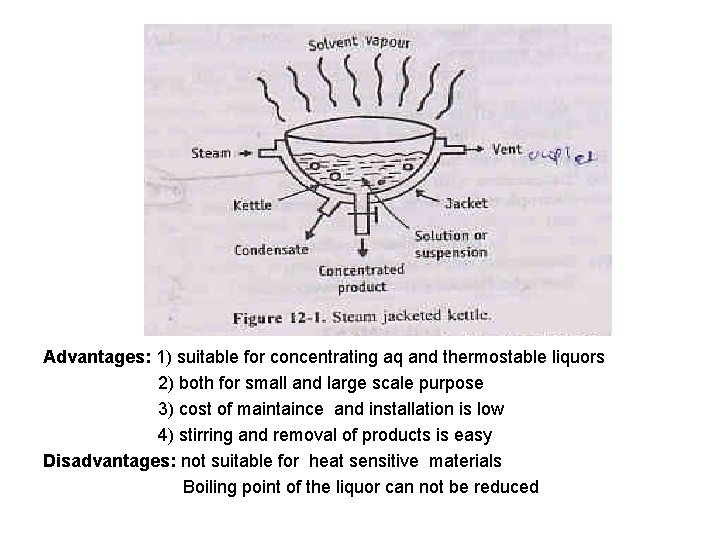
Advantages: 1) suitable for concentrating aq and thermostable liquors 2) both for small and large scale purpose 3) cost of maintaince and installation is low 4) stirring and removal of products is easy Disadvantages: not suitable for heat sensitive materials Boiling point of the liquor can not be reduced

Advantages: 1) suitable for non viscous liquors 2) both for small and large scale purpose 3) cost of maintaince and installation is low Disadvantages: Liquids should be maintained above the tubes

Advantages: 1) no of tubes increases surface area of liquor 2) both for small and large scale purpose 3) circulation enhances the rate of heat transfer Disadvantages: Liquids should be maintained above the calendria

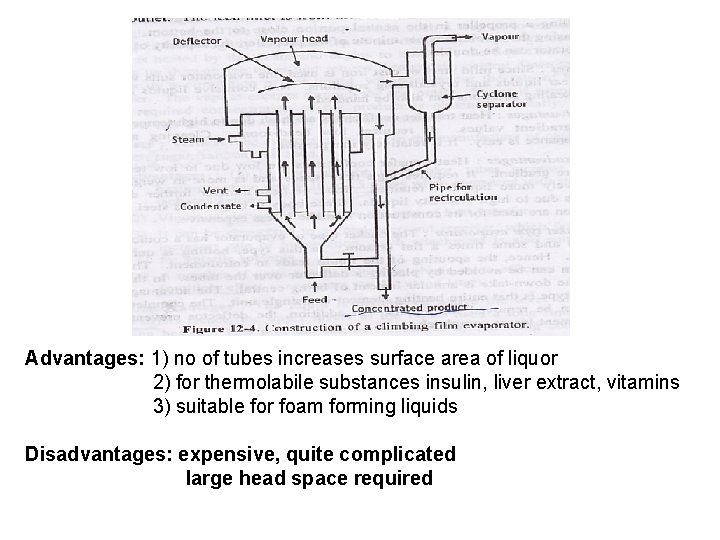
Advantages: 1) no of tubes increases surface area of liquor 2) for thermolabile substances insulin, liver extract, vitamins 3) suitable for foam forming liquids Disadvantages: expensive, quite complicated large head space required

Advantages: 1) no of tubes increases surface area of liquor 2) To separate volatile and non volatile liquids, also for viscous liquids 3) suitable for heat sensitive material, for extraction of yeasts, caffiene Disadvantages: expensive, cyclone separator is needed required


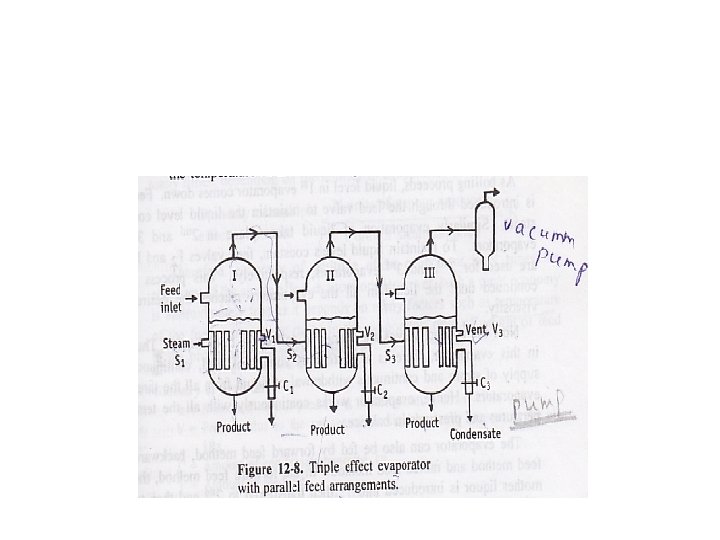
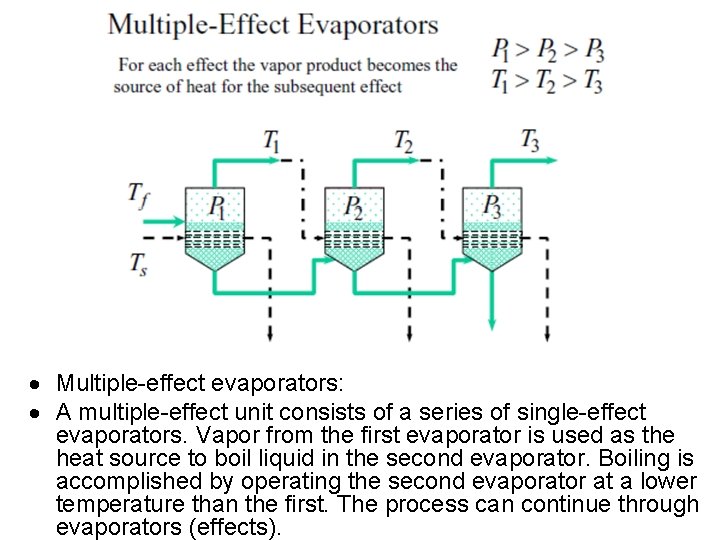
Multiple-effect evaporators: A multiple-effect unit consists of a series of single-effect evaporators. Vapor from the first evaporator is used as the heat source to boil liquid in the second evaporator. Boiling is accomplished by operating the second evaporator at a lower temperature than the first. The process can continue through evaporators (effects).
