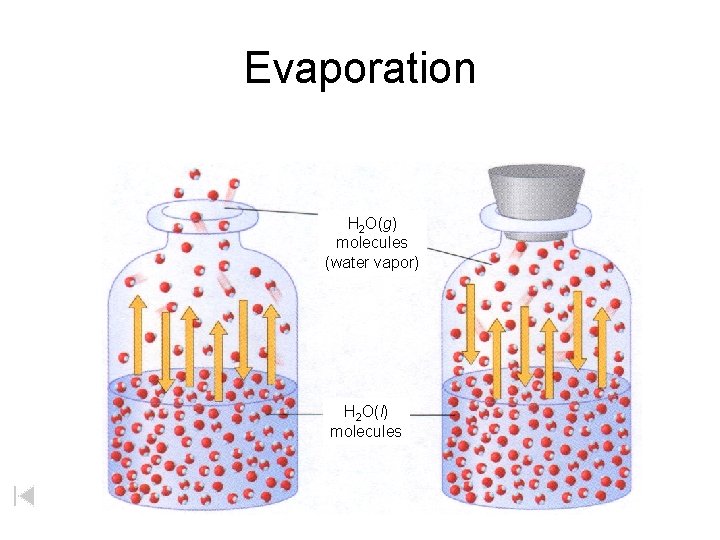
Evaporation H 2 Og molecules water vapor H









- Slides: 32

Evaporation H 2 O(g) molecules (water vapor) H 2 O(l) molecules

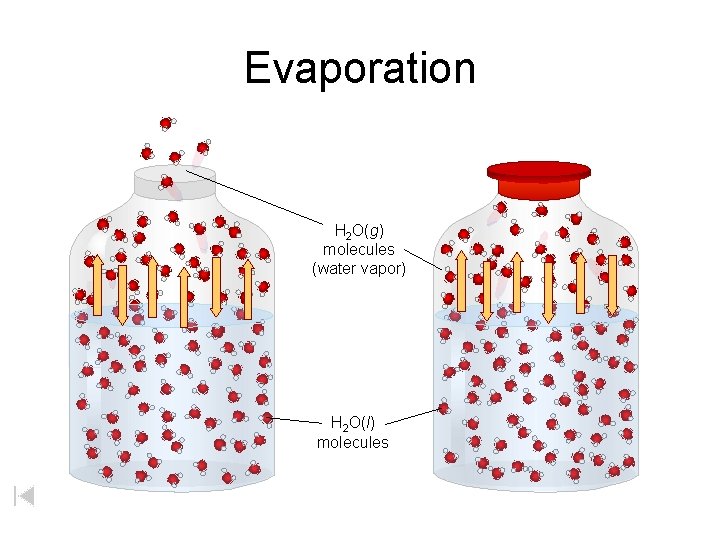
Evaporation H 2 O(g) molecules (water vapor) H 2 O(l) molecules

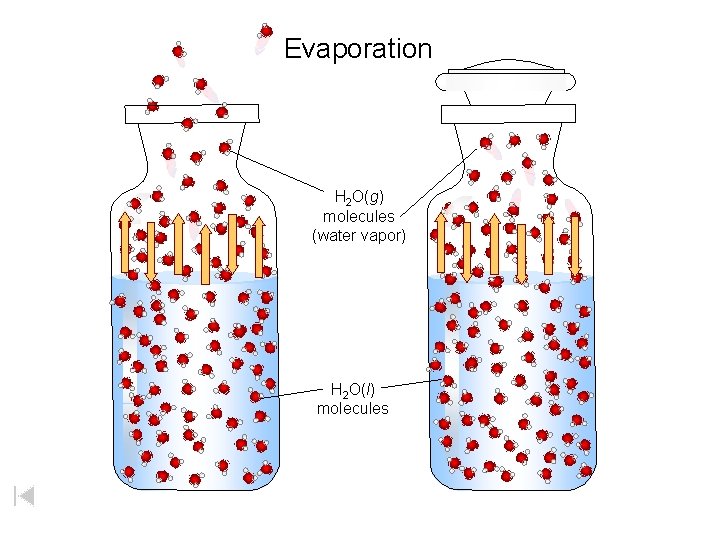
Evaporation H 2 O(g) molecules (water vapor) H 2 O(l) molecules

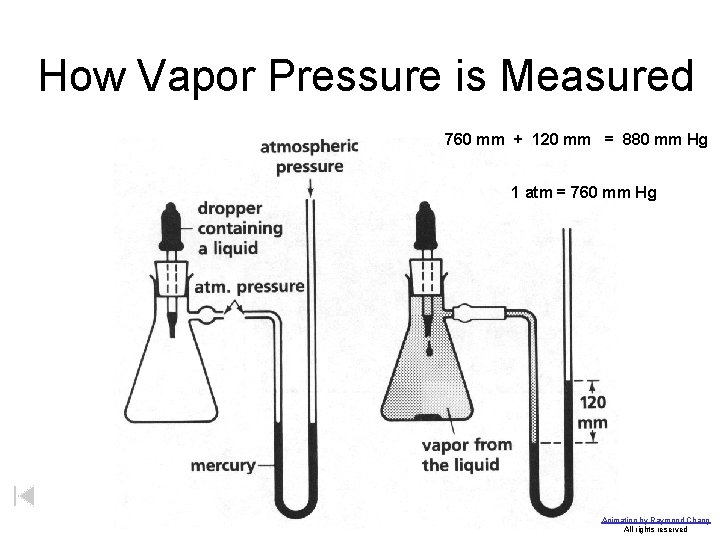
How Vapor Pressure is Measured 760 mm + 120 mm = 880 mm Hg 1 atm = 760 mm Hg Animation by Raymond Chang All rights reserved

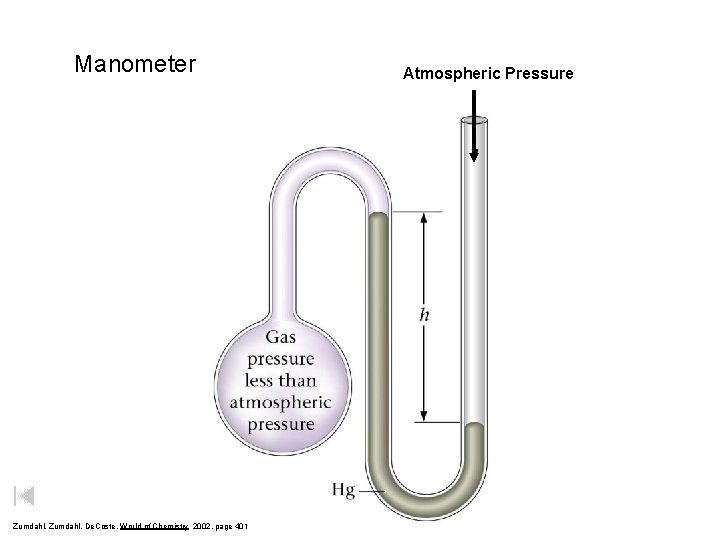
Manometer Zumdahl, De. Coste, World of Chemistry 2002, page 401 Atmospheric Pressure

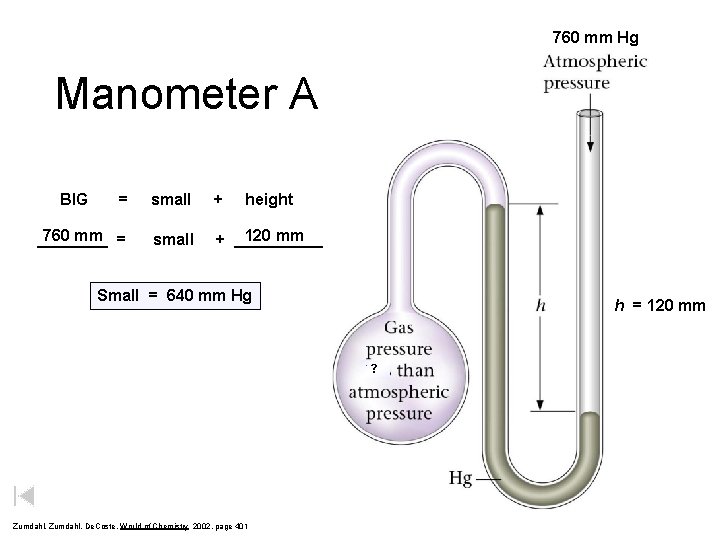
760 mm Hg Manometer A BIG = small + height 760 mm = ____ small 120 mm + _____ Small = 640 mm Hg h = 120 mm ? Zumdahl, De. Coste, World of Chemistry 2002, page 401

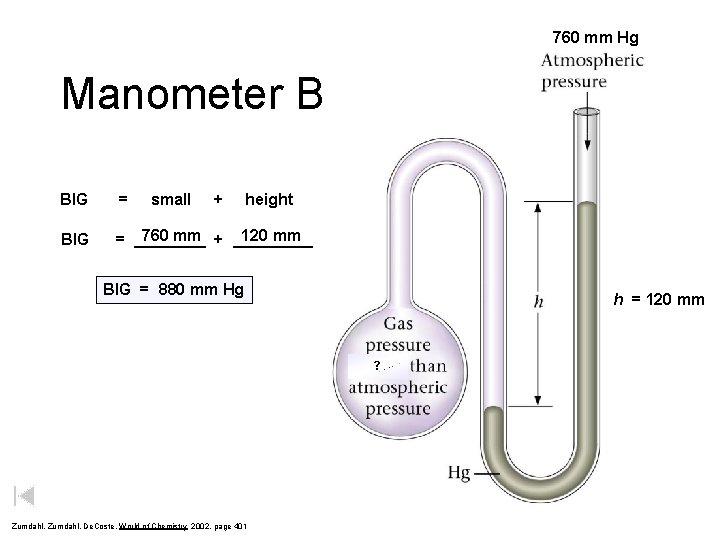
760 mm Hg Manometer B BIG = small + height BIG 760 mm + _____ 120 mm = ____ BIG = 880 mm Hg h = 120 mm ? Zumdahl, De. Coste, World of Chemistry 2002, page 401

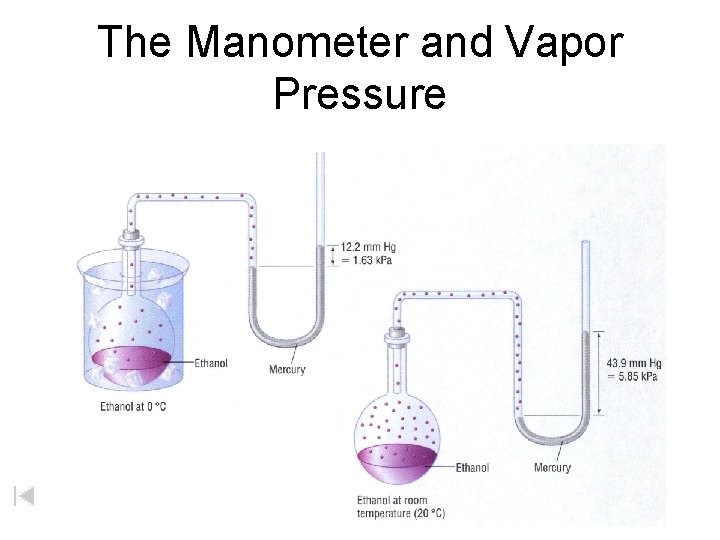
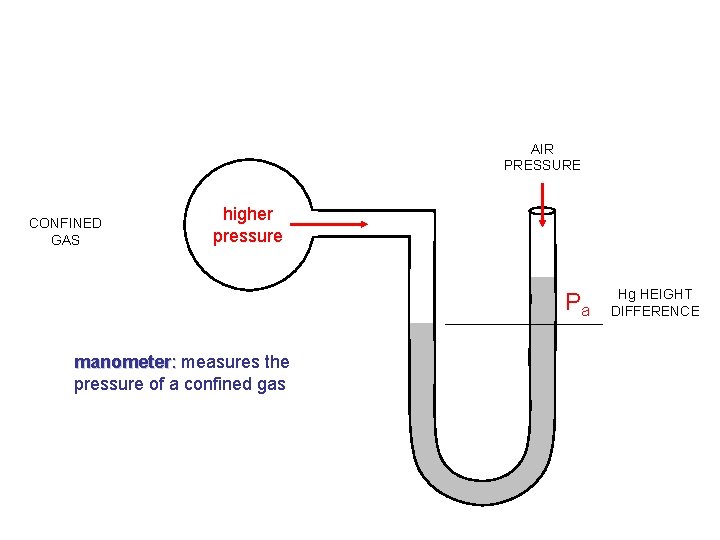
The Manometer and Vapor Pressure

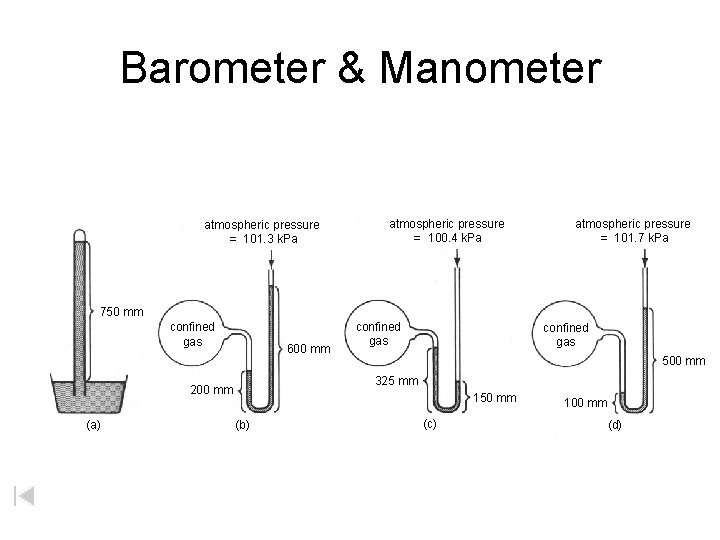
Barometer & Manometer atmospheric pressure = 101. 3 k. Pa atmospheric pressure = 100. 4 k. Pa atmospheric pressure = 101. 7 k. Pa 750 mm confined gas 600 mm confined gas 500 mm 325 mm 200 mm (a) confined gas 150 mm (b) (c) 100 mm (d)

Pressure and Temperature STP (Standard Temperature and Pressure) standard temperature 0 o. C 273 K standard pressure 1 atm 101. 3 k. Pa 760 mm Hg Equations / Conversion Factors: K = o. C + 273 o. C = K – 273 1 atm = 101. 3 k. Pa = 760 mm Hg

Convert 25 o. C to Kelvin. K = o. C + 273 25 o. C + 273 = 298 K How many k. Pa is 1. 37 atm? X k. Pa = 1. 37 atm 101. 3 k. Pa = 138. 8 k. Pa 1 atm How many mm Hg is 231. 5 k. Pa? X mm Hg = 231. 5 k. Pa 760 mm Hg = 1737 mm Hg 101. 3 k. Pa

AIR PRESSURE CONFINED GAS higher pressure Pa manometer: measures the pressure of a confined gas Hg HEIGHT DIFFERENCE

small 96. 5 k. Pa Atmospheric pressure is 96. 5 k. Pa; mercury height difference is 233 mm. Find confined gas pressure, in atm. BIG 1. 26 X atm SMALL + HEIGHT = BIG 233 mm Hg 96. 5 k. Pa + 233 mm Hg = X atm 96. 5 k. Pa 1 atm 101. 3 k. Pa + 233 mm Hg 0. 953 atm + 0. 307 atm = X atm X = 1. 26 atm 1 atm = X atm 760 mm Hg

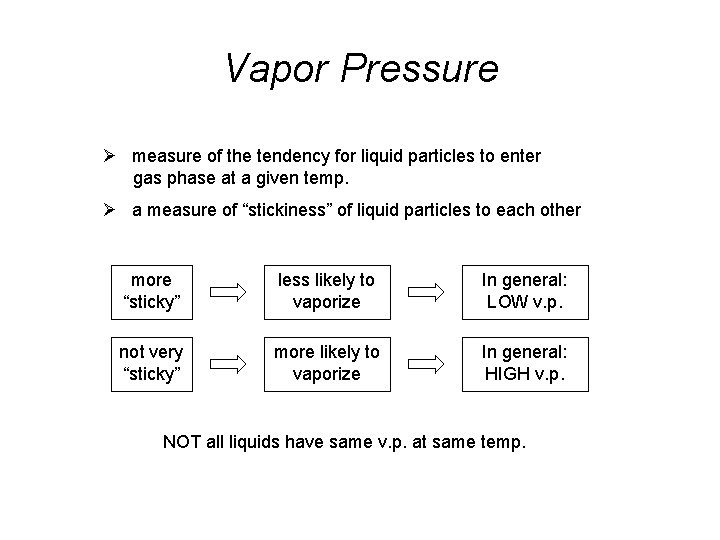
Vapor Pressure Ø measure of the tendency for liquid particles to enter gas phase at a given temp. Ø a measure of “stickiness” of liquid particles to each other more “sticky” less likely to vaporize In general: LOW v. p. not very “sticky” more likely to vaporize In general: HIGH v. p. NOT all liquids have same v. p. at same temp.

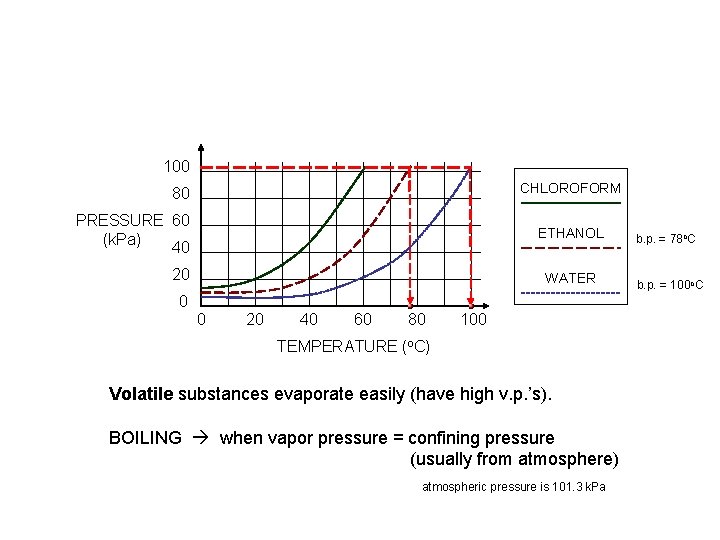
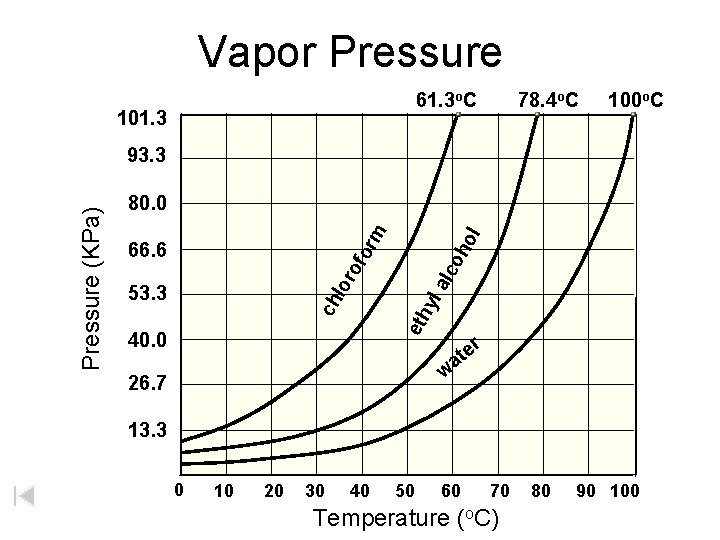
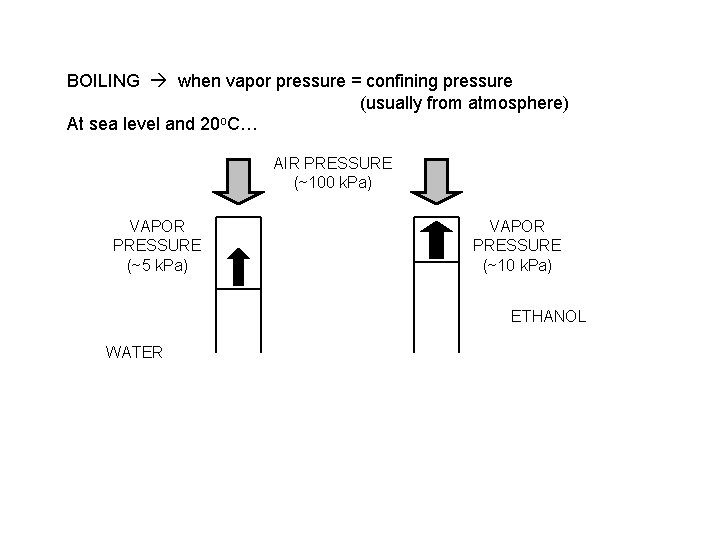
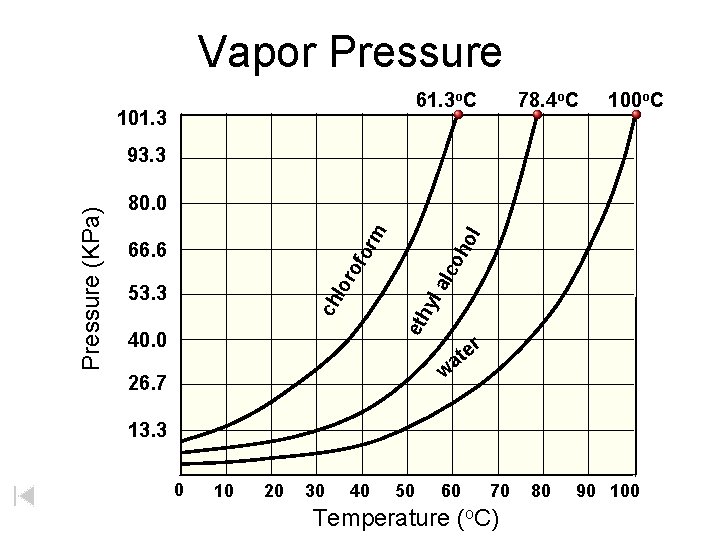
100 CHLOROFORM 80 PRESSURE 60 (k. Pa) 40 ETHANOL 20 WATER 0 0 20 40 60 80 100 TEMPERATURE (o. C) Volatile substances evaporate easily (have high v. p. ’s). BOILING when vapor pressure = confining pressure (usually from atmosphere) atmospheric pressure is 101. 3 k. Pa b. p. = 78 o. C b. p. = 100 o. C

Vapor Pressure 61. 3 o. C 101. 3 78. 4 o. C 100 o. C 53. 3 eth yl lor alc of 66. 6 oh ol or m 80. 0 ch Pressure (KPa) 93. 3 40. 0 e at r w 26. 7 13. 3 0 10 20 30 40 50 60 70 Temperature (o. C) 80 90 100

BOILING when vapor pressure = confining pressure (usually from atmosphere) At sea level and 20 o. C… AIR PRESSURE (~100 k. Pa) VAPOR PRESSURE (~5 k. Pa) VAPOR PRESSURE (~10 k. Pa) ETHANOL WATER

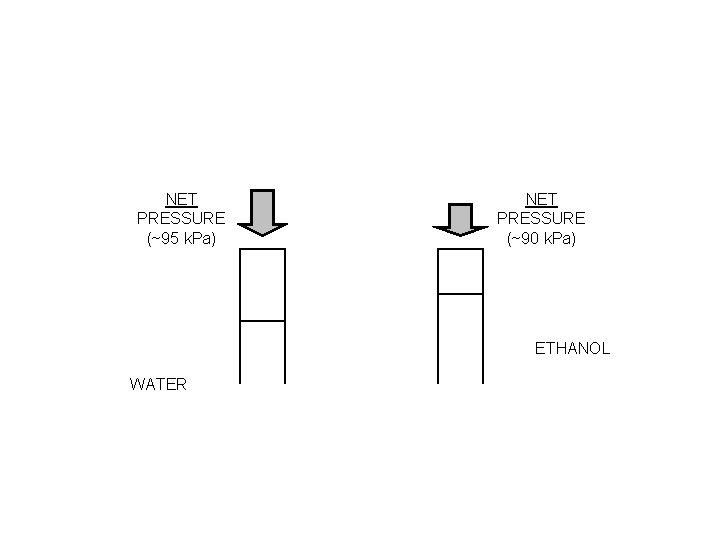
NET PRESSURE (~95 k. Pa) NET PRESSURE (~90 k. Pa) ETHANOL WATER

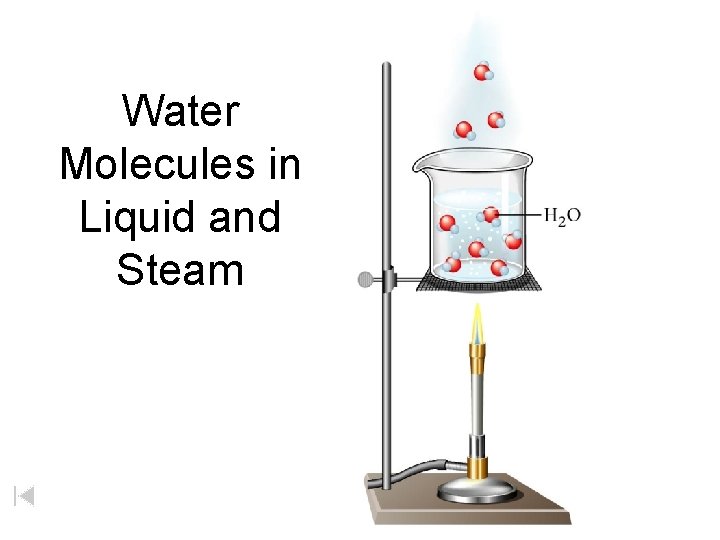
Water Molecules in Liquid and Steam

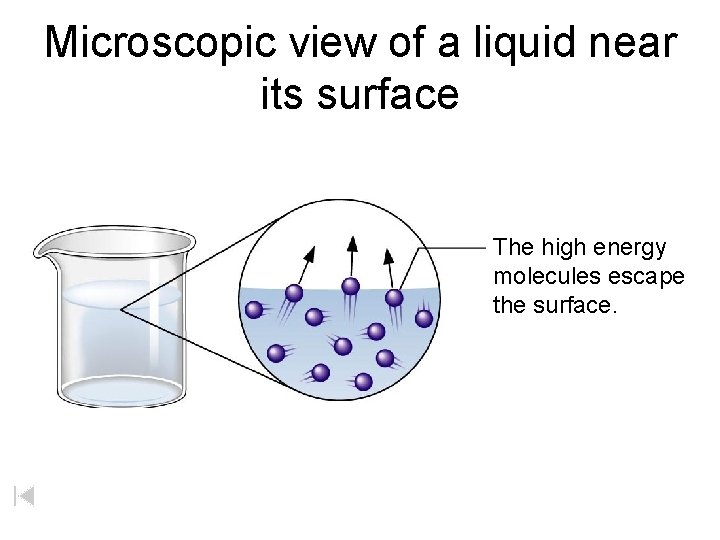
Microscopic view of a liquid near its surface The high energy molecules escape the surface.

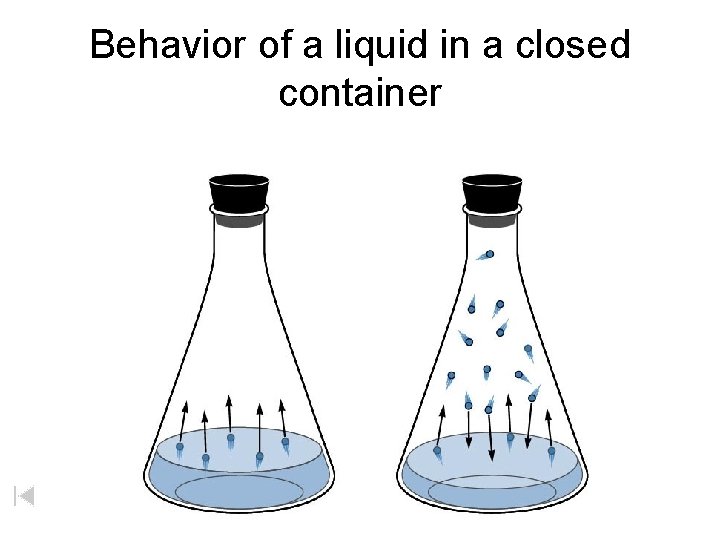
Behavior of a liquid in a closed container

Water rapidly boiling on a stove

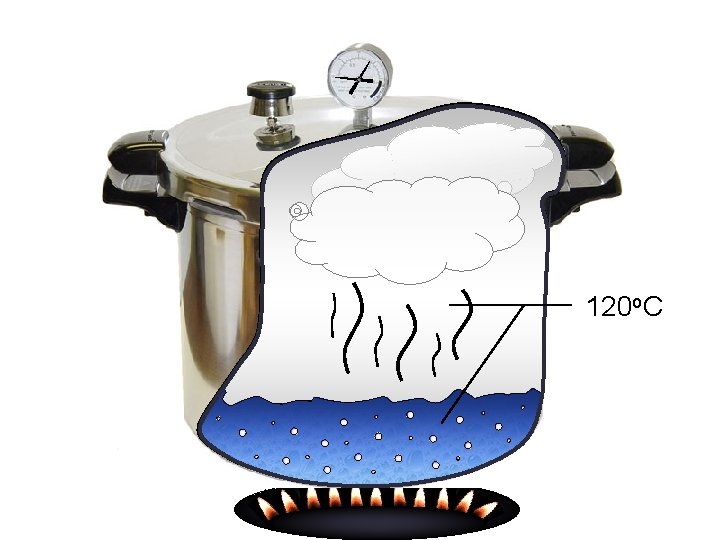
Pressure Cooker Copyright © 2006 Pearson Benjamin Cummings. All rights reserved.

120 o. C

Formation of a bubble is opposed by the pressure of the atmosphere Zumdahl, De. Coste, World of Chemistry 2002, page 452

Vapor Pressure 61. 3 o. C 101. 3 78. 4 o. C 100 o. C 53. 3 eth yl lor alc of 66. 6 oh ol or m 80. 0 ch Pressure (KPa) 93. 3 40. 0 e at r w 26. 7 13. 3 0 10 20 30 40 50 60 70 Temperature (o. C) 80 90 100

Boiling Point and Pressure

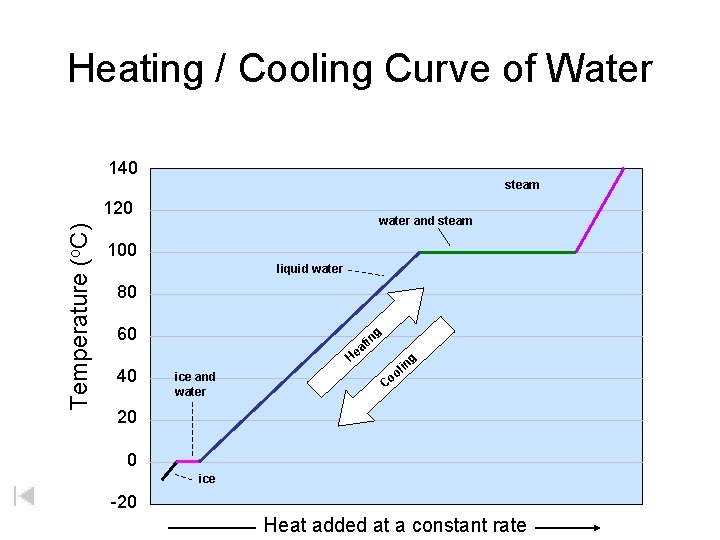
Heating / Cooling Curve of Water 140 steam Temperature (o. C) 120 water and steam 100 liquid water 80 60 tin a e g H 40 ice and water in ol g Co 20 0 ice -20 Heat added at a constant rate

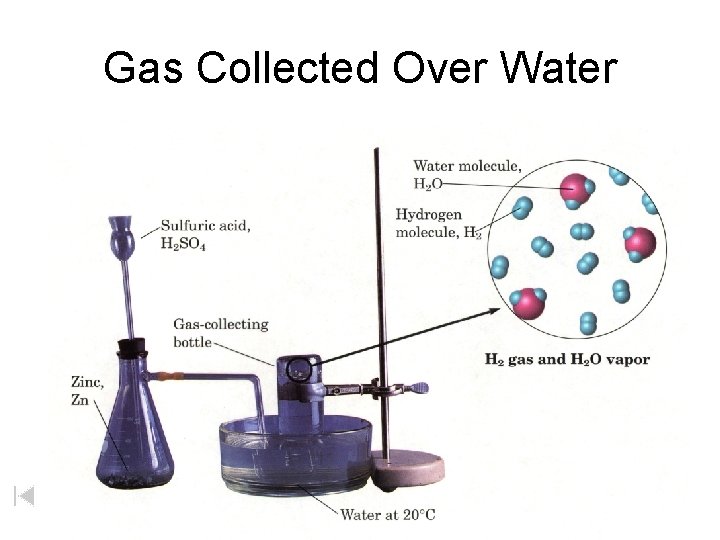
Gas Collected Over Water

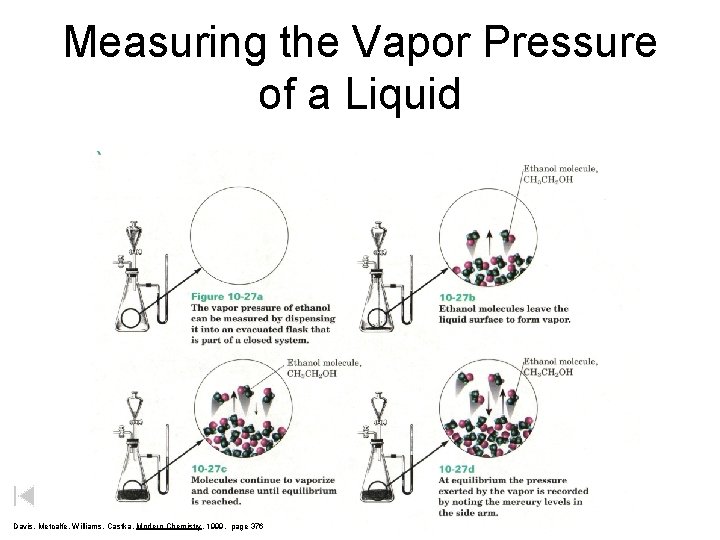
Measuring the Vapor Pressure of a Liquid Davis, Metcalfe, Williams, Castka, Modern Chemistry, 1999, page 376

Gas Mixtures and Dalton’s Law

Gases Dissolved in Liquids