Evaporation Introduction to Food Engineering Evaporation v Removal
















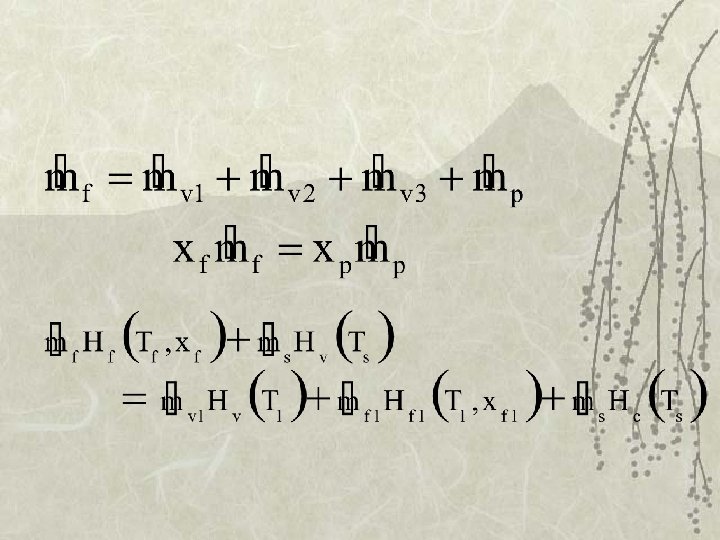
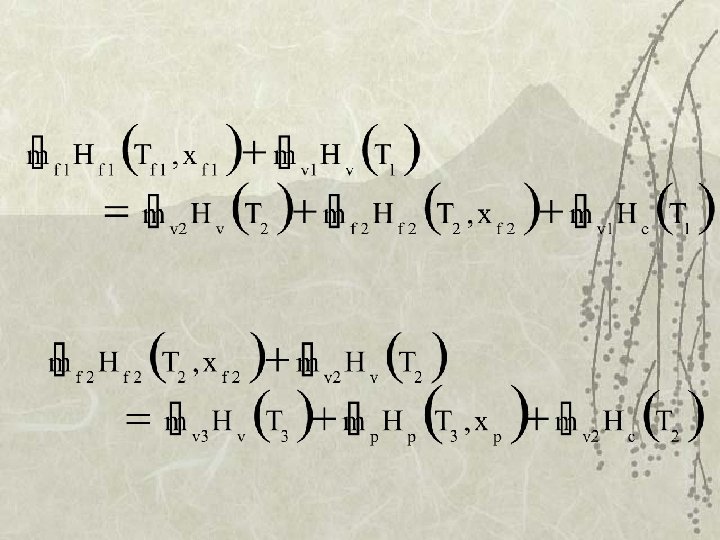
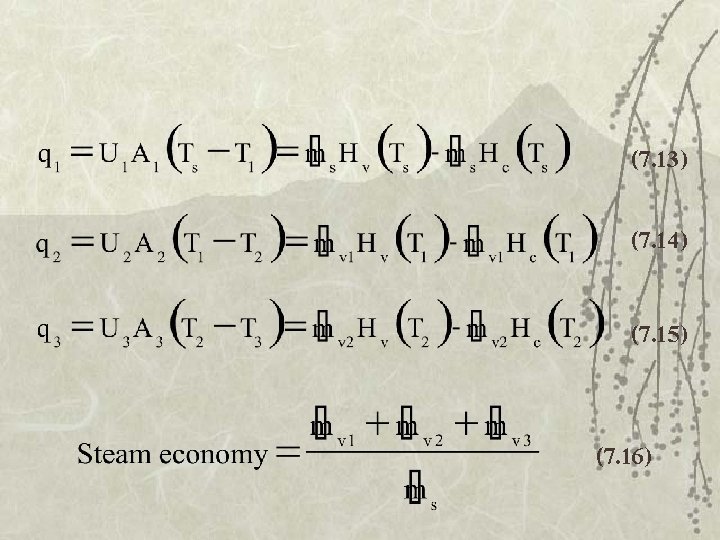
- Slides: 31

Evaporation Introduction to Food Engineering

Evaporation v Removal of water from diluted liquid foods to obtain concentrated products. – Microbiological stability – Reduce transportation costs, storage v Evaporator – Heat exchanger in large chamber – Product under vacuum

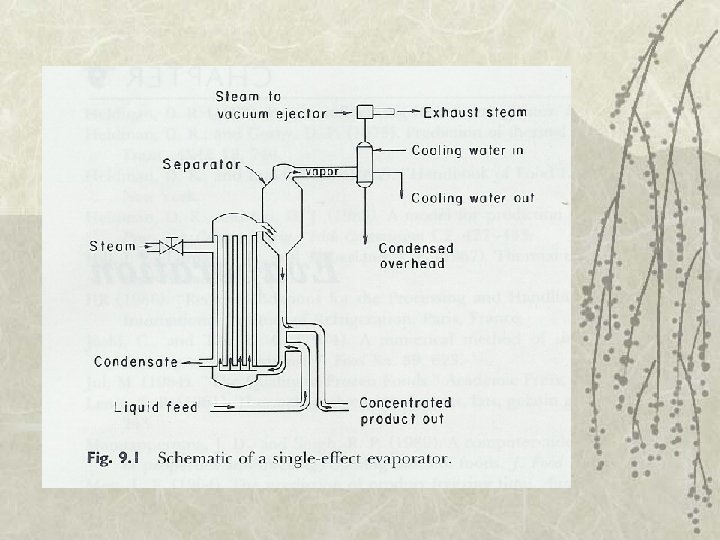
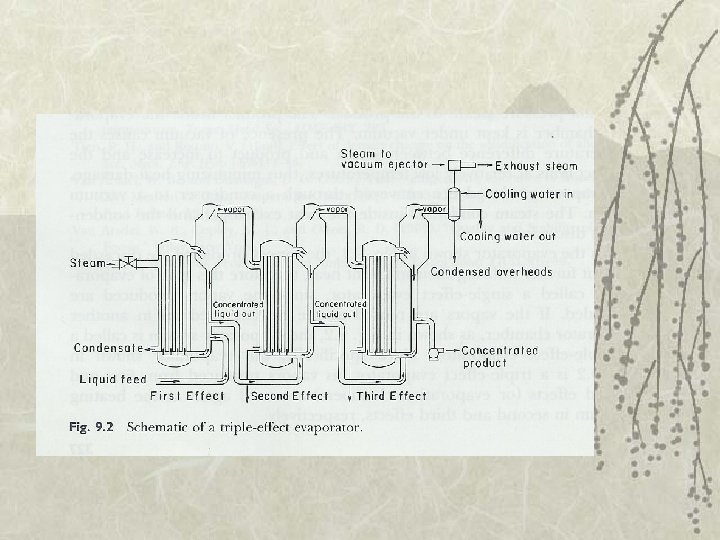
Evaporator v Single-effect evaporator – Vapor discarded v Multiple-effect evaporator – Vapor reused as heating medium



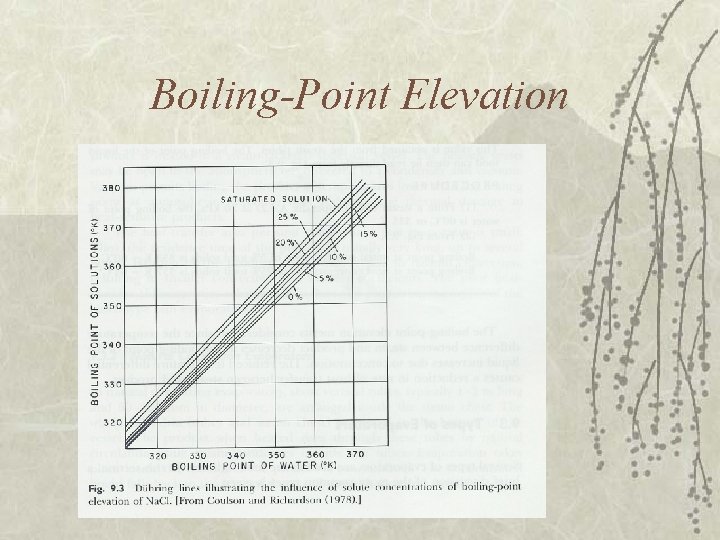
Boiling-Point Elevation

Duhring’s Rule v Linear relationship between boilingpoint temperature of solution and boiling point temperature of water at the same pressure.

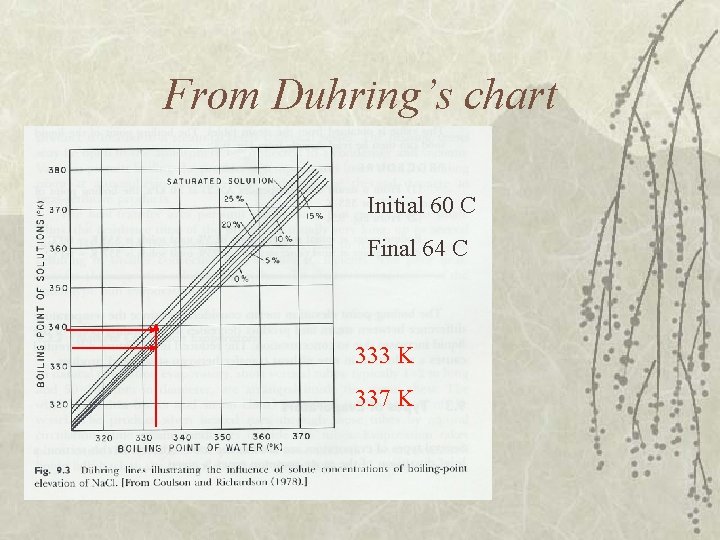
Example v Determine initial and final boiling point of a liquid food. The pressure in evaporator is 20 k. Pa. The product is being concentrated from 5 % to 25 % solids concentration. – Boiling pt. of water from steam table at 20 k. Pa = 60 C (333 K)

From Duhring’s chart Initial 60 C Final 64 C 333 K 337 K

Types of Evaporator v . 1 Batch-type pan evaporator

v Heat v transfer per unit volume is small <=long residence time, limit capacities

Types of Evaporator v 2. Natural circulation evaporators – 1 – 2 m vertical tubes inside steam chest

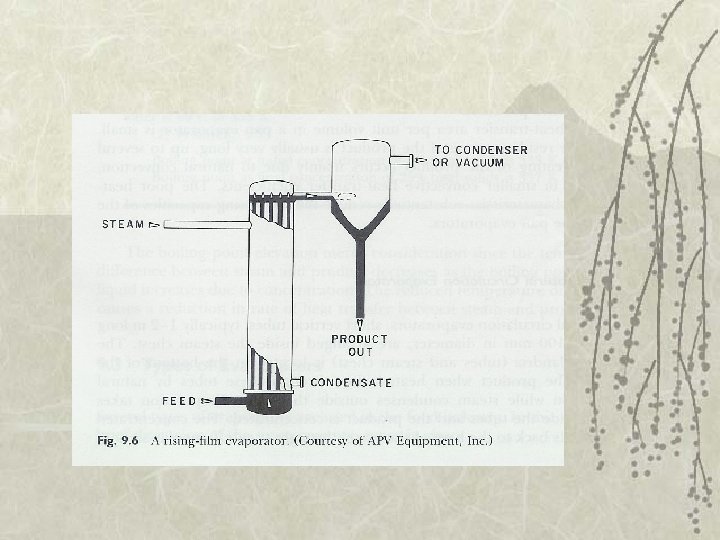
Types of Evaporator v 3. Rising-film evaporator – 10 – 15 m vertical tubes – Film of liquid move upward – Need 14 C difference between heating medium and product


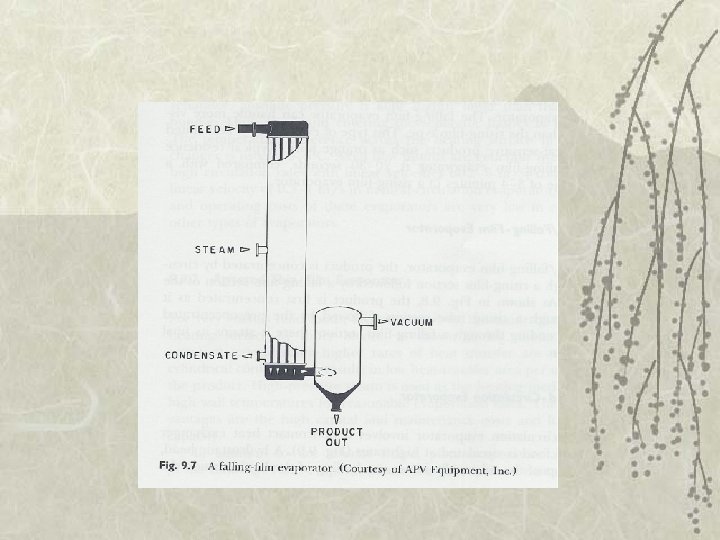
Types of Evaporator v Falling-film evaporator – Thin liquid film move downward – Distribution of liquid in uniform film by spray nozzles – Handle more viscous liquids than risingfilm – Less residence time


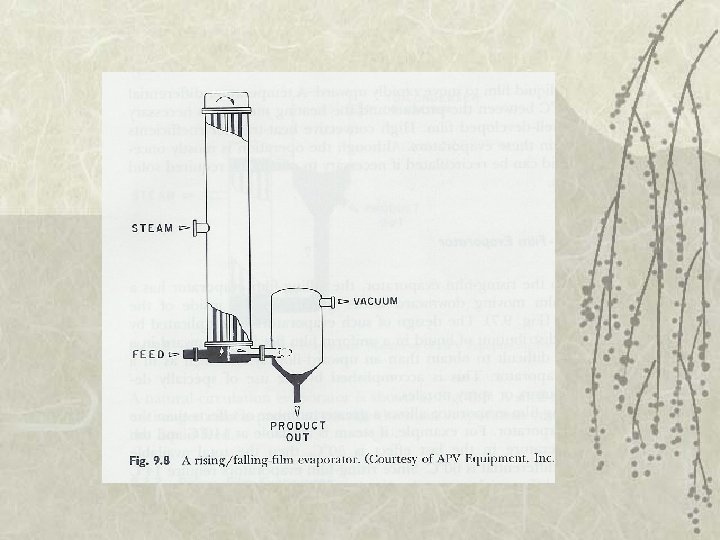
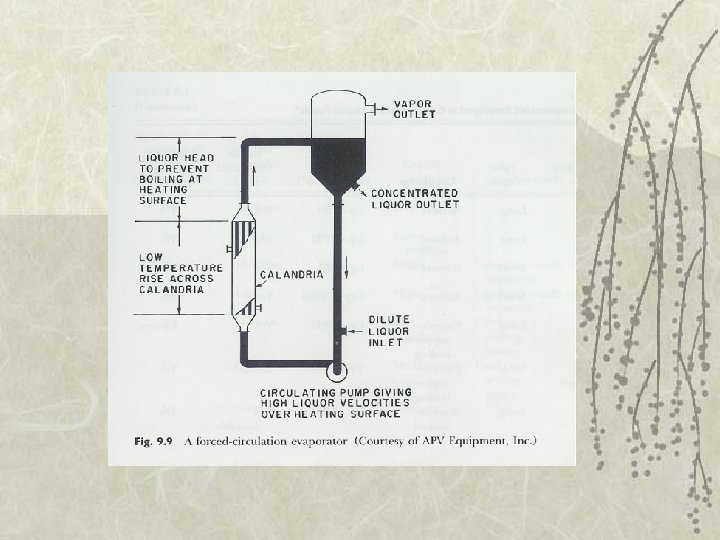
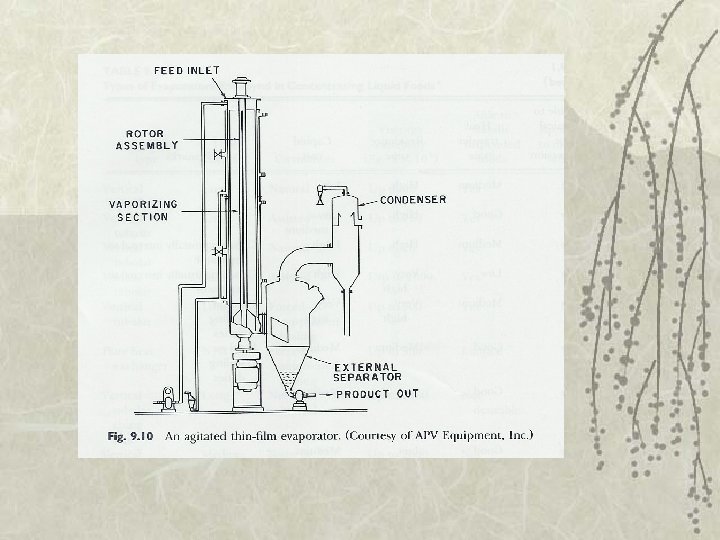
Types of Evaporator v 5. Rising/falling-film evaporator v 6. Forced-circulation evaporator – Use pump to maintain high circulation rates v 7. Agitated thin-film evaporator – Very viscous fluid foods – Feed is spread on heating surface by wiper blades




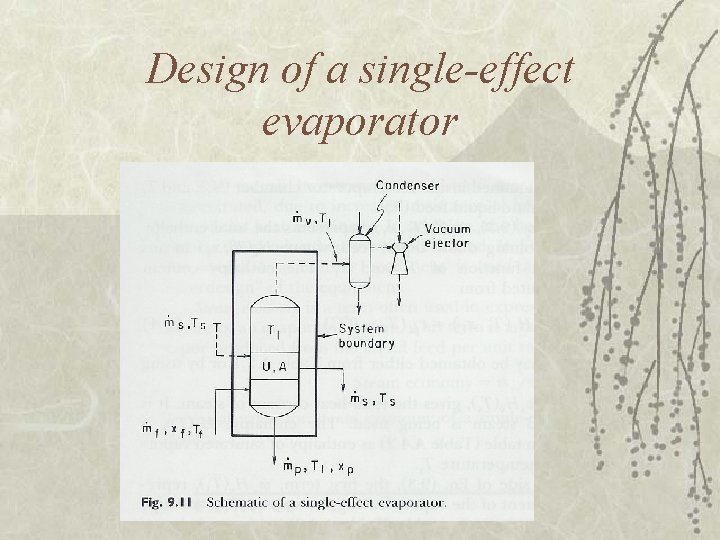
Design of a single-effect evaporator

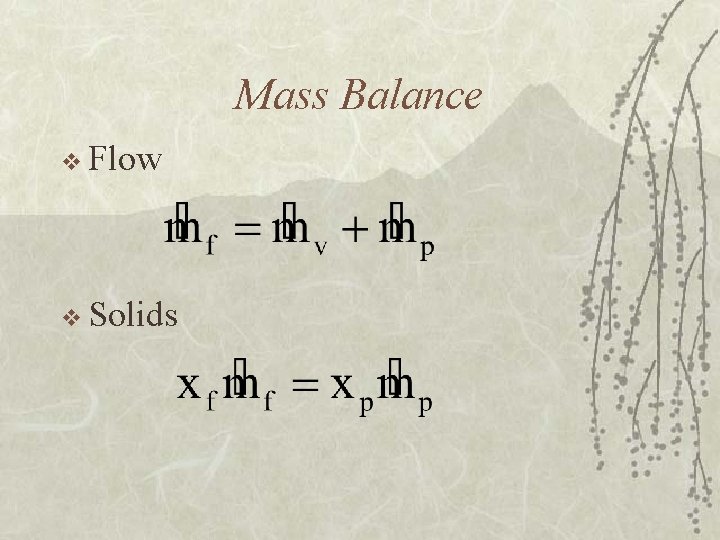
Mass Balance v Flow v Solids

Enthalpy Balance v k. J/kg feed steam vapor product Hv (Ts), Hv (T 1) , Hc (Ts) from steam table condensate

Rate of heat transfer q = rate of heat transfer (W) U = overall heat transfer coefficient (W/m 2 K) A = area, m 2

Rate of heat transfer v. U decreases as product becomes concentrated. – increase resistance of heat transfer – Boiling point elevation v But constant U is used -> overdesign

Steam Economy v Ratio-rate of mass of water vapor produced per unit of steam consumed v Typically -> 1

Example v Apple juice is being concentrated. At steady state, feed = 0. 67 kg/s. Concentration of the juice = 11 % total solids. The juice is concentrated to 75 % TS. Specific heats of diluted feed and concentrate are 3. 9 and 2. 3 k. J/kg. C. The steam pressure is measured to be 304. 42 k. Pa. Inlet feed temp is 43. 3 C. The product inside the evaporator boils at 62. 2 C. Assume U = 943 W/m 2 C, negligible boilingpoint elevation. Calculate mass flow rate of concentrate product, steam requirements, steam economy and heat-transfer area.

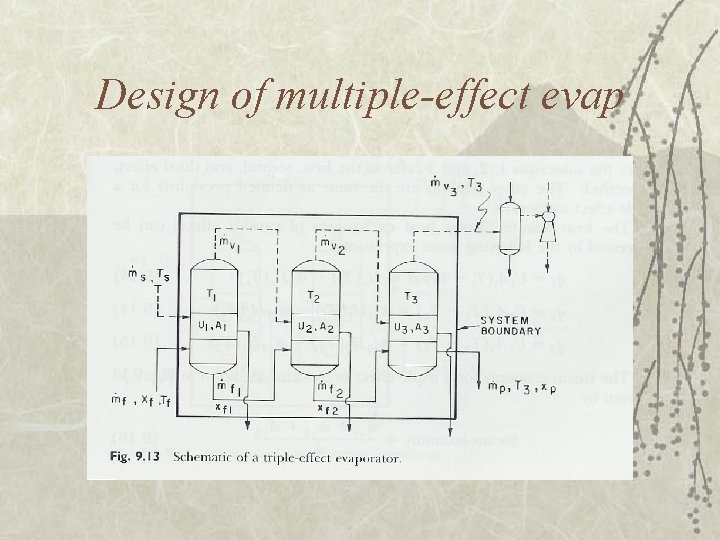
Design of multiple-effect evap
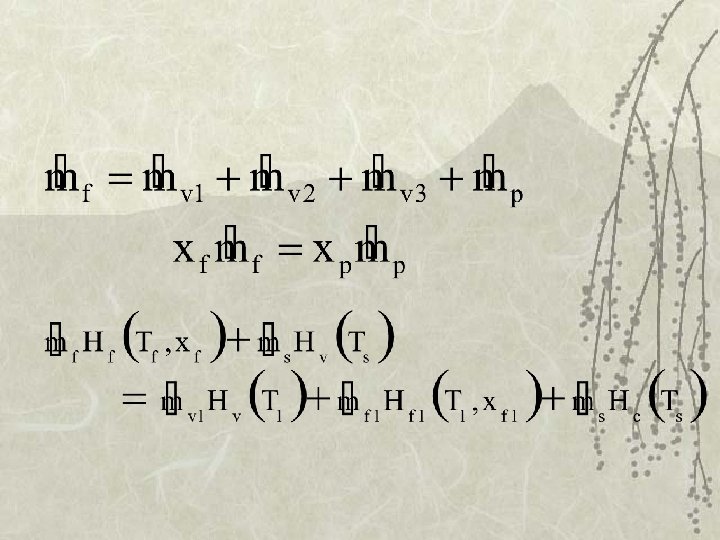
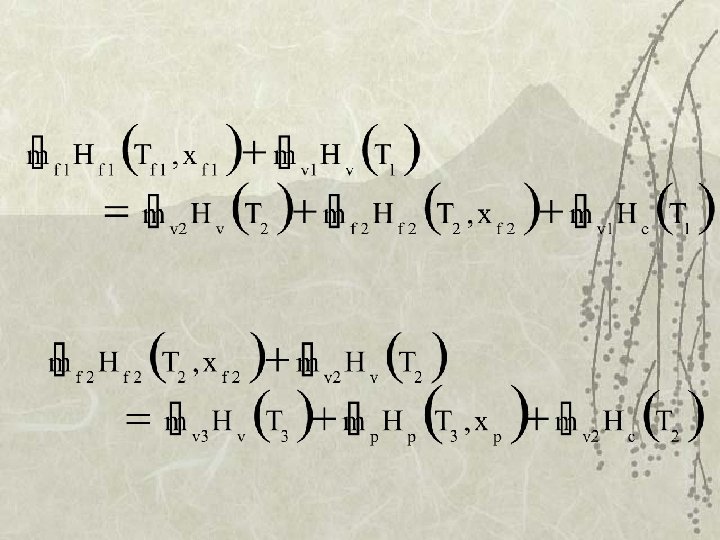
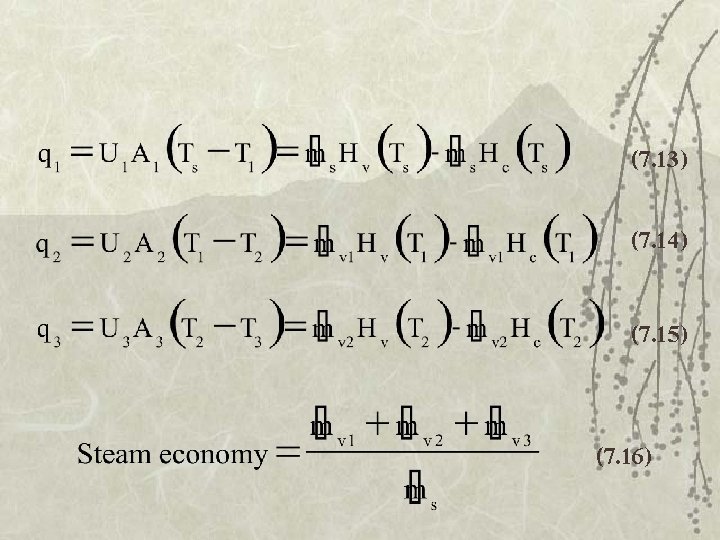
(7. 13) (7. 14) (7. 15) (7. 16)