Evaporation vs Boiling Evaporation Liquid changing into gas

Evaporation vs. Boiling

Evaporation �Liquid changing into gas. �Seems similar to boiling but there are differences.
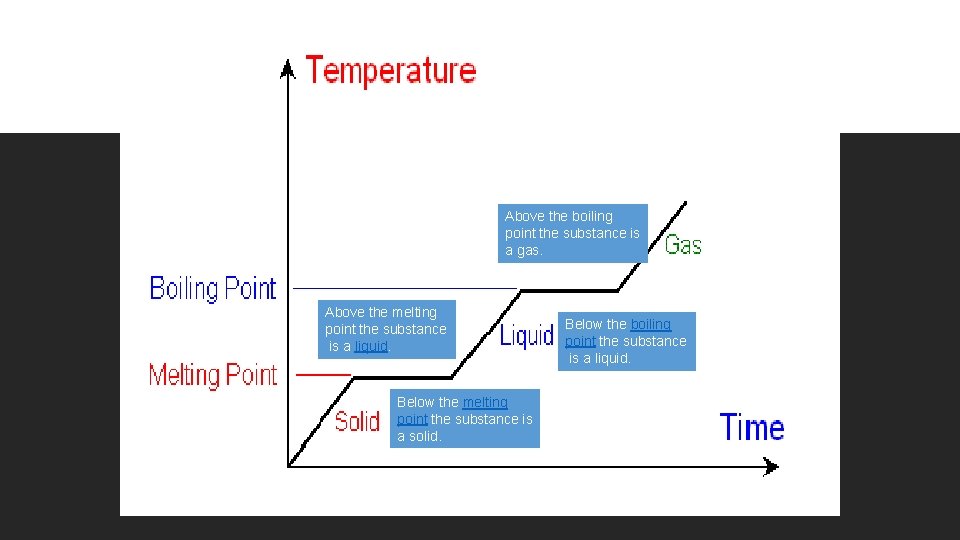
Above the boiling point the substance is a gas. Above the melting point the substance is a liquid. Below the melting point the substance is a solid. Below the boiling point the substance is a liquid.

Evaporation VS Boiling Evaporation occurs on the surface of liquid and it is a vaporization of liquid. It is a state of transition from liquid to gaseous state. The process occurs slowly and cannot be seen as well. It occurs when there is exposure of water to air and water molecules change into vapor and these vapors rise up and form clouds. Boiling occurs on the entire mass of liquid and it is the vaporization of liquid. It occurs rapidly. Boiling occurs when the temperature of the liquid is greater than the boiling point of the substance.
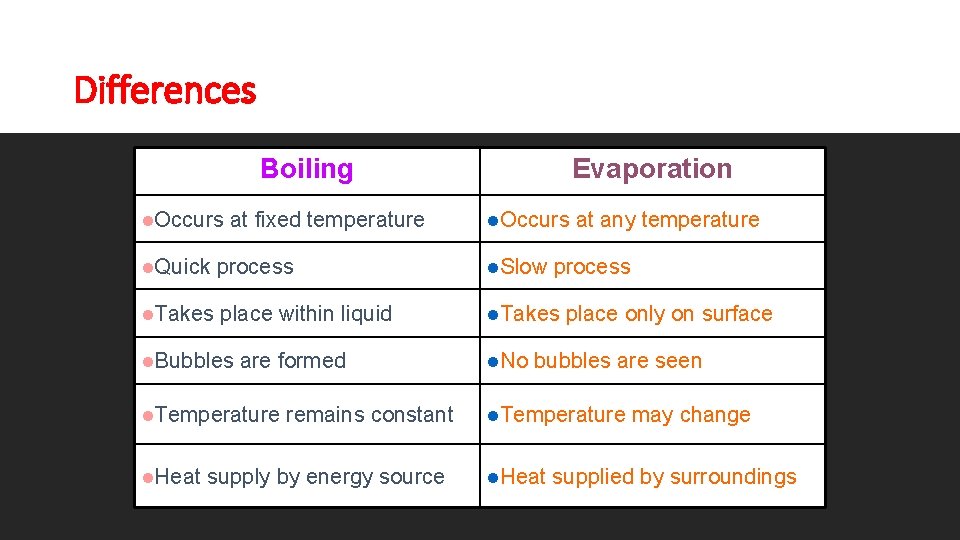
Differences Boiling l. Occurs at fixed temperature Evaporation l. Occurs l. Quick process l. Slow l. Takes place within liquid l. Takes l. Bubbles are formed l. Temperature l. Heat remains constant supply by energy source l. No at any temperature process place only on surface bubbles are seen l. Temperature l. Heat may change supplied by surroundings

Factors Affecting rate of Evaporation • Temperature of liquid and surroundings. • Surface Area • Presence of wind

Planning a Practical! Next week you will be carrying out a practical. Before completing any practical you must first prepare the first part of the lab report…

Make a prediction! • Write the title “Investigating the Heating Curve of Water” in your book…this is the beginning of your first lab report! • Make a sub-heading “Prediction” • Under this you need to make your own prediction – what do you think will happen in the experiment? How do you think the temperature will change as time goes on?

Equipment list • Make a new sub-heading “Equipment list” • Make a list using bullet-points of the equipment we will use: • Ice/Water Mixture This is what we will measure the temperature of. • Hot Plate To heat the water. • Thermometer To measure the temperature. • Stopwatch To keep the intervals between measurements the same. • 250 m. L beaker To hold the water.

Labelled diagram • Make a sub-heading “Labelled Diagram” • Draw a labelled diagram of how the experiment will be set up. Use pencil!!!

Procedure • Make a sub-heading “Procedure” 1. 2. 3. 4. 5. Measure the initial temperature of the ice-water in your beaker and start the timer. Measure the temperature after one minute. Repeat step 2 every minute until the water has boiled away. Combine results with one other group and calculate averages. Plot a heating curve on graph paper.
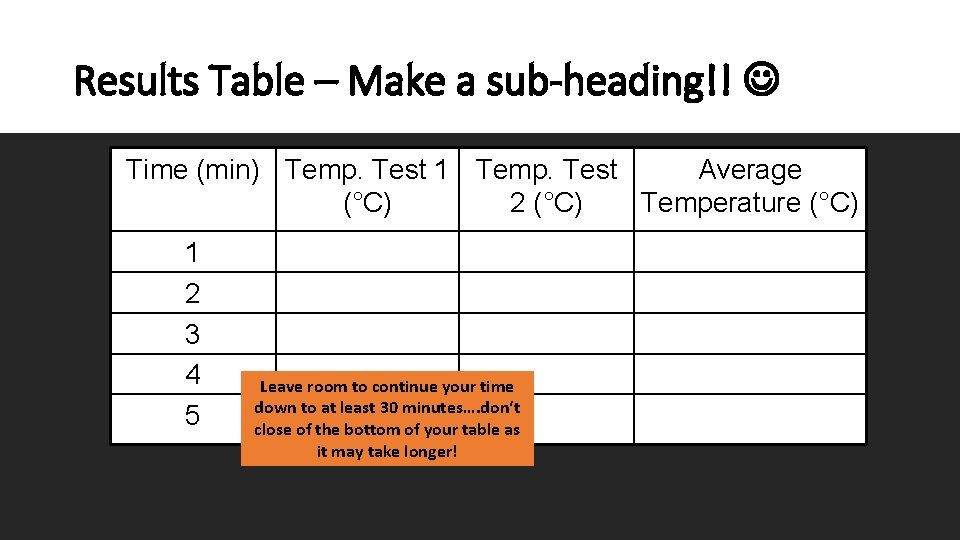
Results Table – Make a sub-heading!! Time (min) Temp. Test 1 Temp. Test Average (°C) 2 (°C) Temperature (°C) 1 2 3 4 5 Leave room to continue your time down to at least 30 minutes…. don‘t close of the bottom of your table as it may take longer!
- Slides: 12