Phases of Matter Kinetic Theory All matter is













- Slides: 23

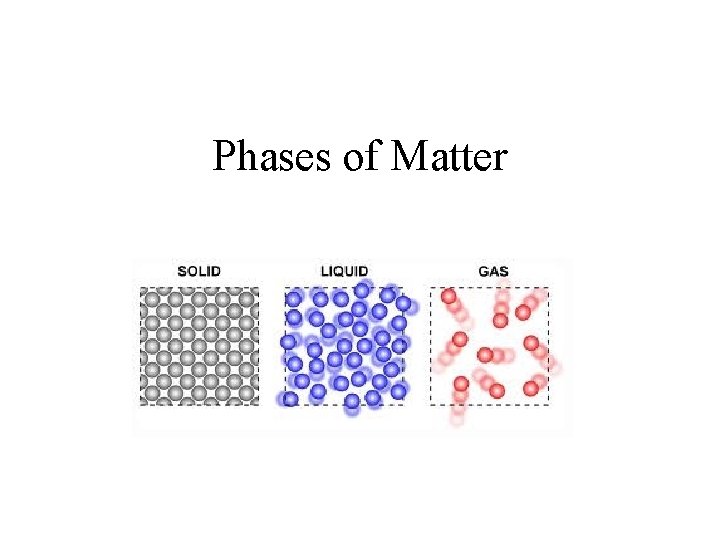
Phases of Matter

Kinetic Theory • All matter is made of atoms and molecules that act like tiny particles. • These tiny particles are always in motion. The higher the temp. , the faster the particles move. • At the same temp. , more massive (heavier) particles move slower than less massive (lighter) particles.

SOLIDS • Definite shape? • YES • Definite volume? • YES • Molecules in a solid are tightly packed and constantly vibrating.

LIQUIDS • Definite shape? • NO • Definite volume? • YES • Some liquids flow more easily than others. The resistance of a liquid to flow is called viscosity. – Honey has a high viscosity compared to water.

GASES • Definite shape? • NO • Definite volume? • NO • The particles in a gas are spread very far apart, but can be compressed by pumping them into a restricted volume.

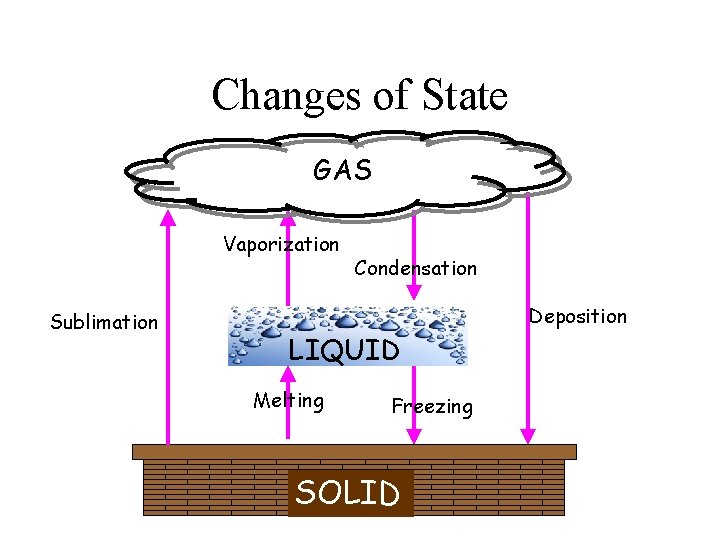
Phase Changes • Changes in phase are examples of physical changes. • • • Melting: solid liquid Freezing: liquid solid Vaporization: liquid gas Condensation: gas liquid Sublimation: solid gas

Changes of State GAS Vaporization Sublimation Condensation Deposition Melting LIQUID Melting Freezing SOLID

• ENERGY is the ability to change or move matter. • Energy is ABSORBED when substances melt or evaporate. – NOTE: our bodies cool down when our sweat evaporates. • Energy is RELEASED when substances freeze or condense.

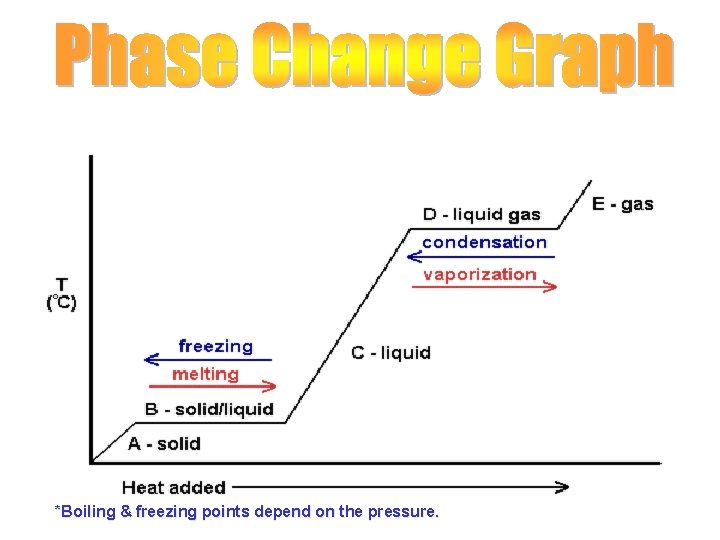
Melting • The change of state from solid to liquid. • Energy (heat) is absorbed by the substance that is melting.

Freezing • The change of state from liquid to solid. Opposite of melting. Energy (heat) is released by the substance undergoing freezing.

• Evaporation The change of state at the surface of a liquid as it passes to a vapor. This results from the random motion of molecules that occasionally escape from the liquid surface. – Energy (heat) is absorbed by the liquid (Cooling of the liquid results) – Can happen at any temperature

Condensation • The change of state from gas to liquid. The opposite of evaporation. – Energy (heat) is released by the liquid (Warming of the liquid results)

Boiling • Change from state from a liquid to a gas. • Occurs throughout the liquid. – boiling point/temperature is determined by pressure – Energy (heat) is absorbed by the liquid.

*Boiling & freezing points depend on the pressure.

Water at normal pressure (1 atm): • For water at normal (every day) pressures: • Melting/freezing point: 0 o. C (32 o. F) • Condensing/boiling point: 100 o. C (212 o. F) Label the points & temperatures on your graph.

Change the pressure Change the Boiling Point

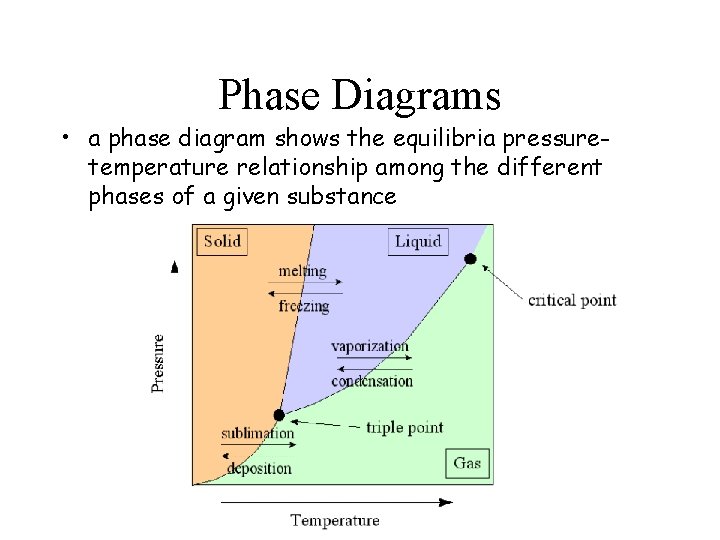
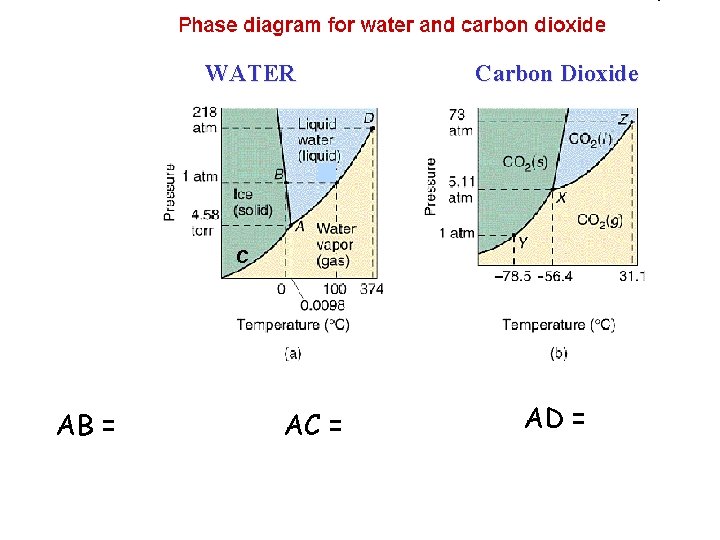
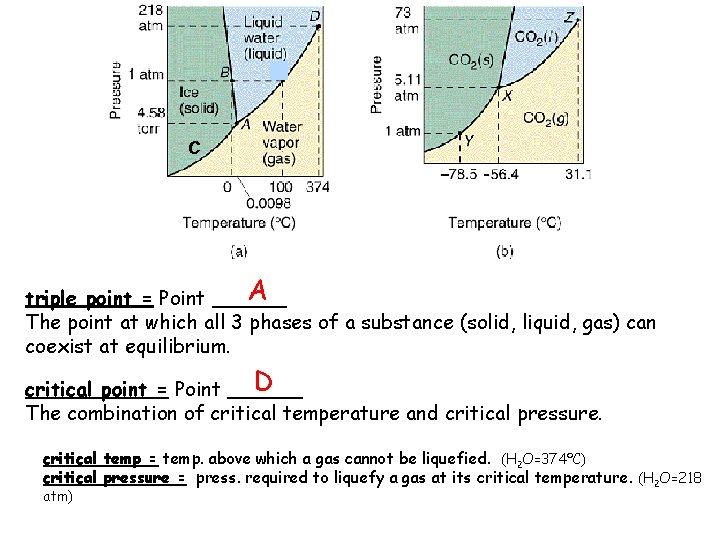
Phase Diagrams • a phase diagram shows the equilibria pressuretemperature relationship among the different phases of a given substance

WATER Carbon Dioxide C AB = AC = AD =

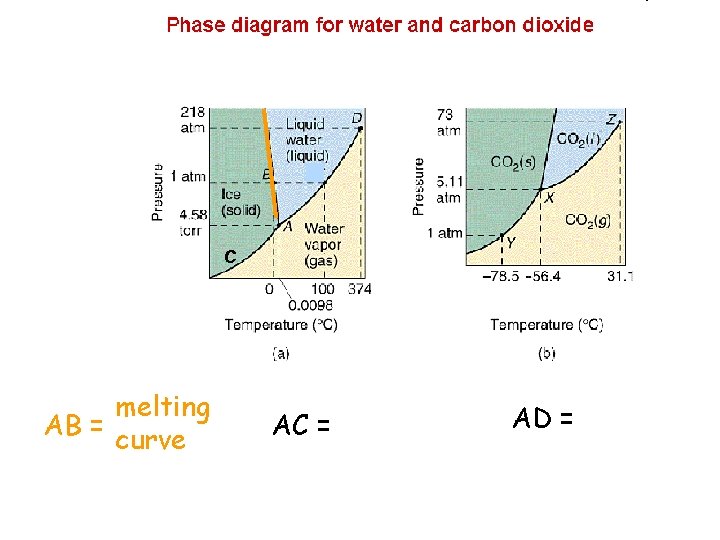
C melting AB = curve AC = AD =

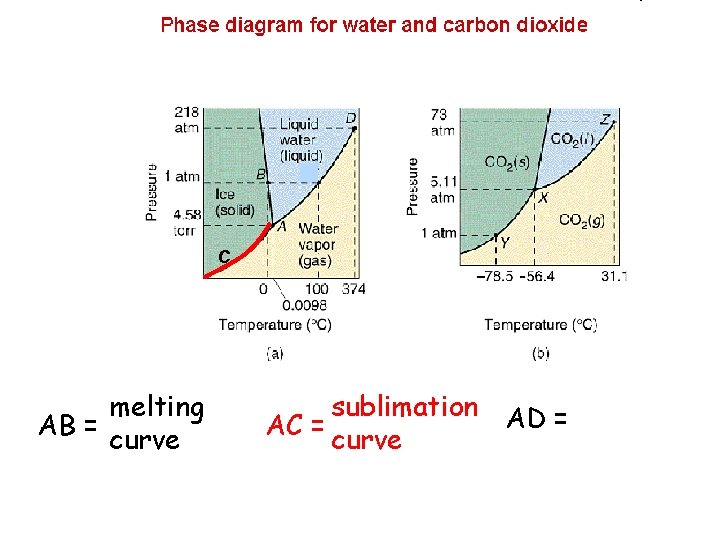
C melting AB = curve sublimation AD = AC = curve

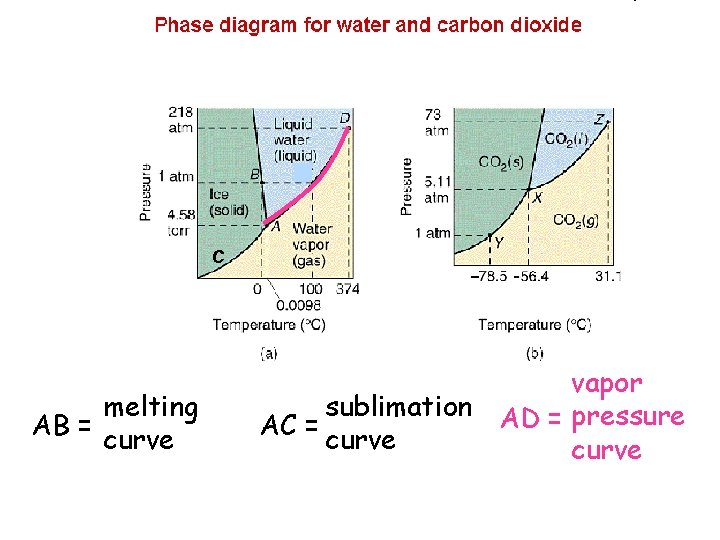
C melting AB = curve vapor sublimation AD = pressure AC = curve

C A triple point = Point ______ The point at which all 3 phases of a substance (solid, liquid, gas) can coexist at equilibrium. D critical point = Point ______ The combination of critical temperature and critical pressure. critical temp = temp. above which a gas cannot be liquefied. (H 2 O=374ºC) critical pressure = press. required to liquefy a gas at its critical temperature. (H 2 O=218 atm)

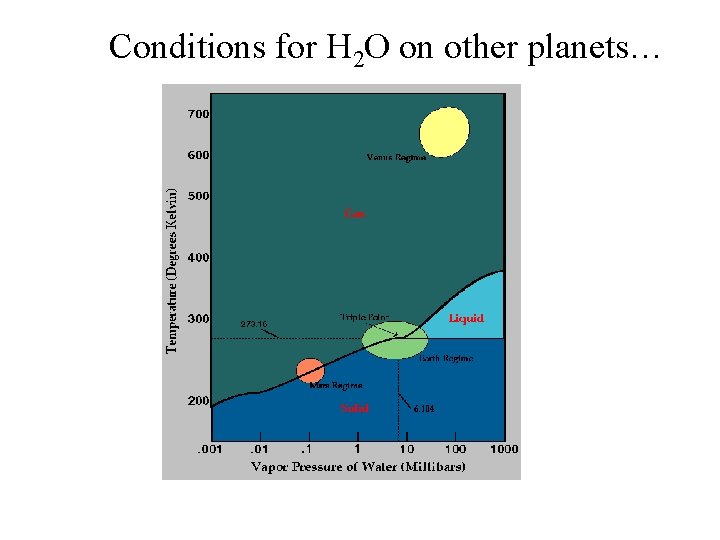
Conditions for H 2 O on other planets…