Thermodynamics Lecture Series Ideal Rankine Cycle The Practical















![Vapor Cycle – Reheat Rankine Cycle Energy Analysis where s 6 = [sf +xsfg]@P Vapor Cycle – Reheat Rankine Cycle Energy Analysis where s 6 = [sf +xsfg]@P](https://slidetodoc.com/presentation_image_h/cd5f8e993329bfa2df924db6ea05cb2a/image-37.jpg)

- Slides: 39

Thermodynamics Lecture Series Ideal Rankine Cycle –The Practical Cycle Applied Sciences Education Research Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@hotmail. com http: //www 5. uitm. edu. my/faculties/fsg/drjj 1. html

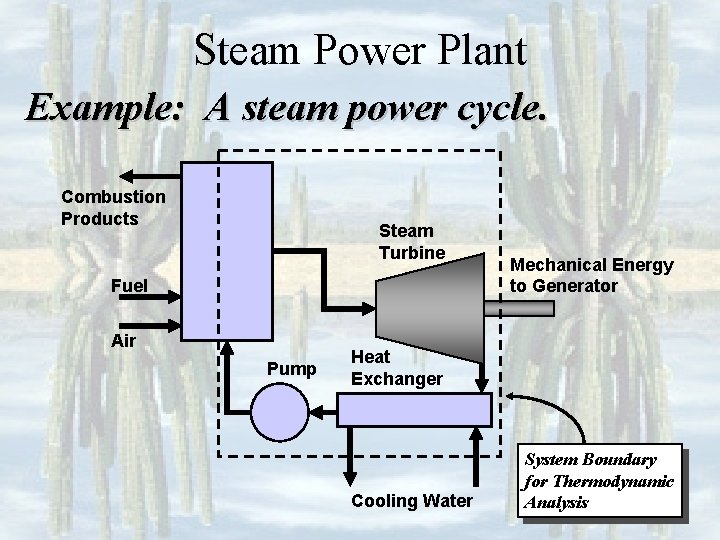
Steam Power Plant Example: A steam power cycle. Combustion Products Steam Turbine Fuel Air Pump Mechanical Energy to Generator Heat Exchanger Cooling Water System Boundary for Thermodynamic Analysis

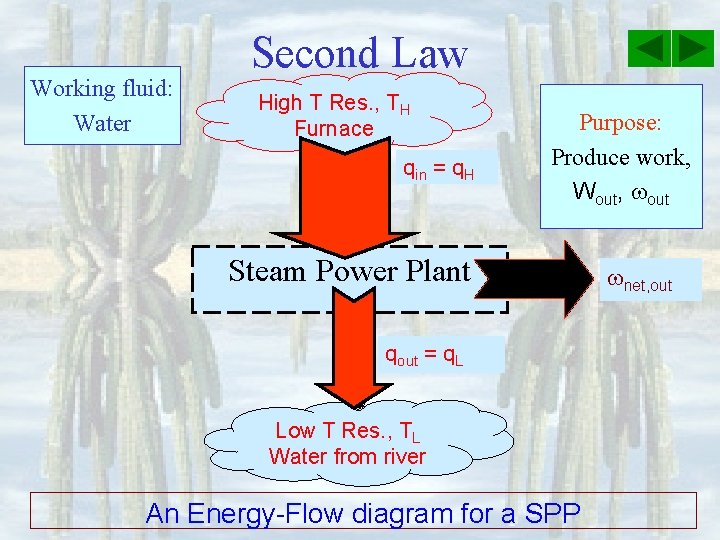
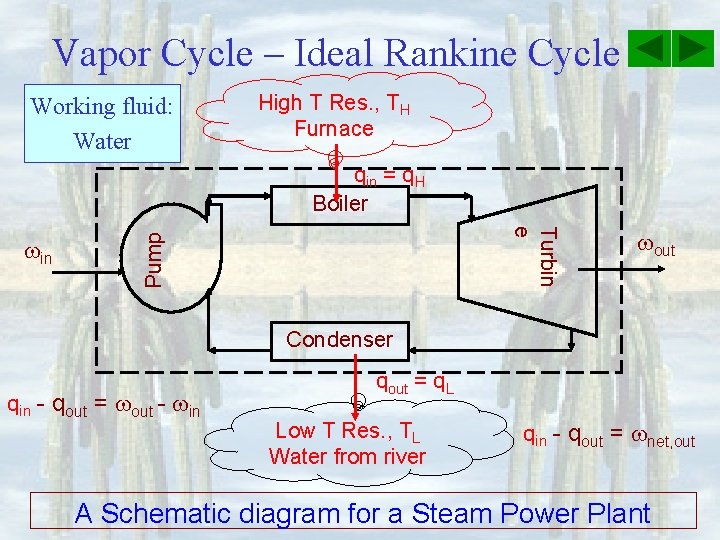
Working fluid: Water Second Law High T Res. , TH Furnace qin = q. H Purpose: Produce work, Wout, out Steam Power Plant qout = q. L Low T Res. , TL Water from river An Energy-Flow diagram for a SPP net, out

Second Law – Dream Engine What is the maximum performance of real engines if it can never achieve 100%? ? Carnot Cycle P - diagram for a Carnot (ideal) power plant P, k. Pa 1 qin 2 4 3 qout , m 3/kg

Second Law – Will a Process Happen Carnot Principles • For heat engines in contact with the same hot and cold reservoir v. P 1: 1 = 2 = 3 (Equality) v. P 2: real < rev (Inequality) Consequence Processes satisfying Carnot Principles obeys the Second Law of

Second Law – Will a Process Happen Clausius Inequality : • Sum of Q/T in a cyclic process must be zero for reversible processes and negative for real processes reversible impossible real

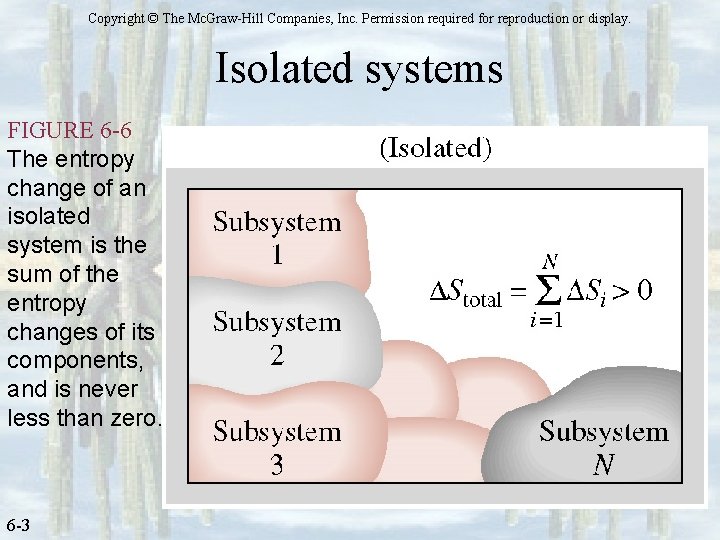
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Isolated systems FIGURE 6 -6 The entropy change of an isolated system is the sum of the entropy changes of its components, and is never less than zero. 6 -3

Entropy – Quantifying Disorder Increase of Entropy Principle – closed system The entropy of an isolated (closed and adiabatic) system undergoing any process, will always increase. For pure substance: and Then Surrounding System

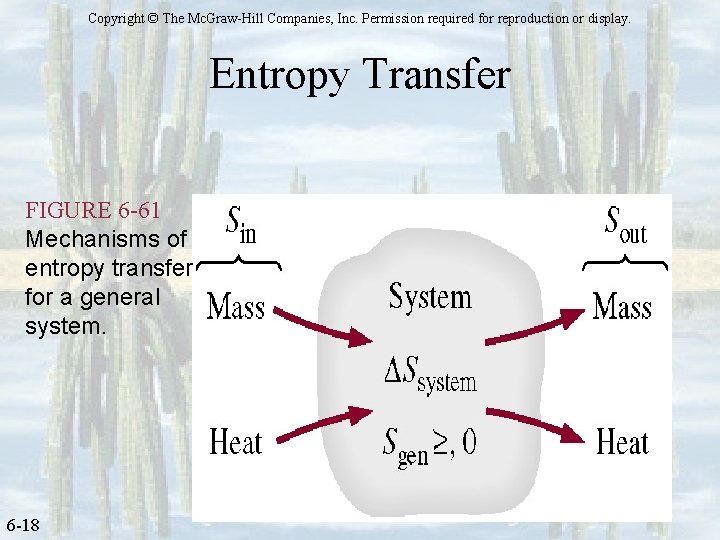
Entropy – Quantifying Disorder Entropy Balance – for any general system ØFor any system undergoing any process, ØEnergy must be conserved (Ein – Eout = Esys) ØMass must be conserved (min – mout = msys) ØEntropy will always be generated except for reversible processes v. Entropy balance is (Sin – Sout + Sgen = Ssys)

Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Entropy Transfer FIGURE 6 -61 Mechanisms of entropy transfer for a general system. 6 -18

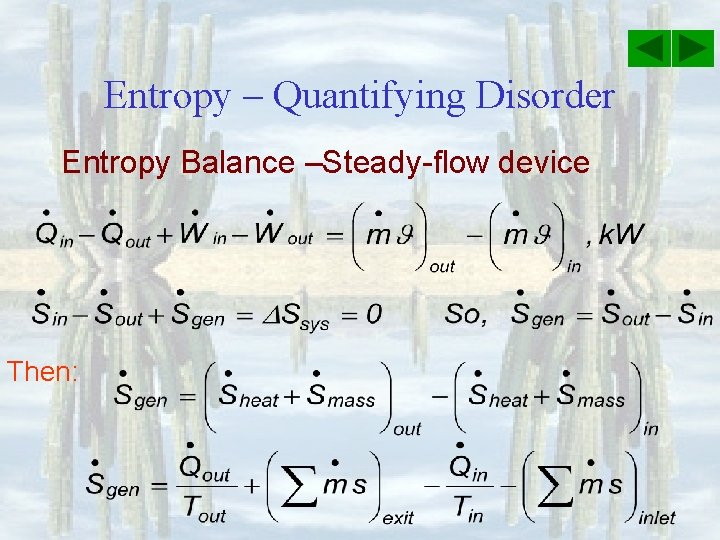
Entropy – Quantifying Disorder Entropy Balance –Steady-flow device Then:

Entropy – Quantifying Disorder Entropy Balance –Steady-flow device Turbine: Assume adiabatic, kemass = 0, pemass = 0 Entropy Balance where In, 3 Out

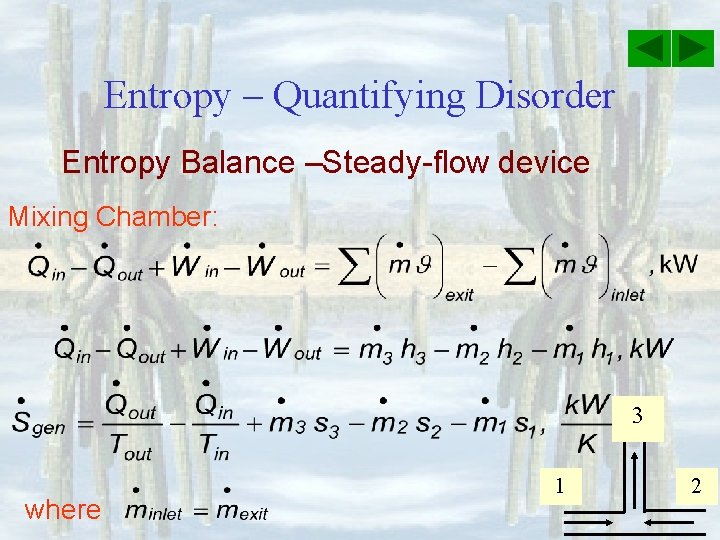
Entropy – Quantifying Disorder Entropy Balance –Steady-flow device Mixing Chamber: 3 where 1 2

Vapor Cycle Steam Power Plant ØExternal combustion ØFuel (q. H) from nuclear reactors, natural gas, charcoal ØWorking fluid is H 2 O Cheap, easily available & high enthalpy of vaporization hfg ØCycle is closed thermodynamic cycle ØAlternates between liquid and gas phase ØCan Carnot cycle be used for representing real SPP? ? ØAim: To decrease ratio of TL/TH

Vapor Cycle – Carnot Cycle Efficiency of a Carnot Cycle SPP

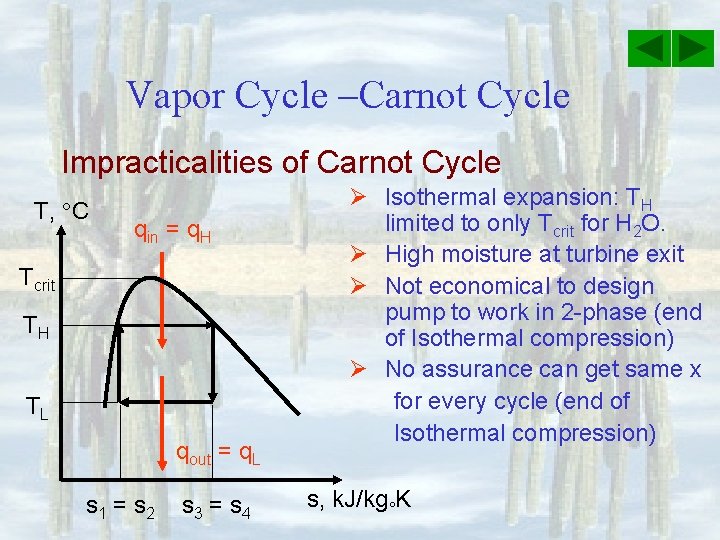
Vapor Cycle –Carnot Cycle Impracticalities of Carnot Cycle T, C qin = q. H Tcrit TH TL qout = q. L s 1 = s 2 s 3 = s 4 Ø Isothermal expansion: TH limited to only Tcrit for H 2 O. Ø High moisture at turbine exit Ø Not economical to design pump to work in 2 -phase (end of Isothermal compression) Ø No assurance can get same x for every cycle (end of Isothermal compression) s, k. J/kg K

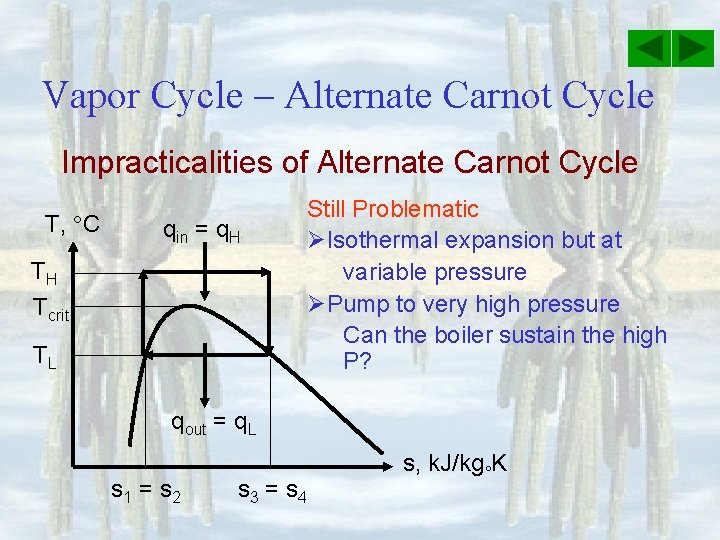
Vapor Cycle – Alternate Carnot Cycle Impracticalities of Alternate Carnot Cycle T, C qin = q. H TH Tcrit TL Still Problematic ØIsothermal expansion but at variable pressure ØPump to very high pressure Can the boiler sustain the high P? qout = q. L s 1 = s 2 s 3 = s 4 s, k. J/kg K

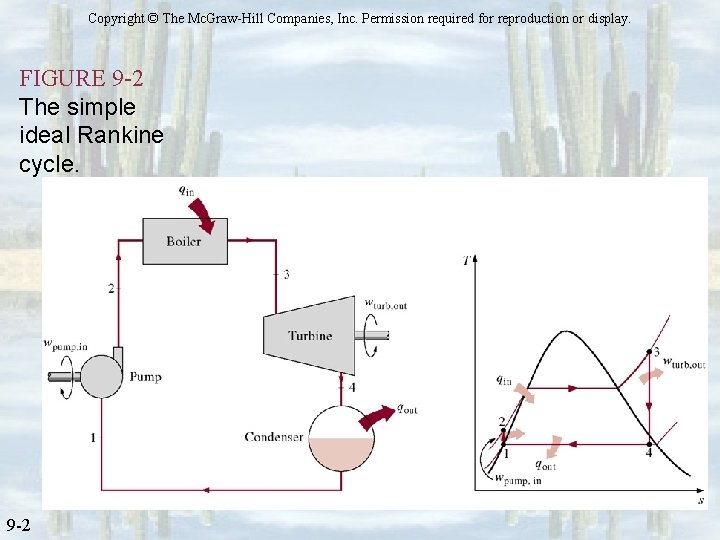
Vapor Cycle – Ideal Rankine Cycle Overcoming Impracticalities of Carnot Cycle ØSuperheat the H 2 O at a constant pressure (isobaric expansion) ü Can easily achieve desired TH higher than Tcrit. ü reduces moisture content at turbine exit ØRemove all excess heat at condenser ü Phase is sat. liquid at condenser exit, hence need only a pump to increase pressure ü Quality is zero for every cycle at condenser exit (pump inlet)

Vapor Cycle – Ideal Rankine Cycle Working fluid: Water High T Res. , TH Furnace qin = q. H Boiler Pump Turbin e in out Condenser qin - qout = out - in qout = q. L Low T Res. , TL Water from river qin - qout = net, out A Schematic diagram for a Steam Power Plant

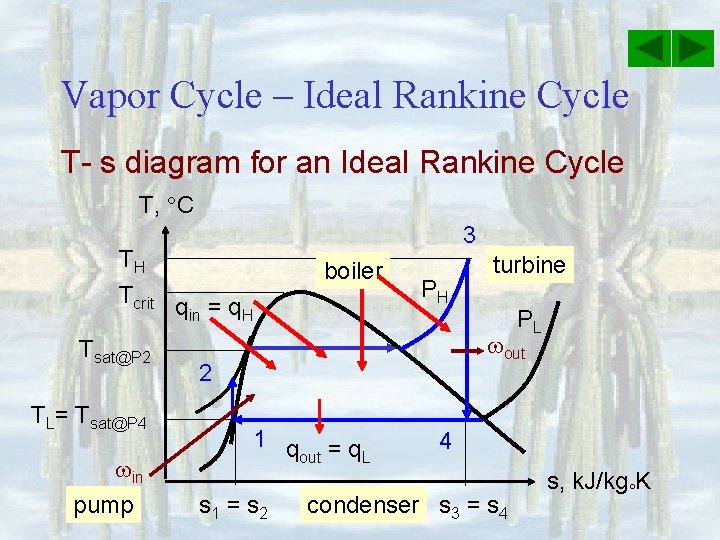
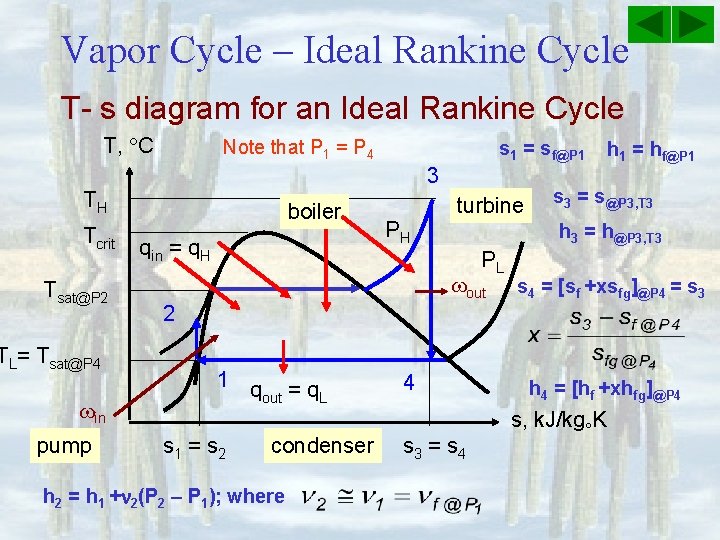
Vapor Cycle – Ideal Rankine Cycle T- s diagram for an Ideal Rankine Cycle T, C 3 TH Tcrit q = q in H Tsat@P 2 TL= Tsat@P 4 in pump boiler PH turbine PL out 2 1 q =q out L s 1 = s 2 4 condenser s 3 = s 4 s, k. J/kg K

Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 9 -2 The simple ideal Rankine cycle. 9 -2

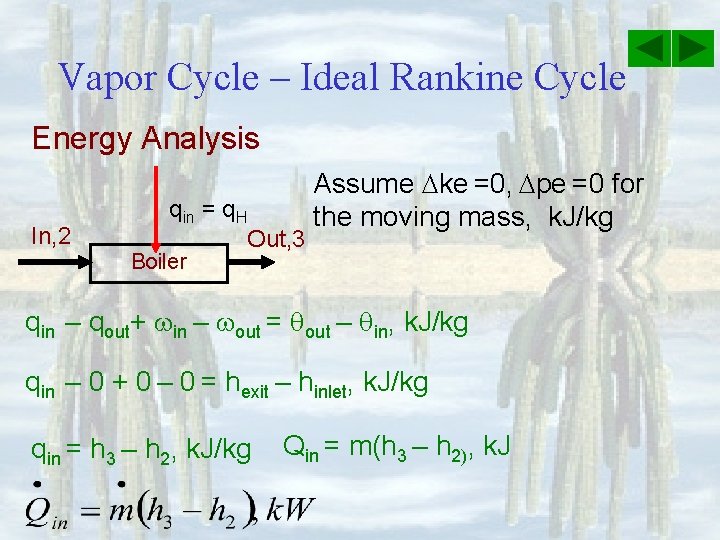
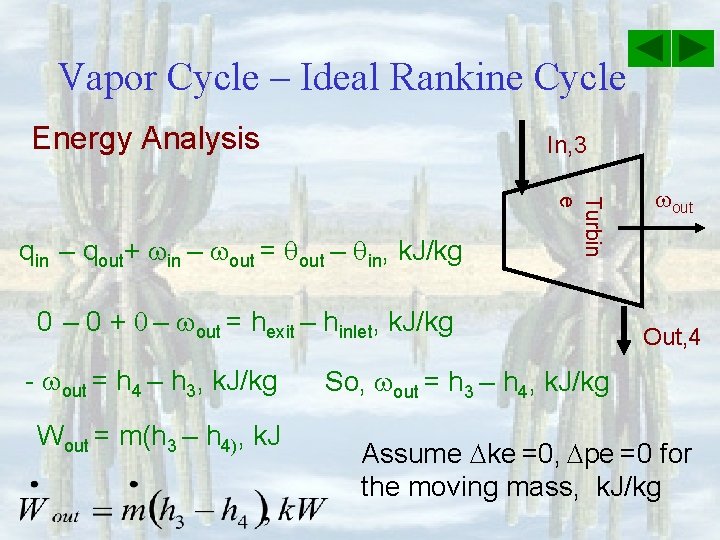
Vapor Cycle – Ideal Rankine Cycle Energy Analysis In, 2 qin = q. H Boiler Out, 3 Assume ke =0, pe =0 for the moving mass, k. J/kg qin – qout+ in – out = qout – qin, k. J/kg qin – 0 + 0 – 0 = hexit – hinlet, k. J/kg qin = h 3 – h 2, k. J/kg Qin = m(h 3 – h 2), k. J

Vapor Cycle – Ideal Rankine Cycle Energy Analysis Assume ke =0, pe =0 for the moving mass, k. J/kg qin – qout+ in – out = qout – qin, k. J/kg 0 – qout + 0 – 0 = hexit – hinlet Out, 1 Condenser In, 4 qout = q. L - qout = h 1 – h 4, So, qout = h 4 – h 1, k. J/kg Qout = m(h 4 – h 1), k. J

Vapor Cycle – Ideal Rankine Cycle Energy Analysis In, 3 Turbin e qin – qout+ in – out = qout – qin, k. J/kg 0 – 0 + 0 – out = hexit – hinlet, k. J/kg - out = h 4 – h 3, k. J/kg Wout = m(h 3 – h 4), k. J out Out, 4 So, out = h 3 – h 4, k. J/kg Assume ke =0, pe =0 for the moving mass, k. J/kg

Vapor Cycle – Ideal Rankine Cycle Energy Analysis qin – qout+ in – out = qout – qin, k. J/kg in 0 – 0 + in – 0 = hexit – hinlet, k. J/kg in = h 2 – h 1, k. J/kg For reversibl e pumps where So, Win = m(h 2 – h 1), k. J Pump Out, 2 In, 1

Vapor Cycle – Ideal Rankine Cycle Energy Analysis Efficiency

Vapor Cycle – Ideal Rankine Cycle T- s diagram for an Ideal Rankine Cycle T, C Note that P 1 = P 4 3 TH Tcrit Tsat@P 2 TL= Tsat@P 4 in pump s 1 = sf@P 1 boiler qin = q. H PH turbine h 1 = hf@P 1 s 3 = s@P 3, T 3 h 3 = h@P 3, T 3 PL out s 4 = [sf +xsfg]@P 4 = s 3 2 1 q =q out L s 1 = s 2 condenser h 2 = h 1 + 2(P 2 – P 1); where 4 s 3 = s 4 h 4 = [hf +xhfg]@P 4 s, k. J/kg K

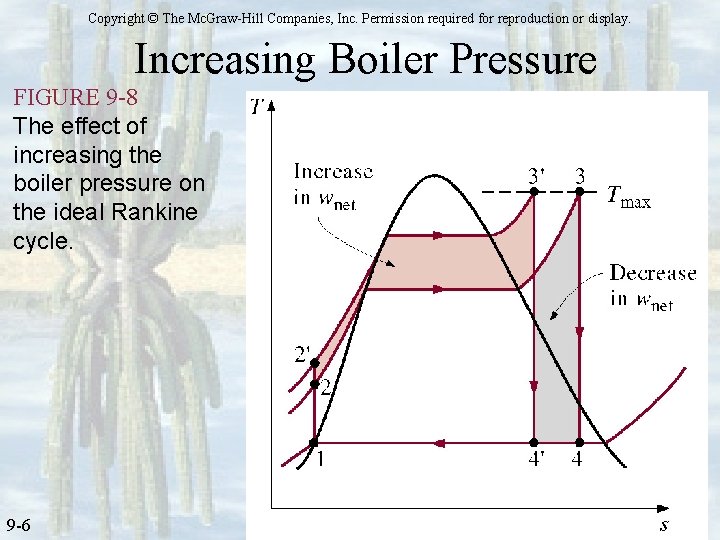
Vapor Cycle – Ideal Rankine Cycle Energy Analysis Increasing Efficiency ØMust increase net, out = qin – qout üIncrease area under process cycle ØDecrease condenser pressure; P 1=P 4 üPmin > Psat@Tcooling+10 deg C ØSuperheat üT 3 limited to metullargical strength of boiler ØIncrease boiler pressure; P 2=P 3 üWill decrease quality (an increase in moisture). Minimum x is 89. 6%.

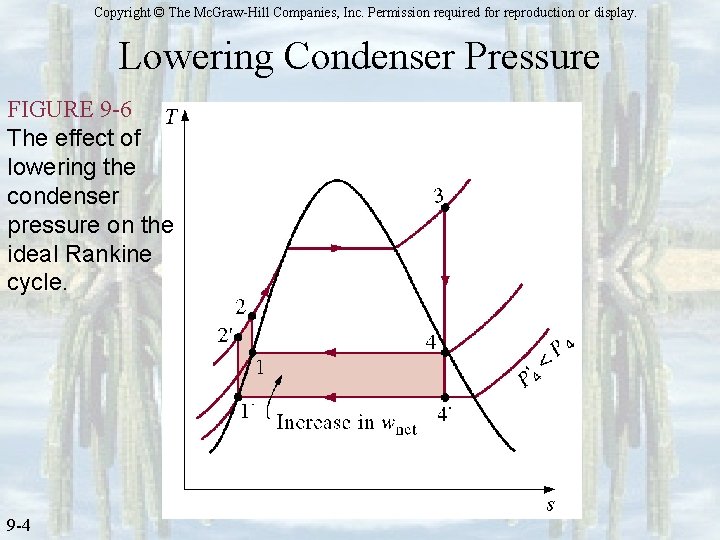
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Lowering Condenser Pressure FIGURE 9 -6 The effect of lowering the condenser pressure on the ideal Rankine cycle. 9 -4

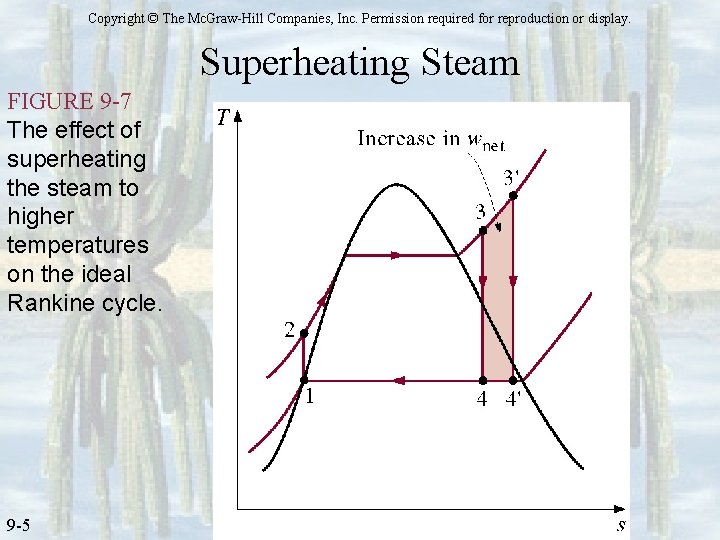
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Superheating Steam FIGURE 9 -7 The effect of superheating the steam to higher temperatures on the ideal Rankine cycle. 9 -5

Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Increasing Boiler Pressure FIGURE 9 -8 The effect of increasing the boiler pressure on the ideal Rankine cycle. 9 -6

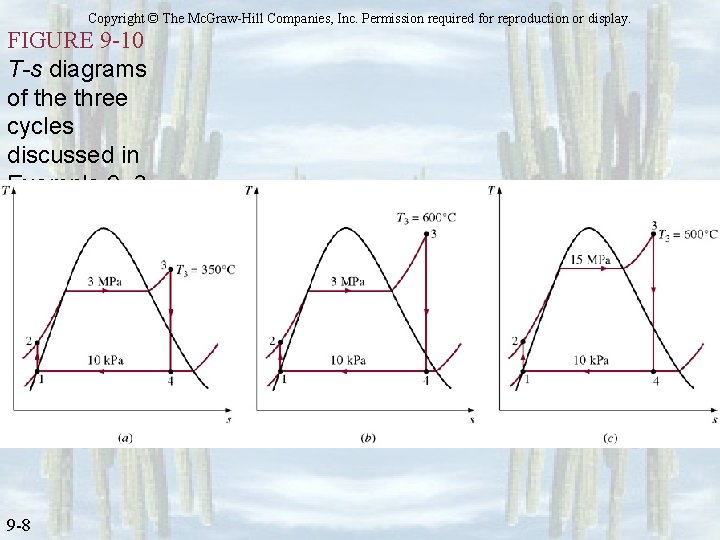
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 9 -10 T-s diagrams of the three cycles discussed in Example 9– 3. 9 -8

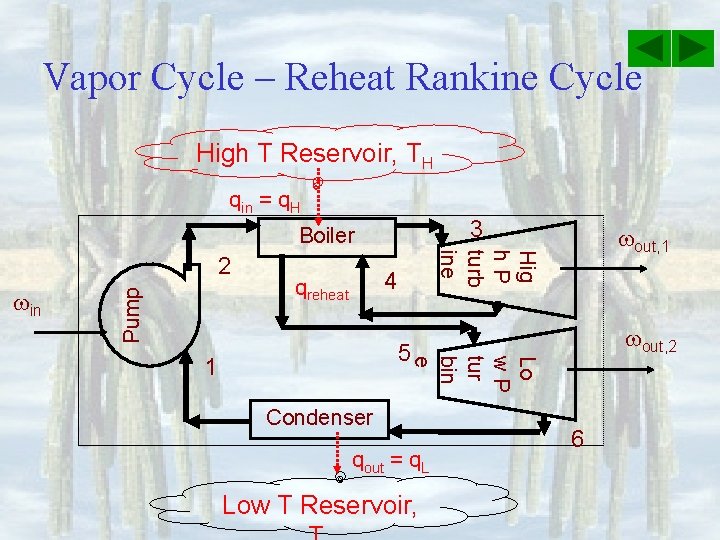
Vapor Cycle – Reheat Rankine Cycle High T Reservoir, TH qin = q. H 3 Boiler Pump in Hig h. P turb ine 2 4 qreheat out, 1 out, 2 5 Lo w. P tur bin e 1 Condenser qout = q. L Low T Reservoir, 6

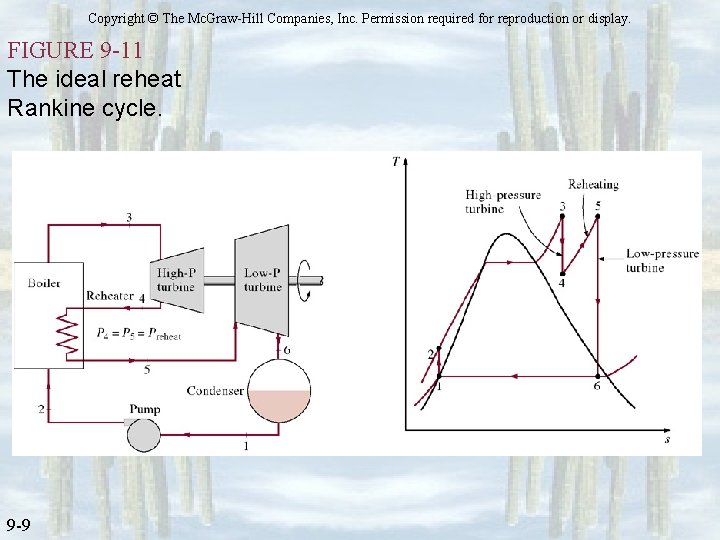
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 9 -11 The ideal reheat Rankine cycle. 9 -9

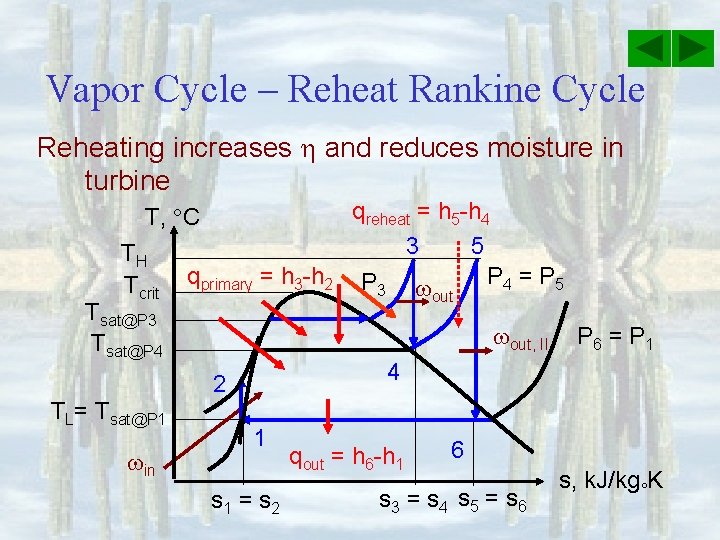
Vapor Cycle – Reheat Rankine Cycle Reheating increases and reduces moisture in turbine T, C TH Tcrit qprimary = h 3 -h 2 Tsat@P 3 Tsat@P 4 in out, II P 6 = P 1 4 2 TL= Tsat@P 1 qreheat = h 5 -h 4 5 3 P 4 = P 5 P 3 1 s 1 = s 2 qout = h 6 -h 1 6 s 3 = s 4 s 5 = s 6 s, k. J/kg K

Vapor Cycle – Reheat Rankine Cycle Energy Analysis q in = qprimary + qreheat = h 3 - h 2 + h 5 - h 4 qout = h 6 -h 1 net, out = out, 1 + out, 2 - in = h 3 - h 4 + h 5 - h 6 – h 2 + h 1
![Vapor Cycle Reheat Rankine Cycle Energy Analysis where s 6 sf xsfgP Vapor Cycle – Reheat Rankine Cycle Energy Analysis where s 6 = [sf +xsfg]@P](https://slidetodoc.com/presentation_image_h/cd5f8e993329bfa2df924db6ea05cb2a/image-37.jpg)
Vapor Cycle – Reheat Rankine Cycle Energy Analysis where s 6 = [sf +xsfg]@P 6. Use x = 0. 896 and s 5 = s 6 h 6 = [hf +xhfg]@P 6 Knowing s 5 and T 5, P 5 needs to be estimated (usually approximately a quarter of P 3 to ensure x is around 89%. On the property table, choose P 5 so that the entropy is lower than s 5 above. Then can find h 5 = h@P 5, T 5.

Vapor Cycle – Reheat Rankine Cycle Energy Analysis where s 1 = sf@P 1 h 1 = hf@P 1 s 3 = s@P 3, T 3 = s 4. h 3 = h@P 3, T 3 h 2 = h 1 + 2(P 2 – P 1); where P 5 = P 4. From P 4 and s 4, lookup for h 4 in the table. If not found, then do interpolation.

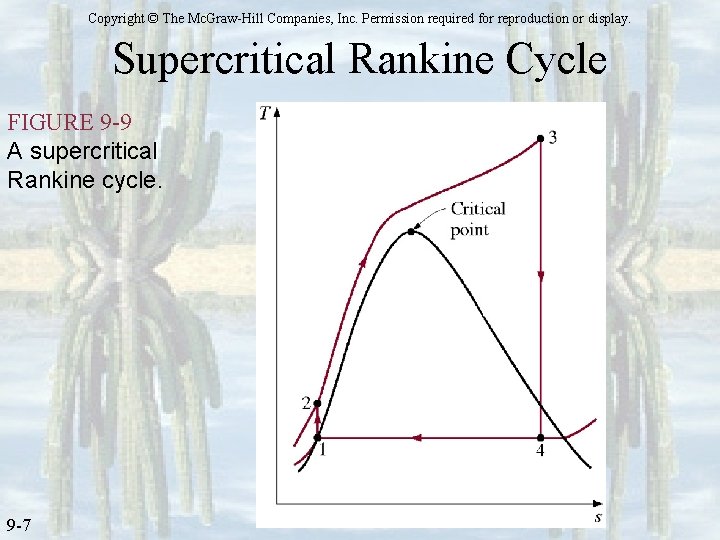
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Supercritical Rankine Cycle FIGURE 9 -9 A supercritical Rankine cycle. 9 -7