Nuclear Chemistry The weird world of the nucleus


























- Slides: 32

Nuclear Chemistry The weird world of the nucleus

Isotope Refresher l l Atoms of the same element all have the same number of protons Atoms of the same element may have different neutrons and therefore mass numbers (p+ + n 0)

Nuclear Instability Not all combinations of protons and neutrons are created equal l Some are more unstable than others. l If they are unstable they will do one of the following: l l Radioactive decay l Nuclear fission l Nuclear fusion

Nuclear “Reactions” l Nuclear “reactions” must still be balanced. Notice 238 = 4 + 234 “Mass balanced” l Notice 92 = 2 + 90 “Charge balanced” l

Radioactive Decay

Radioactive Decay l Radioactive decay – the nucleus of an atom undergoes a change so that it is no longer the same element l Decay is a totally random event. Nothing has an effect when an atom decays Two Main Types of Radioactive Decay 1. Alpha decay 2. Beta decay

Alpha Decay Alpha decay – emission of an alpha particle from the nucleus l Alpha (α) particle - a helium-4 nucleus l l Uranium-235, “enriched uranium, ” decays by alpha decay

Beta Decay Beta decay – emission of a beta particle from the nucleus l Beta (β) particle – electron l l Thorium-231 decays by beta decay

Practice Decay Reactions l Beta decay by cesium-137 l Alpha decay by polonium-210

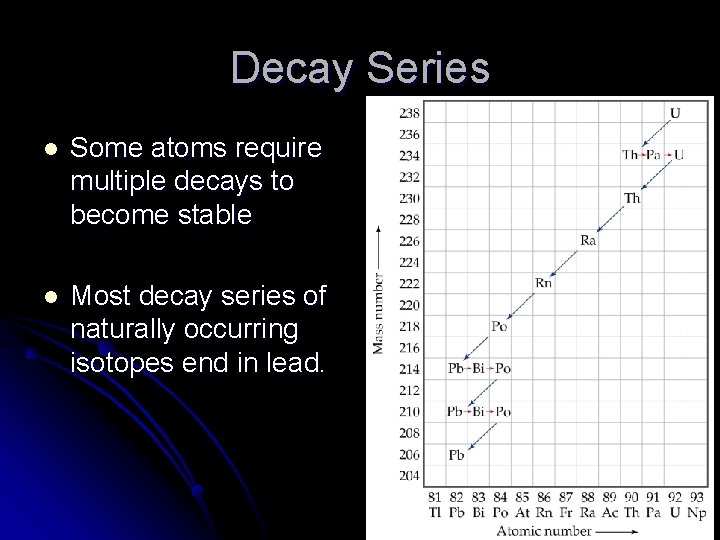
Decay Series l Some atoms require multiple decays to become stable l Most decay series of naturally occurring isotopes end in lead.


Half Life

Half Life l Half life – the amount of time it takes for ½ of a radioactive isotope to decay into something else. l Notice the atoms don’t disappear, they just change their identity.

Half Life Problem l l l A 100. 0 g radioactive sample decays for 5 hours. Only 12. 5 g of the original isotope remains after 5 hours. How long is the half life? 3 half lives = 5 hours 5 hours/3 half lives = 1. 67 hours/halflife Number of half lives Mass of sample Time 0 100. 0 g 0 1 50. 0 g 2 25. 0 g 3 12. 5 g 5 hours

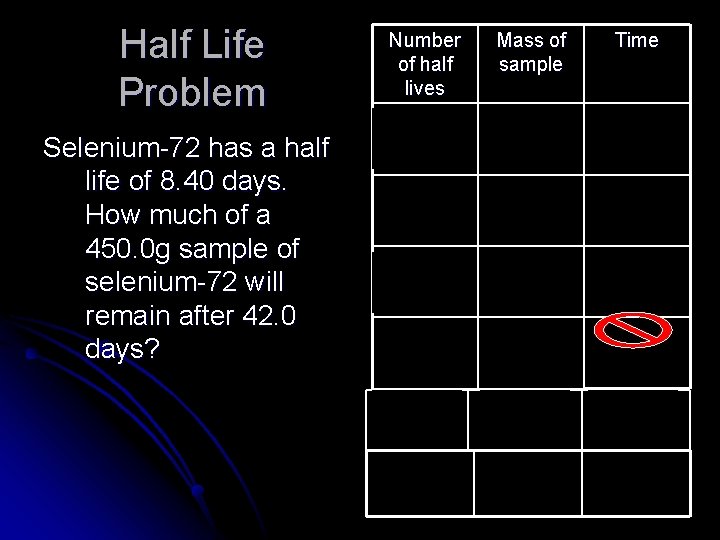
Half Life Problem Selenium-72 has a half life of 8. 40 days. How much of a 450. 0 g sample of selenium-72 will remain after 42. 0 days? Number of half lives Mass of sample Time 0 450. 0 g 0 1 225. 0 g 8. 40 days 2 112. 5 g 16. 8 days 3 56. 25 g 33. 6 days 25. 2 days 4 28. 13 g 33. 6 days 5 14. 06 g 42. 0 days

Radiocarbon Dating Technique l Uses the known half life of C-14 to estimate death of organic matter l Based on the known ratio of C-14 to C-12


Nuclear Fission and Fusion

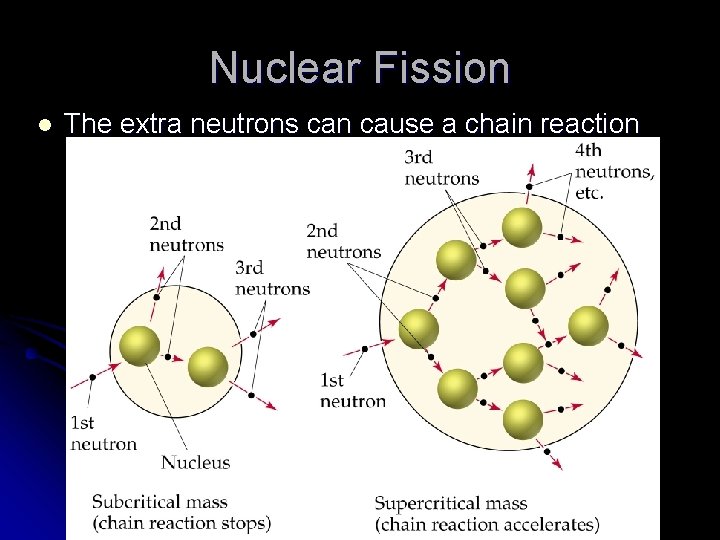
Nuclear Fission l Nuclear fission – one atom’s nucleus splits apart. A neutron strikes a nucleus causing it to split into small pieces l Releases lots of energy. l Extra neutrons are also produced. l

Nuclear Fission l The extra neutrons can cause a chain reaction

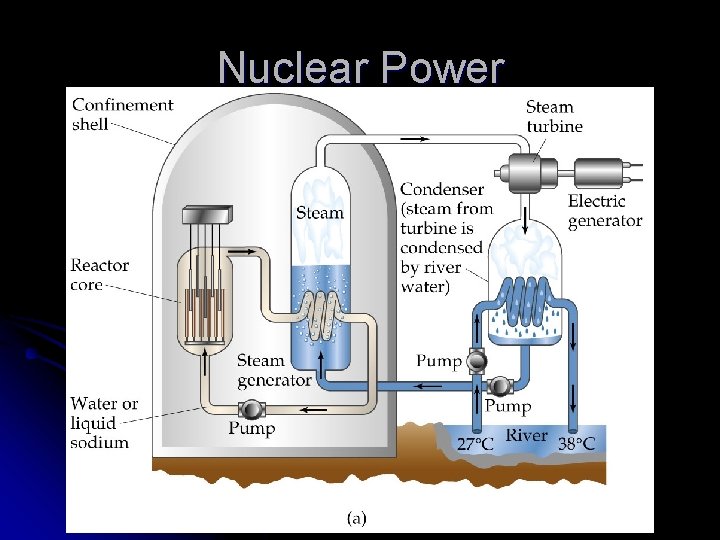
Uses of Nuclear Fission l Nuclear power l Energy produced by fission is transferred as heat to a coolant

Nuclear Power

Uses of Nuclear Fission Mushroom cloud from Nagasaki

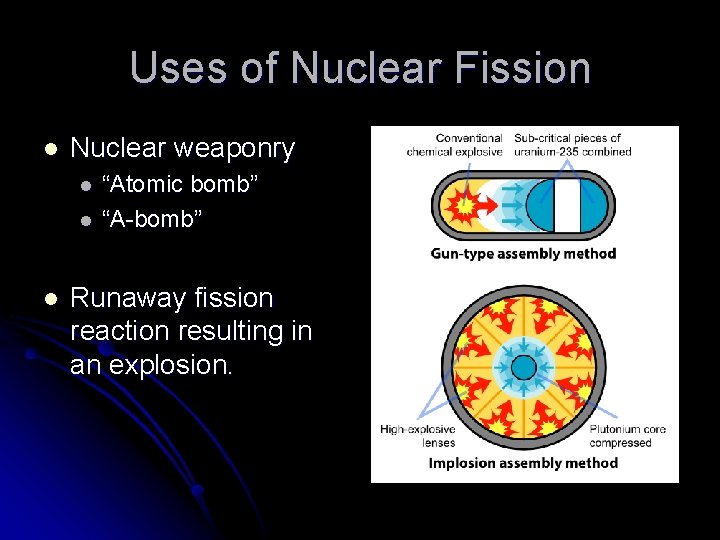
Uses of Nuclear Fission l Nuclear weaponry l l l “Atomic bomb” “A-bomb” Runaway fission reaction resulting in an explosion.


Nuclear Fusion l Fusion – combining of nuclei Above reactions describe fusion in the Sun. l Releases more energy than fission. l Also requires very high temperatures. l

Uses of Nuclear Fusion

Uses of Nuclear Fusion l Fusion weapons l l l Hydrogen bomb “H-bomb” Thermonuclear bomb Uses a fission reaction to start the fusion reaction “A-bomb” is the detonator for an “H-bomb”

Radiation

3 Primary Types of Radiation 1. Alpha radiation – α – alpha particles l l 2. Beta radiation – β –beta particles (electrons) l l 3. Low energy Result of alpha decay Low penetration Stop with a sheet of paper Higher energy Result of beta decay Some penetration ability Stop with several sheets of aluminum foil Gamma radiation – γ –high energy gamma rays l l Very high energy Can result from any type of decay Very highly penetrating and damaging Stop with several centimeters of lead or very thick concrete

3 Primary Types of Radiation

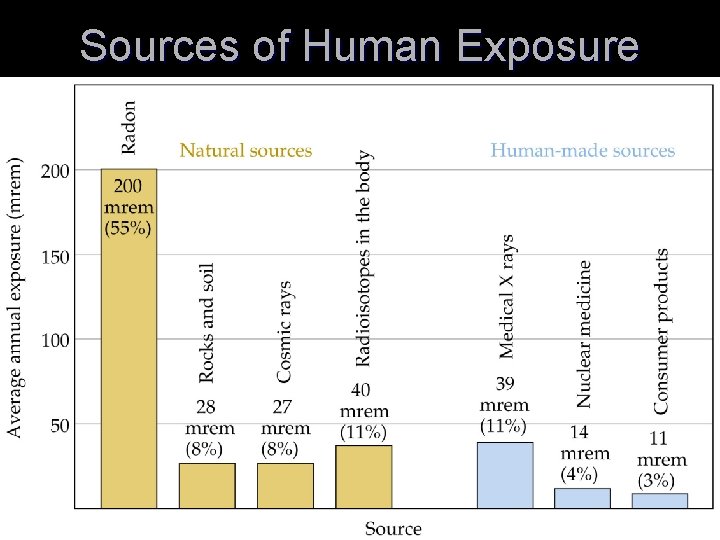
Sources of Human Exposure