Neonatal Neurology 1 Seizures HIE and Cooling CHO























































- Slides: 67

Neonatal Neurology 1: Seizures, HIE, and Cooling CHO NICU Lecture D Durand Revised 10/17/12

Incidence of Seizures • Higher in neonates than any other age group – 3/1000 live births • Most frequent in the first 10 days of life • Most frequent neonatal neurological problem

Pathophysiology • Excessive synchronous depolarization from large group of neurons Depolarization- Na influx into cell Repolarization- efflux of K out of cell • Disruption of Na/K ATP pump

Adverse Effects of Seizures • • • Disturbances in cerebral blood flow Cell division and migration Formation and expression of receptors Synaptogenesis and apotosis Long term effects: – Seizure threshold, – Learning and cognition

Why Are Neonates More Susceptible to Sz? • Increased Excitatory pathways: – Glutamate is the major excitatory neurotransmitter in CNS – Over-expression of glutamate receptors – Neonatal glutamate receptors are more permeable to Ca++, causing increased excitability • Decreased Inhibitory pathways: – GABA is principle inhibitory neurotransmitter – Neonate has decreased binding and expression of receptor

Clonic Seizures • Focal or multifocal, rhythmic movements with slow return movement • May be associated with generalized or focal brain abnormality • Most commonly associated with EEG seizures

Tonic Seizures • Sustained flexion or extension of one extremity or the whole body • Extensive neocortical damage with uninhibited subcortically generated movements • + EEG correlate – Brainstem release?

Nonepileptic Movements • Tremulousness or jitteriness • Stimulous evoked myoclonus from metabolic encephalopathies, CNS malformation • Stopped by gentle passive flexion • Benign sleep myoclonus

Benign Sleep Myoclonus • Onset 1 st week of life • Synchronous jerks of upper and lower extremities during sleep • No EEG correlate • Provoked by benzodiazepines • Ceases upon arousal • Resolves by 2 months • Good prognosis

Causes of Seizures • • Hypoxic ischemic encephalopathy (60%) Arterial and venous stroke (15%) Intracranial hemorrhage (5%) CNS infection (5%) Metabolic disorders (5%) Congenital anomalies (5%) Other (5%)

Diagnose Underlying Etiology • Maternal and labor history – cord gases, placental pathology • Neuroimaging – MRI, US, occasionally CT • R/O infection – CBC, blood cx, LP, viral PCR • Metabolic – Glucose, Electrolytes, Ca++ – NH 3, lactate, pyruvate – Serum AA, Urine OA, very long chain fatty acids

Meningitis/Encephalitis Causes 5% of Seizures • Viral: – TORCH, Enterovirus, Parvovirus – Sz 1 st 72 hrs • Bacterial: – GBS, Listeria, E coli, Strep pneumoniae – Presents at end of 1 st week to 3 months of age • Clues: – Maternal risk factors – Late onset after 1 st week of life

CNS Malformations Causes 5% of Seizures • Abnormal neuronal migration resulting in cortical dysgenesis – – Lissencephaly Pachygyria Polymicrgyria Linear Sebaceous Nevus syndrome • Often present later in infancy/childhood • Poor prognosis • Clues: – refractory seizures – severe encephalopathy – microcephaly

Metabolic Causes 5% of Seizures Etiology Clues • Glucose • Ca/Mg • ↑ or ↓ Na IDM, SGA, VLBW, IDM, CHD Dehydration, meningitis • Inborn Error Severe encephalopathy • Mitochondrial • Peroxisomal • Pyridoxine responsive Severe seizures Lactic acidosis Severe seizures, 1 st hours of life – – Amino acids Organic acids Ammonia Lactate/Pyruvate

Other Benign Causes 5% of Neonatal Seizures • Benign familial neonatal seizures – – Autosomal dominant Inter-ictal exam normal Good outcome Voltage-gated K+ channel • Benign idiopathic neonatal seizures – Term, normal birth – Normal inter-ictal state, EEG – Clonic, occur day 5, may be Zn deficiency

Other Malignant Causes 5% of Neonatal Seizures • Ohtahara syndrome (epileptic encephalopathy) – – 10 d -3 mo refractory seizures with burst suppression pattern underlying dysgenesis or metabolic disorder prognosis very poor

Neurologic Exam • Alertness – Lethargy, irritability • Spontaneous motor activity – Smooth, symmetric • Tone and muscle strength – Normal increased flexor tone – Muscle bulk • Cranial nerves – Corneal reflexes, facial symmetry, suck, swallow • Reflexes – DTR, clonus • Sensory – Response to touch

Neuroimaging • Ultrasound – IVH in premature infants • MRI – Developmental anomalies – Diagnostic and prognostic info for term infants with HIE • CT scan – Hemorrhagic lesions in term infants – Portable, radiation risk depends on urgency

Clinical Seizures Without EEG Correlate • May represent uninhibited brainstem reflexes • Discharges from deep cerebral structures and brainstem may not reach the cortical surface • Or may be normal movement – Myoclonic jerks

EEG Seizures Without Clinical Correlate • Occurs after treatment with anticonvulsants • Approximately 50% of EEG seizures not controlled by phenobarbital • Remember when using paralytic agents

Treatment • More difficult to suppress than in older children • Treatment is worthwhile because seizures: – May cause hemodynamic or respiratory compromise – Disrupt cerebral autoregulation – May result in cerebral energy failure and further injury

Treatment Options • Phenobarbitol • Dilantin • Kepra (? ) • Benzodiazepams

Prognosis by Etiology of Seizure • Chance of normal development: – Late hypocalcemia-100% – Subarachnoid hemorrhage-90% 50% normal outcome – HIE- 50% – Meningitis-50% – Hypoglycemia-50% – Severe IVH- 10% – Developmental anomaly- 0% Almost all are normal

Prognosis by EEG • • Severe background abnormalities –poor outcome Certain electrographic seizure types- poor outcome Status epilepticus > 30 minutes- poor outcome Normal background- good outcome

Hypoxemic Ischemic Encephalopathy & Asphyxia

ACOG & AAP “Asphyxia should be reserved to describe a neonate with all of the following: • Umbilical artery p. H < 7. 00 • Apgar 0 -3 for longer than 5 minutes • Neonatal neurologic manifestations – eg seizures, coma, hypotonia • Multisystem organ dysfunction. ” Guidelines for Perinatal Care, 5 th ed (2002)

Apgar Score • Developed in 1953 • Designed to help with decisions about clinical care, not prognosis • Poor correlation with long-term outcome – 1 minute Apgar does not correlate with outcome – 5 min Apgar poorly correlated with outcome

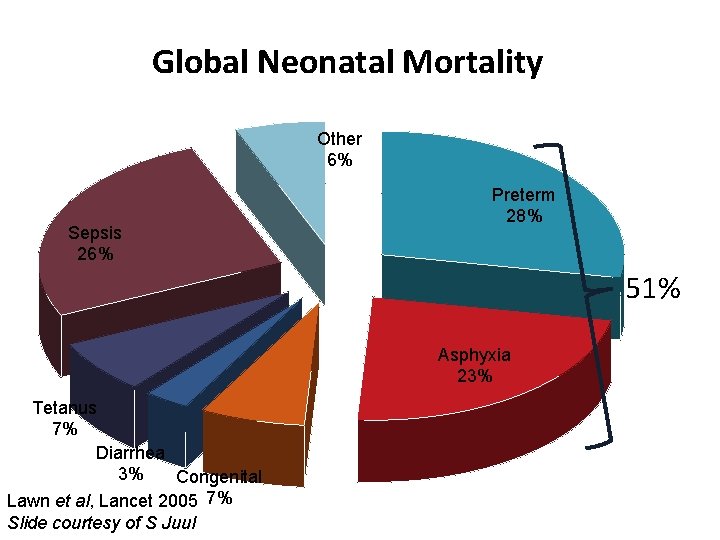
Global Neonatal Mortality Other 6% Sepsis 26% Preterm 28% 51% Asphyxia 23% Tetanus 7% Diarrhea 3% Congenital Lawn et al, Lancet 2005 7% Slide courtesy of S Juul

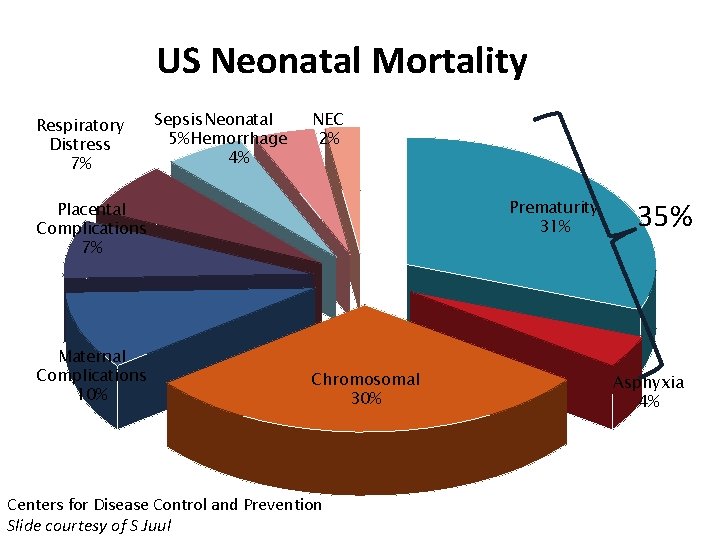
US Neonatal Mortality Respiratory Distress 7% Sepsis Neonatal 5% Hemorrhage 4% NEC 2% Prematurity 31% Placental Complications 7% Maternal Complications 10% Chromosomal 30% Centers for Disease Control and Prevention Slide courtesy of S Juul 35% Asphyxia 4%

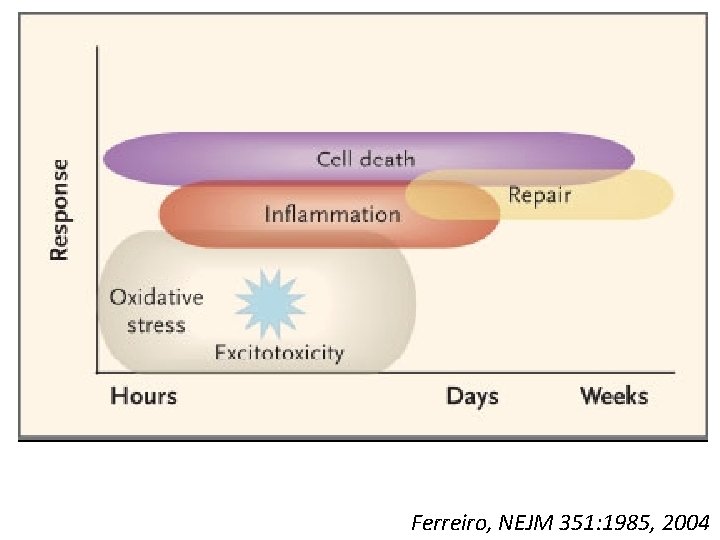
Early Cerebral Response to Asphyxia • Substrate levels fall – Decreased Oxygen and Glucose utilization • Decrease in ATP (energy failure) • Sets up a cascade leading to later necrosis and/or apoptosis

Pathogenesis of HIE: Secondary Neuronal Injury • • Occurs 6 -48 hours after initial injury Extracellular glutamate leads to “excitotoxicity” Intracellular Ca++ accumulation Free-radical formation Intracellular edema and membrane damage Loss of endothelial integrity Apoptosis and necrosis

Ferreiro, NEJM 351: 1985, 2004

Standard Therapies for Neonatal HIE • Basic support: – Maintain blood pressure in normal range – Maintain blood gases in normal range – Maintain electrolytes in normal range – Maintain blood glucose in normal range • Treat obvious seizures with anticonvulsants

Cerebral Hypothermia: Mechanisms of Action • Reduce cerebral metabolism • Reduce excessive accumulation of cytotoxins – e. g. glutamate, free radicals • Reduce inflammatory second messengers • Inhibit apoptosis and necrosis Gunn, Early Human Devel 1998; 53: 19

Cerebral Hypothermia: Depth of Cooling • 32 -34 degrees is protective • Below 32 -34 degrees, systemic complications become significant – Prolonged QT, bradycardia, decreased CO – Hyperviscosity – Platelet dysfunction – Decreased neutrophil function Gunn, Early Human Devel 1998, 53: 19

Cerebral Hypothermia: Animal Model • Near-term fetal sheep • Cerebral ischemia x 30 min • 5. 5 hrs later, randomized to cooling or sham cooling • Rx’d 72 hrs • Cooled animals did better – Improved EEG, decreased neuronal loss Gunn, Pediatrics 1998; 102: 1098

Cerebral Hypothermia: Animal Model • Animals did better if cooling started earlier – 90 min after cerebral ischemia • No protection seen after 8. 5 hours

Cool Cap Study “Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial” Gluckman et al, Lancet, 2005

Inclusion Criteria: History • Gestation > 36 wks • One of the following: – Apgar score < 5 at 10 minutes – Continued need for resuscitation at 10 min of age – Umbilical cord or arterial p. H < 7. 0 within 60 min of birth – Umbilical cord or arterial Base Deficit > 16 within 60 min of birth

Inclusion Criteria: Physical Exam • Moderate to severe encephalopathy • Altered state of consciousness and at least one of the following: – Lethargy, stupor, coma – Hypotonia – Abnormal reflexes – Absent or weak suck – Seizures

Inclusion Criteria: Screening a. EEG • Abnormal amplitude integrated EEG – 20 minute test – Abnormal background or seizures • a. EEG has significant positive predictive value for adverse outcome Toet, Arch Dis Child Fetal Neon 81: F 19, 1999 Naqeeb, Pediatrics 103: 1263, 1999 de. Vries, Arch Dis Child Fetal Neon 90: F 201, 2005

Cerebral Function Monitor (CFM) • • 3 lead amplitude integrated EEG (a. EEG) Easy to apply 20 minute tracing Easy to interpret

Cerebral Function Monitor

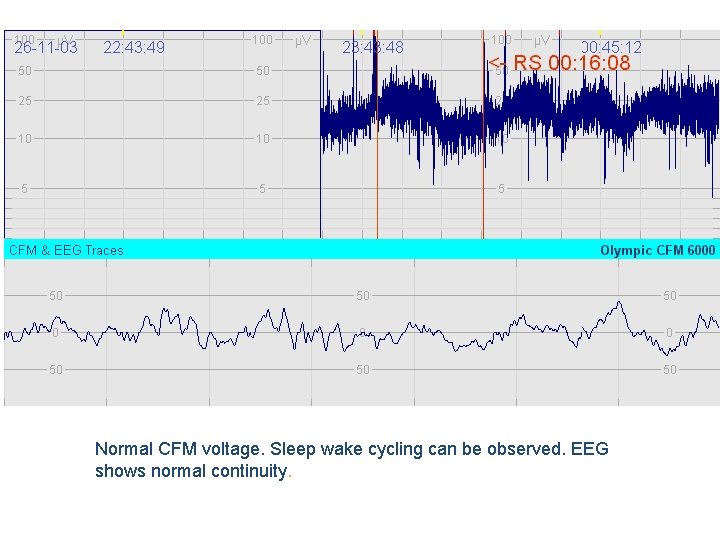
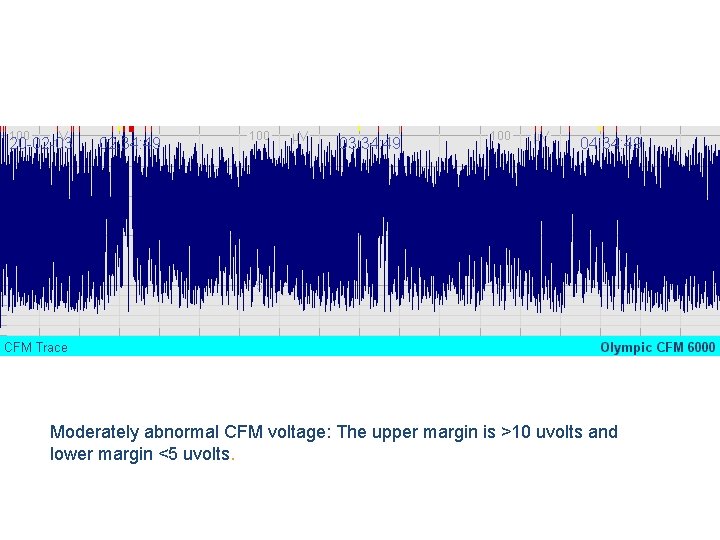
a. EEG Interpretation • Amplitude: – Normal lower border above 5 µv – Normal upper border above 10 µv – Lower amplitude means is abnormal • Seizures • Does not give information about regional abnormalities • Under diagnose subtle abnormalities

Normal CFM voltage. Sleep wake cycling can be observed. EEG shows normal continuity.

Moderately abnormal CFM voltage: The upper margin is >10 uvolts and lower margin <5 uvolts.

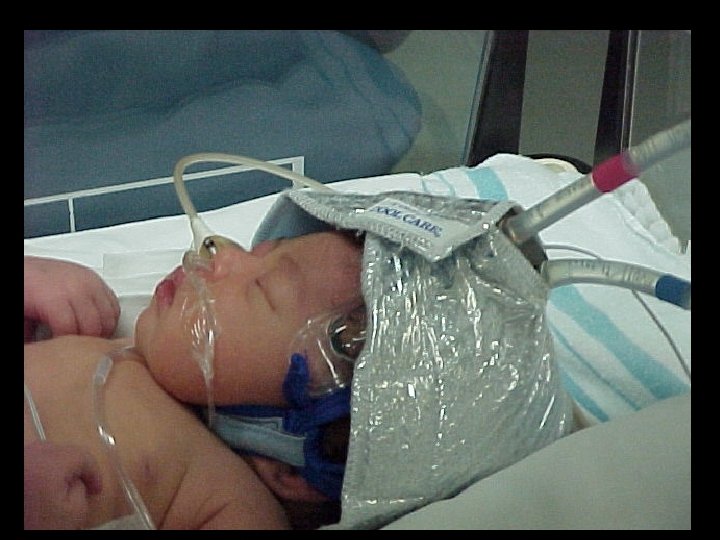
Cerebral Cooling Technique • Goal: Maximal cerebral cooling with only mild core cooling • Cool water (approx 10 degrees) circulates through cap • Radiant warmer adjusted to maintain rectal temp 34 -35 degrees • Both cooling and re-warming done gradually – 0. 5 degree/hour


Results • 234 infants randomized – Moderate to severe encephalopathy in > 90% – Seizures in > 50% • 18 month follow up data on 218 (93%) • Death or severe disability at 18 months – Cooled 55% – Controls 66% – P = 0. 1

Sub-Analysis • Moderate encephalopathy at entry (n=172) • Death or severe disability at 18 months – Cooled 48% – Controls 66% – P = 0. 02 • Severe encephalopathy at entry (n=46) • Death or severe disability at 18 months – Cooled 79% – Controls 68% – P = 0. 5

Whole Body Cooling NICHD Network • • Cooling blankets Esophageal temperature 33. 5 o. C No radiant heat 72 hrs cooling, 6 hrs re-warming Shankaran, N Engl J Med 2005, 353: 1574


Whole Body Cooling NICHD Network • 208 patients randomized • 18 -24 month outcome data for 205 • Death – 24% in Cooled Group – 37% in Control Group – P=0. 08 • Death or moderate/severe disability – 44% in Cooled group – 62% in Control group – P=0. 01 Shankaran, N Engl J Med 2005, 353: 1574

Current US Consensus • Cooling improves outcome from HIE – No longer experimental • Selective cerebral cooling and whole body cooling are (probably) similarly effective • Cooling should (probably) be offered to patients with either moderate or severe HIE • Cooling should only be done as part of an organized program

Cooling is Not Easy! • Careful servo of cooling device to keep temperature in narrow range • With over-cooling: – Pulmonary hypertension – Bradycardia • With re-warming: – Vasodilation – Hypotension

Avoid Hyperthermia! • Reviewed patients in NICHD (whole body) trial • Control (un-cooled) arm • Risk of death or severe disability increased with highest temp: – For each increase in temp by 1 o. C, risk increased by 4 x Laptook, Pediatrics 2008; 122: 491

Passive Cooling • • • Begin at delivery hospital Do not turn on radiant warmer Measure rectal temperature every 15 minutes Maintain rectal temperature 33 -35 °C Watch for complications of cooling: – Pulmonary hypertension – Systemic hypotension

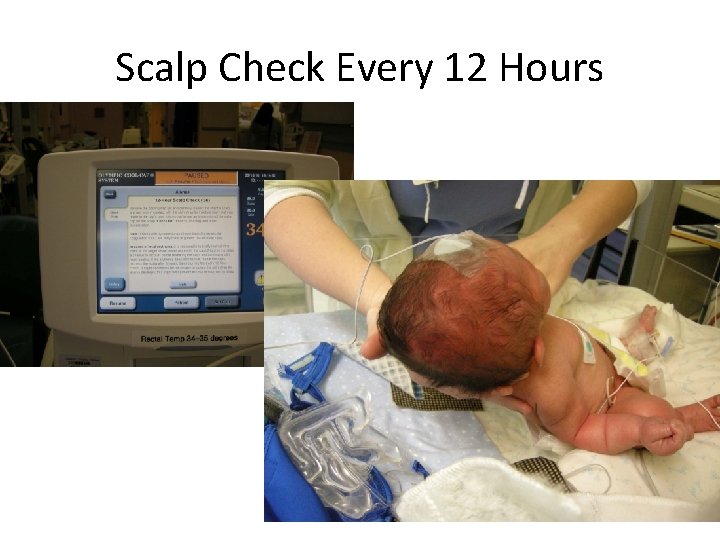
Scalp Check Every 12 Hours

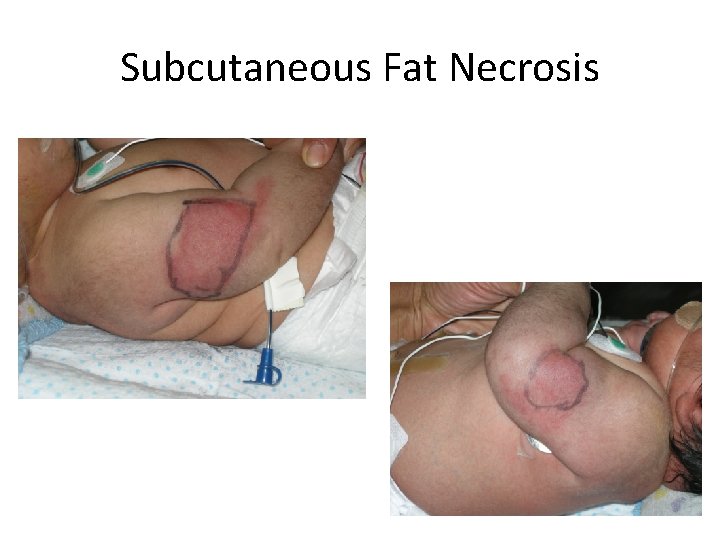
Subcutaneous Fat Necrosis

Passive Cooling • • • Begin at delivery hospital Do not turn on radiant warmer Measure rectal temperature every 15 minutes Maintain rectal temperature 34 -35 °C Watch for complications of cooling: – Pulmonary hypertension – Systemic hypotension

Cooling On Transport • • • Retrospective review from U. Virginia All patients 2005 -2009 40 out-born 35 cooled on transport 34% were “over-cooled” to < 32 o. C Fairchild, J Perinatology 2009

Other Neuroprotective Rx? • Mg++ – Small trial of post-natal Rx in term HIE Bhat, Pediatrics 2009; 123: e 764 • Allopurinol – Animal data encouraging – Most data focused on prenatal administration to mother • Xenon – Animal data very encouraging – Given by inhalation to intubated mother – Expensive! • Stem Cells • Erythropoietin (Epo)

Erythropoietin – Not Just for RBCs • Erythropoietin Receptor (Epo. R) is expressed by multiple cell types • Somatic – – Erythrocytes Endothelial cells Myocardiocytes Enterocytes • Nervous System – – – Neurons Oligodendrocytes Schwann cells Astrocytes Microglia

Endogenous Erythropoeitin in Brain • Epo is required for normal brain development • Epo and Epo. R are expressed in the human fetal brain by 7 weeks gestation • Epo. R is expressed in mouse neural tube neuroepithelium, and in neural crest and mesenchyme-derived cells • Epo expression follows by 0. 5 – 1 d • Epo production in brain is increased by hypoxia

Erythropoietin Safety • Extensively studied in preterm infants for RBC production and neuroprotection – More than 30 clinical trials – More than 2500 babies – No significant safety issues – Still some concerns about ROP in preemies • Widely used in preterm and term infants to prevent or treat anemia

Erythropoietin Neuroprotection Animal Studies • Erythropoietin is neuroprotective: – Benefit in short-term and long-term studies – Protects both white matter and gray matter • Multiple mechanisms: – – – – Decreases glutamate toxicity Decreases oxidative injury Decreases NO mediated injury Decreases inflammation Decreases apoptosis Promotes cell growth Promotes angiogenesis

Erythropoietin for HIE Early Clinical Trials • 167 term patients with mod-severe HIE randomized to Erythropoietin vs Placebo (without cooling) • Erythropoietin patients did better Zhu, Pediatrics 2009; 124: e 218 • Non-randomized study, showed term infants with HIE who received Erythropoietin had improved neurologic outcome at 6 months Elmahdy, Pediatrics 2010; 25: e 1135 • NEAT Trial (Neonatal Erythropoietin in Asphyxiated Term Infants) – Dose escalation and safety trial